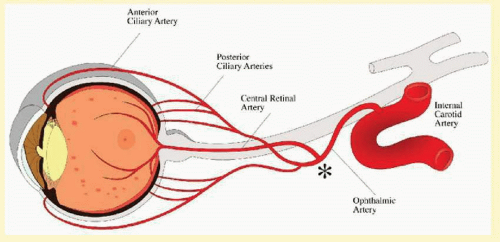
The ocular ischemic syndrome refers to the constellation of ophthalmic features that result from chronic hypoperfusion of the entire arterial supply to the eye, including the central retinal, the posterior ciliary, and the anterior ciliary arteries. In clinical practice, the occlusion is seen most frequently at the level of the carotid artery, but it may occur anywhere proximal to the point where the central retinal and the ciliary arteries branch from the ophthalmic artery (
Fig. 12.1).
The ocular ischemic syndrome is most frequently caused by atherosclerosis of the carotid artery. Less commonly, signs of ocular ischemia may be part of a larger clinical picture, such as the aortic arch syndrome, which in turn is most often caused by Takayasu arteritis
1,
2 and
3 or giant cell arteritis. Rarely, a chronic subtype of ophthalmic artery stenosis may result in the ocular ischemic syndrome. Ocular ischemia has also been associated with diabetes mellitus
4 and the antiphospholipid antibody syndrome, presumably due to the increased risk of thrombosis within the ophthalmic artery.
5,
6The clinical presentation of the ocular ischemic syndrome typically involves patients over the age of 50 years, more frequently men than women, who relate a loss of vision in one or both eyes over a period of weeks to months.
7,
8 There may or may not be associated ocular or periocular pain, and one may elicit a history of difficulty adjusting from bright light to relative darkness. Additionally, patients may relate a history of transient focal neurologic deficits.
The clinical signs of chronic ocular ischemia are legion, yet often subtle. Classic features include midperipheral fundus hemorrhages and dilated (but usually not tortuous) retinal veins.
9,
10 However these classic features are not always present, and it can easily be confused with diabetic retinopathy, central retinal vein occlusion (CRVO), or even idiopathic iritis.
11 Because of its broad presentation, the ocular ischemic syndrome is probably underdiagnosed. Yet its detection often has important implications for the patient, both ophthalmologically and systemically.
12,
13In 1963, Kearns and Hollenhorst
3 reported the ocular symptoms and signs that occurred secondary to severe carotid artery obstruction. At that time they called the disease venous stasis retinopathy, presumably due to the venous dilation and the intraretinal hemorrhages, which in some ways resemble a mild case of CRVO. This term is confusing, since it has, in fact, later been used by certain authors to indicate mild, or nonischemic, CRVO,
14 an entity that is different in origin and clinical manifestations from the ocular ischemic syndrome. It is therefore recommended that the term venous stasis retinopathy be avoided altogether. Other terms that have been applied to describe the ocular condition occurring secondary to marked carotid artery stenosis include ischemic ocular inflammation
11,
15,
16 and ischemic oculopathy.
16Because atherosclerosis accounts for most cases of the ocular ischemic syndrome, it is not surprising that most patients are first diagnosed in their 50s or 60s. The mean age at the time of presentation is approximately 65 years.
7,
11,
15,
16,
17 and
18 In fact, a patient with manifestations of ocular ischemia under the age of 50 years should raise the suspicion of some other underlying systemic condition, such as large vessel arteritis or an abnormality of lipid metabolism causing premature atherosclerosis.
The true incidence of the ocular ischemic syndrome is unknown. The ocular signs are not always obvious and, when present, may mimic other ocular disorders such as diabetic retinopathy and retinal vein obstruction. Kearns and Hollenhorst
3 noted this entity in 5% of their patients with internal carotid artery obstruction. Sturrock and Mueller
7 found six affected patients over a 2-year period in an English hospital serving a population of approximately 400,000, an estimated incidence of 7.5 per million.
