13 The Eyelid and Lacrimal Drainage System
INTRODUCTION
The eyelids are flaps of skin with highly modified epidermal appendages that cover and protect the eye. The structure of the eyelids is discussed in Chapter 1 and illustrated in (Fig. 13-1). The eyelids form a moist chamber lined by mucous membrane (conjunctiva) that is absolutely essential for the maintenance of corneal transparency. The importance of the eyelids in the maintenance of corneal health and transparency becomes evident when facial paralysis or stupor prevent normal eyelid closure and produce corneal exposure or lagophthalmos (rabbit eye). If congenital defects in the eyelid called colobomas (coloboma—“a defect”) are not corrected expeditiously, corneal ulceration and/or opacification caused by epidermalization invariably result. The corneal epithelium literally turns to skin if it is not continuously moistened.
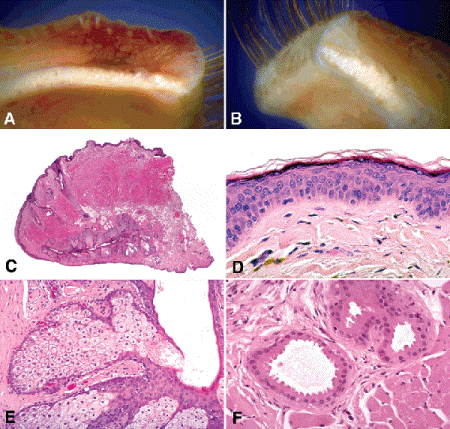
Fig. 13-1. A. Upper eyelid. The upper eyelid is roughly rectangular compared to the lower lid, which is much shorter and triangular in shape. The anterior surface of the lid is covered by skin. A row of cilia (eyelashes) arises near the lid margin (at right) and curves away from the globe. The white foci seen below are the lobules of a Meibomian gland in the tarsal plate, the eyelid’s fibrous skeleton. The palpebral conjunctiva is tightly adherent to the back surface of the tarsal plate. B. Lower eyelid. The lower eyelid is roughly triangular and has a much shorter tarsal plate and fewer meibomian glands. C. Histology, lower eyelid. The anterior surface of the eyelid is covered by a delicate layer of skin. The posterior surface is covered by the palpebral conjunctiva, which is firmly adherent to the tarsal plate. The tarsal plate contains large sebaceous glands called the Meibomian glands. Eosinophilic bundles of orbicularis muscle are seen in cross section in the connective tissue anterior to the tarsus. The tarsal plate of the lower lid is much shorter. The lower lid is roughly triangular in shape. D. Eyelid skin. The skin of the eyelid is extremely delicate and lacks rete pegs. The layers of the epidermis include the basal cell layer, the malpighian or prickle cell layer, the granular cell layer, and the superficial keratin layer. E. Meibomian gland lobule, eyelid. Each Meibomian gland is composed of multiple sebaceous gland lobules arranged along a central duct, which is oriented perpendicular to the lid margin. Sebaceous glands are holocrine glands; lipidized cells shed into the duct comprise the secretory product. Cellular division occurs in the basal cell layer in the periphery of the lobules. The nuclei become increasingly pyknotic as the cells mature and become lipidized. A flaplike valve of ductal epithelium covers the opening of this lobule. F. Glands of Moll. The dilated lumina of these apocrine sweat glands are lined by tall eosinophilic cells capped with the apical snouts that characterize apocrine decapitation secretion. (C. H&E ×10, D. H&E ×50, E. H&E ×50, F. H&E ×100)
The external surface of the eyelid is covered by one of the thinnest and most delicate layers of skin in the body (Fig. 13-1D). The epidermis of the eyelid skin is generally only five or six cells in thickness and is covered by a thin layer of surface keratinization. Unlike skin elsewhere, eyelid epidermis lacks rete ridges. The basal cell or germinative layer of the epidermis, where cellular division normally occurs, rests on a delicate basement membrane. Compared to the squamous cells of the overlying epidermis, the basal cells have relatively little cytoplasm. Hence, lesions composed of basal cells such as basal cell carcinoma appear blue or basophilic on low power microscopy. Most of the epidermis is composed of the prickle cells of the stratum spinosum or Malpighian layer. These cells are polygonal in shape and are joined by bundles of tonofilaments evident in light microscopy as intercellular bridges. Squamous cell lesions appear pink or eosinophilic under low power microscopy because the cells have abundant eosinophilic cytoplasm.
