Fig. 12.1
The silicone tube is inserted into the fibrous capsule encasing the scleral buckle through a small incision on top of the scleral buckle. The tube exits the capsule through a 23-gauge needle anteriorly before scleral fistulization, anterior chamber insertion, and placement of a scleral patch graft
7.
Balanced salt solution on a blunt 27-gauge cannula is injected into the proximal end of the tube to test resistance and elevation of the fibrous capsule.
8.
Tube ligation: The tube can be ligated with 7-0 polyglactin for early flow restriction and prevention of immediate postoperative hypotony (Fig. 12.2).
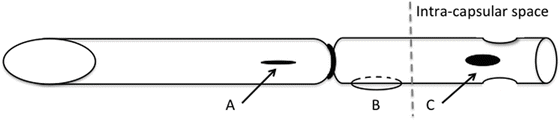

Fig. 12.2
Tube modifications include (A) fenestrations anterior to the ligature, (B) placing a securing suture to the sclera, and (C) side ports at the intracapsular end of the tube. A 7-0 polyglactin cutting needle, a 9-0 nylon suture, and curved Westcott scissors can be used to perform these modifications, respectively. Modified from Lee et al. [27]
9.
Sclerostomy: A sharp 23-gauge needle is used to enter the anterior chamber 1–2 mm posterior to the limbus and parallel to the iris plane. The tube is trimmed to the appropriate length with an anterior bevel, and is inserted into the anterior chamber through the needle tract. After ensuring that the tube is adequately positioned in the anterior chamber away from the corneal endothelium, it may be fixated to the sclera using a 9-0 nylon suture. Verify that the anterior chamber is holding stable depth.
10.
Patch graft: To prevent erosion through the conjunctiva, cover the tube with a donor tissue patch graft (cornea, sclera, or pericardium), secured to the sclera using 7-0 polyglactin sutures.
11.
Conjunctival closure: The conjunctiva is sutured using 8-0 polyglactin on a tapered needle in a watertight fashion.
12.
Subconjunctival injections of an antibiotic and corticosteroid are administered and the traction suture, lid speculum, and drapes are removed.
12.3 Procedure Modifications and Alternatives
1.
2.
3.
4.
A Baerveldt glaucoma implant (or other glaucoma drainage devices) may be used [30, 31]. The device should be placed in the quadrant with least amount of hardware (away from the area where the ends of the scleral buckle are tied); adequate posterior dissection is needed to create enough space for the endplate. The endplate is positioned over the recti muscles and the encapsulated scleral buckle. The endplate can be secured to the sclera or buckle. Either 250 or 350 mm endplates may be used and trimmed to fit in tighter spaces if significant subconjunctival scarring is present.
12.4 Outcomes
The initial ACTSEB procedure described by Schocket and colleagues consisted of shunting aqueous to an encircling band in eyes with NVG [25, 26]. Final IOP in 18 of 19 eyes (95 %) was less than 20 mmHg (average IOP 16.2 mmHg, mean follow up 59 weeks). Of these 18 eyes, only 3 required IOP-lowering medications. The original Schocket faired well when compared to the initial data on the Krupin and Molteno glaucoma drainage devices [25]. The original Schocket was also compared to the double-plate Molteno in a randomized, prospective study in eyes with refractory glaucoma [32]. In this study, the final IOP was similar between groups (14.4 mmHg with Molteno and 15.1 mmHg with Schocket, mean follow up 26 months), although the Schocket group required fewer medications (mean 0.95 vs. 0.43 number of medications for the Molteno and Schocket groups, respectively). Nevertheless, more frequent complications were encountered in the Schocket group. Also, newer glaucoma drainage devices, such as the Baerveldt and Ahmed implants, are similarly effective and do not require placement of a 360° encircling band, making them a more popular choice than the original Schocket procedure for treatment of refractory glaucoma.
The modified Schocket procedure originally described by Sidoti and colleagues—and discussed here—consists of shunting aqueous to the fibrous capsule of a previously existing scleral buckle in eyes with a history of retinal detachment [24]. Successful control of IOP (defined as IOP between 6 and 21 mmHg with or without IOP-lowering medications) after 1 year was achieved in about 85 % of the eyes in 2 independent small case series [27, 24]. Ten out of twenty-one eyes required at least one IOP-lowering medication, and vision improved in nineteen of twenty-one eyes in the two series combined. Thus, shunting aqueous to an encircling band placed at the time of surgery (original Schocket) or into the capsule surrounding a preexisting scleral buckle (modified Schocket) is similarly effective in lowering IOP.
Using glaucoma drainage devices in eyes with preexisting scleral buckle is also a good surgical option. Small retrospective case series have looked at outcomes of Baerveldt drainage implants in eyes with preexisting scleral buckle and found similar results compared to the modified Schocket procedure [30, 31]. Twenty-eight of thirty-three eyes (85 %) achieved successful control of IOP (defined as IOP between 6 and 21 mmHg with or without IOP-lowering medications) at last follow up in both studies combined. In one of the series, 7 of 16 eyes required at least one IOP-lowering medication. Of the 9 eyes that did not need postoperative IOP-lowering medications, 2 had a Baerveldt 250 mm and 7 had a Baerveldt 350 mm device [30]. In general, vision remained stable in both these studies at last follow up.
There is no prospective, randomized clinical trial to date to compare the safety and efficacy between the modified Schocket procedure and glaucoma drainage implants in eyes with preexisting scleral buckles. Nevertheless, the outcomes reported for both procedures are comparable, and both are effective options in the treatment of refractory glaucoma in eyes with preexisting scleral buckles.
12.5 Complications
In the original series by Sidoti and colleagues, all complications related to the modified Schocket procedure were observed within 4 months postoperatively [24]. Complications included serous choroidal detachment, conjunctival dehiscence and wound leak, hyphema, and tube obstruction by lens, vitreous, or episcleral fibrous tissue. Interestingly, a more recent series by Lee et al. reports significantly fewer complications [27]. Lee et al. report tube exposure in one patient, and self-limited choroidal effusions in three eyes during the immediate postoperative period. They attribute the decreased rate of complications to securing the tube to the sclera, and creating side ports on the intracapsular end of the tube (Fig. 12.2). Indeed, intracapsular obstruction was the most common complication, and obstruction inside the eye could be attributed to tube migration. Thus, albeit limited evidence, these simple modifications appear to be beneficial in decreasing complications.
Placement of Baerveldt drainage implants in eyes with preexisting scleral buckle is associated with similar postoperative complications, including limited choroidal effusion, limited choroidal hemorrhage, hyphema, and conjunctival dehiscence and wound leak [30, 31]. Of note, there were more serious complications observed in these series, including retinal detachment recurrence, limited vitreous hemorrhage, and significant choroidal hemorrhage with subsequent phthisis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