Histopathologic findings in eyes with ocular ischemic syndrome due to carotid occlusion are consistent with retinal capillary endothelial damage from chronic hypoperfusion. Histologic examination demonstrates intraretinal hemorrhages and numerous microaneurysms, particularly in the retinal midperiphery. The retinal capillaries demonstrate a decrease of the pericyte-to-endothelial cell ratio.
19 The ophthalmoscopic appearance of midperipheral intraretinal hemorrhages has been reproduced in a rat model after bilateral carotid artery ligation.
20
SYMPTOMS
Visual loss is the most frequent symptom encountered in the ocular ischemic syndrome, seen in over 90% of affected people at the time of presentation (
Table 12.1).
7 In most cases, this loss of vision is gradual, occurring over a period of weeks to months. It is caused by chronic posterior segment ischemia, macular edema, and/or cataract occurring secondary to anterior segment ischemia.
Less commonly, abrupt loss of vision may occur due to severe hypoperfusion of the retinal arterial system, marked by a cherry-red spot in the macula. In some cases, this hypoperfusion is caused by an embolus from a carotid atherosclerotic plaque that occludes the central retinal artery. In other cases, the perfusion pressure of the central retinal artery falls below that of the intraocular pressure because of an abnormally low arterial perfusion pressure from carotid artery occlusion, or from an abnormally high intraocular pressure from neovascular glaucoma, or both.
Sometimes the cause of reduced vision is not immediately obvious. There is no cataract, no macular edema, and no signs of retinal ischemia, and the intraocular pressure is normal. In these eyes, fluorescein angiography usually reveals impaired choroidal perfusion, which is the probable cause of reduced vision. Color Doppler studies of the orbital vessels have correlated poor visual acuity with absence of blood flow in the posterior ciliary arteries,
21 supporting the theory that choroidal hypoperfusion is the cause of poor vision in many patients.
A history of amaurosis fugax may be elicited in up to 10% of patients.
7 These episodes of transient loss of vision are caused by fibrin-platelet emboli originating from the atherosclerotic carotid plaques, which may temporarily occlude part of the retinal arterial circulation before being resorbed.
In 10% of patients diagnosed with the ocular ischemic syndrome, there is an absence of any visual symptoms.
7 In some cases, the detection of the ocular ischemic syndrome may be the patient’s first indication of underlying systemic problems such as atherosclerosis, arterial hypertension, or hypercholesterolemia, as well as the first indication of the patient’s risk for cerebrovascular accident and myocardial infarction.
Persons with ocular ischemic syndrome will often relate a difficulty in seeing after exposure to bright light.18 Typically, the patient will complain of prolonged difficulty in adjusting to a darker indoors when moving from a brightly lit outdoors. This subjective complaint can be more objectively tested with the photopic stress test, which measures the amount of time the patient requires to recover visual function following exposure to a bright light source.
The symptom of pain is found in about 40% of persons with the ocular ischemic syndrome.
7 Typically, the pain is described as a dull ache over the eye or brow. Ischemia to the anterior segment structures, or ocular angina, is thought to be the cause of this pain in some cases. In more advanced cases, an elevated intraocular pressure from neovascular glaucoma may cause the patient pain due to an increased perfusion gradient to the anterior segment causing ischemia, or due to rupture of a corneal epithelial microcyst of acute glaucoma. Ipsilateral dural ischemia has also been proposed as a cause of the pain.
SIGNS
The ocular ischemic syndrome may present with any of a constellation of signs in both anterior and posterior segments of the eye (
Table 12.2). The classic triad includes midperipheral dot hemorrhages, dilated retinal veins, and iris neovascularization. Yet, in its early stages, none of these features may be present, and the diagnosis may rely on the recognition of other clues, such as the elicitation of central retinal artery pulsations with gentle digital pressure on the globe.