TERMS USED IN SKIN PATHOLOGY
Thickening of the prickle cell layer is termed acanthosis (from Gk, akantha, thorn) (Fig. 13-2A). As basal cells mature and approach the surface of the skin, they become flattened and squamoid. The nuclei undergo apoptosis as the cells near the surface, releasing intensely basophilic granules of nucleoprotein that form the granular cell layer. The dead, desiccated, anucleate cellular remnants form a thin horny layer of keratin called the stratum corneum (from Latin, cornu, a horn). Abnormal thickening of this normally thin surface layer of keratin is called hyperkeratosis (Fig. 13-2A). Hyperkeratosis is commonly found on the surface of benign squamous papillomas and is responsible for the greasy or scaly character of seborrheic keratoses. Colonies of yeast or bacteria frequently are found in the thick layer of keratin.
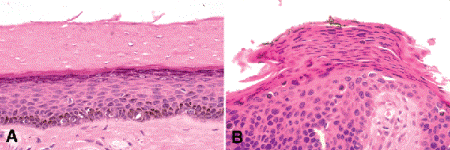
Fig. 13-2. A. Hyperkeratosis. The superficial layer of eosinophilic keratin is markedly thickened. The dead cells comprising the mass of keratin lack nuclei. A granular cell layer is present beneath the keratin. The epidermis in this specimen is mildly thickened. B. Parakeratosis, actinic keratosis. The keratin layer retains flattened nuclei, and no granular cell layer is present. Parakeratosis is typically found in actinic keratosis. In this example, the epidermis is composed of mildly atypical squamous cells. (A. H&E ×50, B. H&E ×100)
Retention of nuclei in the stratum corneum is termed parakeratosis (Fig. 13-2B). Parakeratosis generally occurs in pathologic states when the epidermis is rapidly proliferating. When parakeratosis is present, there is no granular cell layer in the underlying epidermis. Parakeratosis is relatively rare and should alert one that he/she may be dealing with a premalignant lesion such as actinic keratosis.
Dyskeratosis refers to the keratinization of single cells within the prickle cell layer. Dyskeratotic cells are round and intensely eosinophilic and have pyknotic nuclei. Dyskeratosis is particularly striking in the conjunctival lesions of hereditary benign intraepithelial dyskeratosis.
Congenital and Developmental Lesions
Developmental anomalies of the eyelid include ablepharon, cryptophthalmos, colobomas, microblepharon, ankyloblepharon, euryblepharon, blepharophimosis, congenital ectropion, epicanthal folds, and dystopia canthorum. An intact sheet of skin covers the eye in cryptophthalmos. Affected patients may have Fraser syndrome, which also includes syndactyly, renal agenesis, and aural and genital malformations. Colobomas are partial or full-thickness defects in the lid that affect the lid margin. Lid colobomas are common in Goldenhar syndrome and mandibulofacial dysostosis. Isolated strands of skin bridge the palpebral fissure in ankyloblepharon filiform adnatum. These are related to the fusion of the upper and lower lids that normally occurs in utero. Dystopia canthorum, iris heterochromia, a white forelock, synophrys, and deafness comprise Waardenburg syndrome. An accessory row of eyelashes arises from the meibomian glands in distichiasis. Phakomatous choristoma or Zimmerman tumor is a rare congenital neoplasm that involves the lower medial eyelid or anterior orbit. The ultimate choristoma, Zimmerman tumor is composed of extraocular eye lens tissue including lens epithelium, neoplastic lens capsule, and bladder cells like those found in posterior subcapsular cataract (Fig. 13-3).
