Fig. 15.1
The Hydrus micro-stent consists of a curved implant with an inlet, three windows, and an open lumen designed to scaffold open Schlemm’s canal
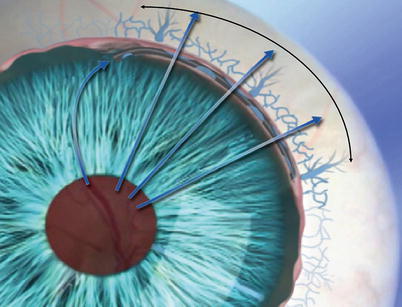
Fig. 15.2
The Hydrus micro-stent with anterior chamber inlet providing direct aqueous access to the canal, and three windows to permit aqueous flow through the inner wall and scaffolded Schlemm’s canal into collector channels and aqueous veins
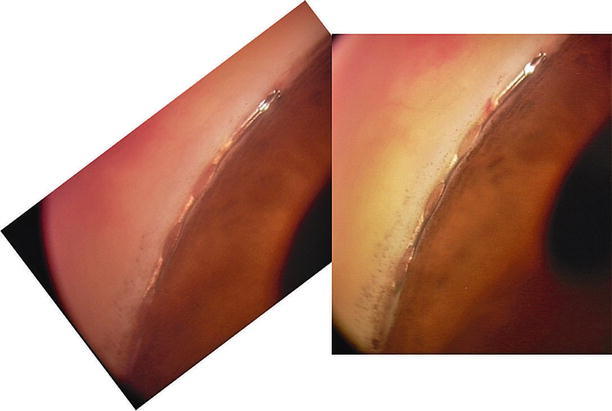
Fig. 15.3
Postoperative gonioscopic photos of the Hydrus micro-stent in position
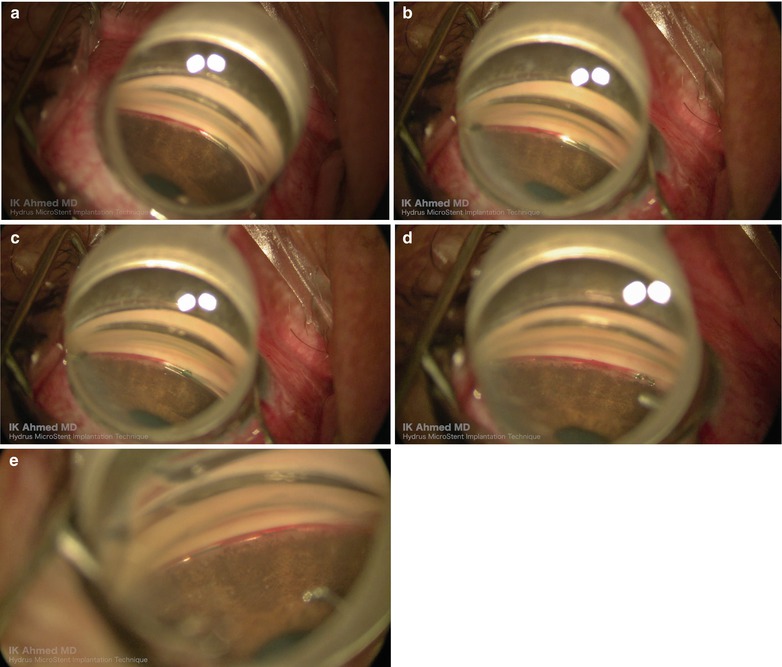
Fig. 15.4
Surgical technique. The beveled cannula is used to incise the inner wall (a). The Hydrus implant is advanced into Schlemm’s canal (b, c). Once implanted, the inlet is visible in the anterior chamber angle (d). The three windows are visible in the canal (e)
Biocompatibility of the device has been studied histologically in both rabbit and nonhuman primates (Fig. 15.5) [1]. At 6 months, the device showed minimal inflammation in implanted New Zealand rabbits. In an analysis 13 weeks after implantation in nonhuman primates, there was no evidence of an inflammatory, foreign body response or fibrosis around the device when placed in the canal region. Based on these findings, the Hydrus micro-stent appears to be biocompatible in the eye.
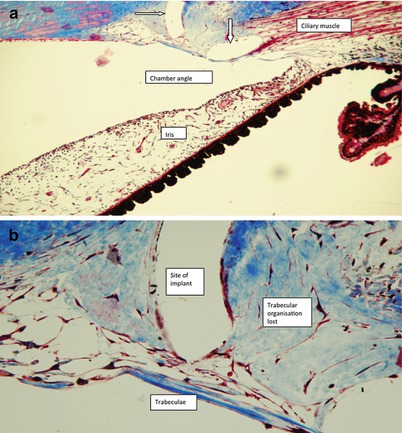

Fig. 15.5
(a) Histopathology section in nonhuman primate shows two segments of the Hydrus scaffold in white (arrows) with no evidence of chronic inflammatory reaction in the outflow system or in adjacent tissues. (b) At higher magnification shows a segment of the Hydrus scaffold in white with distortion to the TM but no chronic inflammation
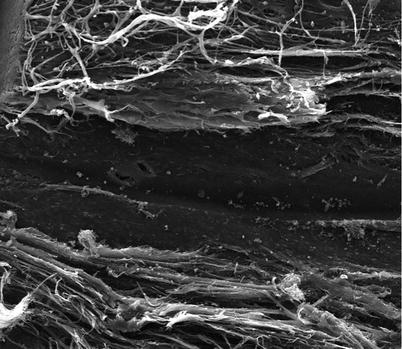
A cadaveric injury study was performed to assess the impact of placement of the Hydrus into the canal using scanning electron microscopy (SEM) (Fig. 15.6) [2]. Using fresh human cadaveric anterior segments, SEM analysis showed patency of the canal, and collector channels were maintained with minimal alteration of normal Schlemm’s canal anatomy. The low profile device appears to be inserted with minimal disruption of normal canal architecture.