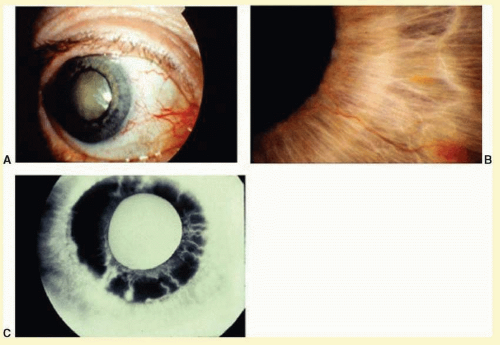
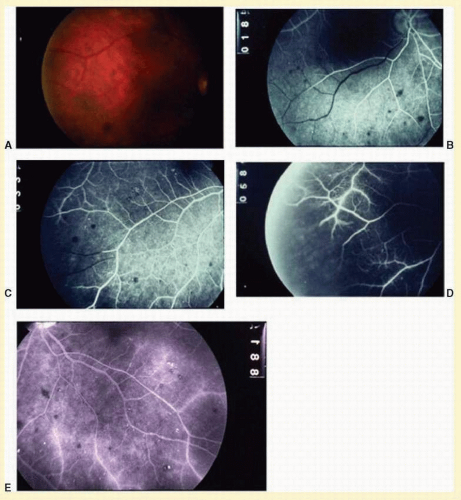
Eyes with ocular ischemic syndrome appear to be at high risk for rubeosis iridis, found in 67% of eyes at the time of presentation
7 (
Figs. 12.2 and
12.3). By comparison, only 2.8% of nonischemic CRVOs, 58% of ischemic CRVOs,
22 and 15% to 20% of central retinal artery occlusions will develop neovascularization of the iris.
23,
24 When the clinician faces a patient with rubeosis iridis and no obvious etiology (there are no signs of diabetic retinopathy or retinal vascular occlusion), this should prompt consideration of the ocular ischemic syndrome. The high incidence of rubeosis is probably related to the combined ischemia of both anterior and posterior segments of the globe.
The risk of neovascular glaucoma is likewise very high; 35% of patients at the time of presentation will have neovascularization of the iris and an intraocular pressure >22 mm Hg.
7 It is interesting to note that in some cases, because of poor ciliary body perfusion and decreased aqueous production, the intraocular pressure remains low even when fibrovascular tissue completely occludes the iridocorneal angle. Eyes with normal or well-controlled intraocular pressures have been reported to experience a dramatic rise in intraocular pressure after carotid endarterectomy, and it is therefore recommended that eyes be followed closely immediately following carotid surgery.
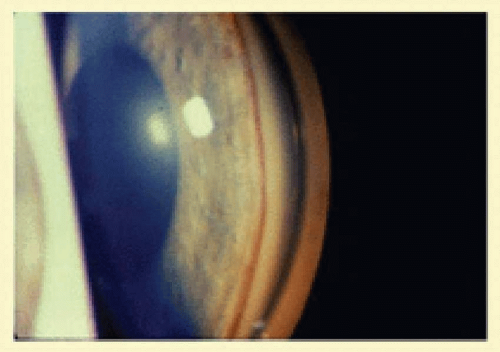
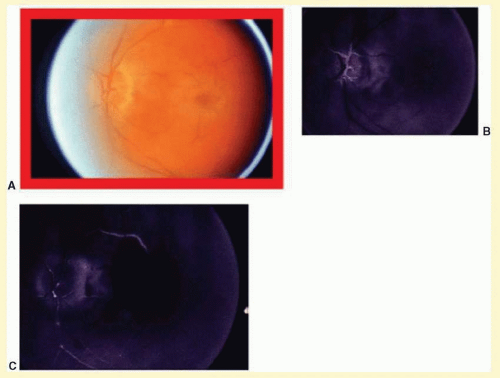
Anterior segment ischemia may cause conjunctival and episcleral vessel injection, corneal edema, and folds in Descemet’s membrane (
Fig. 12.2A).
9 Aqueous flare is seen in most eyes with iris neovascularization, but a mild anterior chamber reaction is also seen in 18% of eyes with the ocular ischemic syndrome, even in the absence of rubeosis.
7 In some cases, this anterior chamber inflammation may be the only obvious feature of the disease, and it may be easily mistaken for anterior uveitis.
11 Progressive cataracterous changes are often seen (
Fig. 12.2A), presumably related to anterior segment ischemia, anterior chamber inflammation, or both.
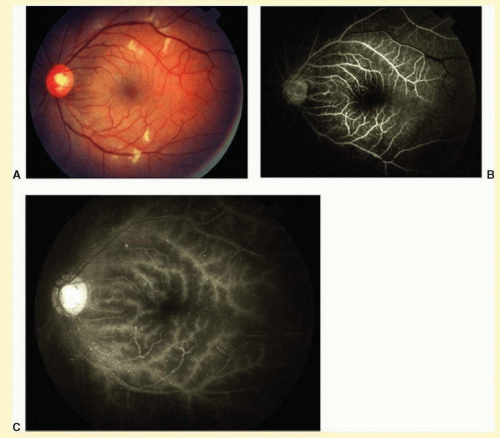
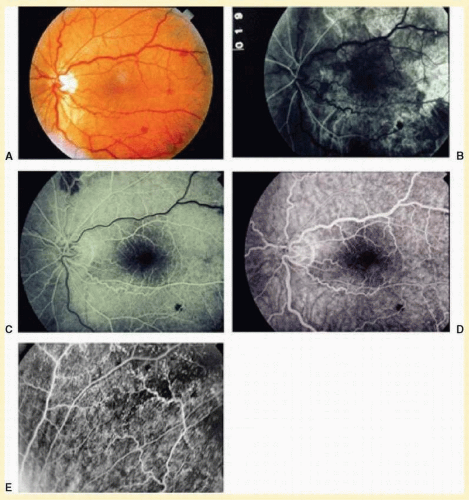
The retinal arteries are generally narrowed and straightened in eyes with the ocular ischemic syndrome (
Figs. 12.4A and
12.5A).
10 Although such arterial narrowing can be seen in systemic hypertension or as a normal variation in elderly people, it is important to note that retinal arterial narrowing can also be caused by the ocular ischemic syndrome in and of itself. A significant asymmetry in retinal arterial narrowing between the two eyes may be seen in unilateral cases of the ocular ischemic syndrome.
Dilated retinal veins are frequently seen in eyes with the ocular ischemic syndrome (
Figs. 12.4A and
12.5A). Irregularity of the vessel caliber may be present, similar to the venous beading seen in preproliferative or proliferative diabetic retinopathy. In contrast to central retinal vein obstruction, the veins in eyes with the ocular ischemic syndrome are generally not tortuous. The fact that the ocular ischemic syndrome occurs secondary to impaired inflow, whereas central retinal vein obstruction is usually associated with compromised outflow, may account for the difference.
Intraretinal dot and blot hemorrhages are seen in about 80% of eyes with the ocular ischemic syndrome (
Figs. 12.4A and
12.5A).
7 As with diabetic retinopathy, these hemorrhages probably arise secondary to the rupture of microaneurysms, which in turn are caused by endothelial damage. Unlike background diabetic retinopathy, in which the intraretinal hemorrhages are distributed throughout the posterior pole, the hemorrhages of the ocular ischemic syndrome tend to be located mainly in the midperipheral fundus. The hemorrhages of CRVO tend to be more numerous and larger than those of the ocular ischemic syndrome.
Neovascularization of the retina or optic disc may occur in the ocular ischemic syndrome. Optic disc neovascularization is quite common, seen in 35% of patients.
7 This incidence is much higher than that seen in ischemic CRVO (5%)
22 and CRVO (2%).
23 This high incidence of disc involvement may be reflective of the generalized hypoxic status of the eye in the ocular ischemic syndrome. In an older patient with no history of diabetes mellitus or vascular occlusive disease, the presence of neovascularization of the optic disc should arouse the clinical suspicion of the ocular ischemic syndrome.
Neovascularization of the retina is seen in 8% of patients with the ocular ischemic syndrome, usually seen concurrently with neovascularization of the disc.
7 Secondary vitreous hemorrhage may occur as a result of traction upon the new vessels by the vitreous gel.
A cherry-red spot is seen at presentation in about 12% of eyes with the ocular ischemic syndrome.
7 It may occur acutely due to inner-retinal-layer ischemia and whitening from emboli to the central retinal artery, or it may occur more chronically. The chronic form occurs when the intraocular pressure encroaches upon the perfusion pressure of the central retinal artery, and blood flow to the inner layer of the retina is compromised. The visual prognosis is generally grim when a cherry-red spot develops.
Cotton-wool spots are observed in about 5% of eyes with the ocular ischemic syndrome (
Fig 12.6A).
7 The cottonwool spots are generally located in the posterior pole.
Spontaneous pulsations of the central retinal artery are highly suggestive of the ocular ischemic syndrome. When the diastolic pressure within the central retinal artery falls below the intraocular pressure, the retinal arteries collapse during diastole, and spontaneous pulsation of the central retinal artery is observed. This phenomenon is present in 5% of eyes with the ocular ischemic syndrome.
7When not present spontaneously, retinal arterial pulsations may be induced by light digital pressure to the globe, thereby artificially raising the intraocular pressure above the diastolic retinal arterial pressure. With slightly more pressure, the retinal arterial tree may even be seen to collapse as the intraocular pressure is raised above the systolic retinal arterial pressure. Correspondingly, as would be expected, ophthalmodynamometry and oculoplethysmography reveal decreased readings in eyes with the ocular ischemic syndrome.
Ischemic optic neuropathy has been reported in an eye with the ocular ischemic syndrome.
25 Hypoperfusion of the posterior ciliary circulation to the optic disc due to impaired carotid blood flow or emboli of carotid origin is the probable cause of this complication.
The carotid pulse is often diminished or absent in the presence of carotid occlusive disease. The finding of a cervical bruit is significant, although its absence does not exclude a significant or even a complete carotid occlusion.
26,
27
DIFFERENTIAL DIAGNOSIS
Diabetic retinopathy and nonischemic central retinal vein obstruction are the two entities most likely to be confused with the ocular ischemic syndrome. Differentiation from diabetic retinopathy can be especially difficult, since many persons with the ocular ischemic syndrome have diabetes mellitus as well, and the retinal manifestations of each may be superimposed. Both the ocular ischemic syndrome and diabetic retinopathy tend to display dilated and beaded retinal venules, dot and blot intraretinal hemorrhages, and microaneurysms.
9,
28 Diabetic retinopathy tends to have bilateral involvement, while the ocular ischemic syndrome is more often unilateral than bilateral. The ocular ischemic syndrome generally affects an older population in the age range of 50s to 80s, while diabetic retinopathy may affect any age group. Retinal hemorrhages and microaneurysms tend to be midperipheral in the ocular ischemic syndrome, while they mainly involve the posterior pole in diabetic retinopathy. Hard exudates are common in diabetic retinopathy but are usually absent in the ocular ischemic syndrome unless the patient has concomitant diabetic retinopathy. On fluorescein angiography, the ocular ischemic syndrome typically displays delayed and patchy choroidal filling, prolonged arteriovenous transit time, and arterial staining, while in diabetic retinopathy these features are usually absent.
CRVO and the ocular ischemic syndrome both tend to affect an older patient population and display dilated venules with dot and blot intraretinal hemorrhages.
29,
30 In CRVO, venules may display tortuosity in addition to dilation. Bilateral involvement is uncommon in CRVO and occurs in a minority of cases of the ocular ischemic syndrome. In contrast to the ocular ischemic syndrome, there may be optic disc swelling and more pronounced macular edema in CRVO. On fluorescein angiography, both CRVO and the ocular ischemic syndrome may display a prolonged arteriovenous transit time, but in CRVO there is no defect in the choroidal filling pattern. In the ocular ischemic syndrome, vessel staining is predominantly arterial, while in CRVO it is mostly venous. Ophthalmodynamometry may also help in the diagnosis; the retinal arterial perfusion pressure is decreased in the ocular ischemic syndrome, while it is normal in CRVO.
A fundus appearance similar to that found in the ocular ischemic syndrome has been reported to occur secondary to an embolus within the central retinal artery.
31 Presumably, this occurred on the basis of hypoperfusion.