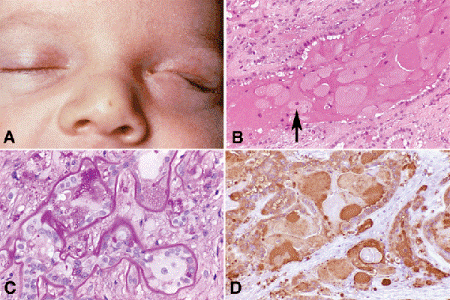
Fig. 13-3. Zimmerman tumor (phakomatous choristoma). A. Phakomatous choristoma is a rare congenital tumor of lenticular anlage that always occurs in the lower medial eyelid or anterior orbit. B. The tumor is composed of neoplastic lens epithelial cells. Arrow denotes cell resembling bladder or Wedl cell found in posterior subcapsular cataract. C. Lens epithelial cells rest on thick PAS-positive basement membrane that mimics lens capsule. D. Cells are immunoreactive for lens crystallin. (B. H&E ×50, C. PAS ×250, D. IHC for beta crystalline ×100)
AGING CHANGES
Aging produces atrophy and laxity of eyelid skin (dermatochalasis), loss of orbital fat and subcutaneous tissue, and relaxation of eyelid ligaments. Folds of redundant skin overhang the upper lid margin, and the orbital fat protrudes through the attenuated orbital septum, producing bags under the eyes. Chronic light damage (senile or actinic elastosis) is evident histologically as basophilic degeneration of dermal collagen.
Laxity of eyelid tissues predispose to senile entropion or ectropion. Entropion occurs when the preseptal part of the orbicularis muscle overrides the pretarsal part. The lateral canthal tendon is lax in senile ectropion, and the exposed conjunctiva becomes inflamed and undergoes epidermalization. The floppy eyelid syndrome occurs in obese men who have eyelids with a rubbery tarsus that are easily everted. Spontaneous eversion of the eyelids or loss of eyelid contact occurs during sleep, causing chronic papillary conjunctivitis. Patients with floppy eyelid syndrome should be evaluated for obstructive sleep apnea, a potentially fatal condition.
COMMON INFLAMMATORY LESIONS OF THE EYELID
Hordeolums or styes and chalazia are common inflammatory lesions of the eyelid. Hordeolums are acute infections of eyelash follicles (external hordeolums) or meibomian glands (internal hordeolums) that usually are caused by staphylococci. Internal hordeolums are more painful because the inflammation is localized within a confined space. Hordeolums usually respond to conservative therapy, for example, hot compresses, and therefore are rarely examined histopathologically.
Chalazion
Chalazia typically appear as mildly tender nodules, or areas of localized nodular thickening on the eyelid (Fig. 13-4A). Inflammatory signs such as pain and redness are not especially prominent because chalazia are chronic inflammatory lesions. Chalazia are chronic lipogranulomas, that is, “endogenous foreign body reactions” to the lipid-rich secretions of the meibomian and Zeis glands. When sebum escapes from its normal confines in these sebaceous glands, it is extremely irritating and stimulates chronic granulomatous inflammation. Histopathology discloses epithelioid histiocytes and inflammatory giant cells that typically surround empty spaces (lipid vacuoles) (Fig. 13-4B). In vivo the vacuoles contained lipid. During tissue processing solvents such as alcohol and xylene, dissolve out the lipid. Other chronic inflammatory cells such as lymphocytes and plasma cells contribute to the chronic inflammatory infiltrate in chalazion specimens. If the chalazion has ruptured spontaneously, or if prior incision and drainage has been performed, granulation tissue is often found. Although granulation tissue typically is nongranulomatous, the granulation tissue associated with chalazion may contain epithelioid cells or giant cells, which are residua of the lipogranulomatous response. Chalazion curettings often include fragments of tarsal plate.
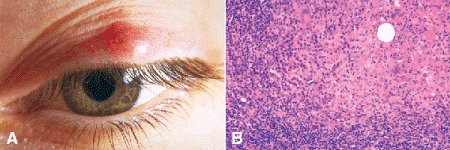
Fig. 13-4. Chalazion. A. The focal area of nodular thickening in the upper eyelid represents a chronic granulomatous inflammatory response to irritating lipid material that has escaped from its normal compartment in the lid. Chalazia usually are only mildly tender and inflamed. Recurrent or atypical “chalazia” should be examined pathologically to exclude simulating lesions such as sebaceous carcinoma. B. Pink epithelioid histiocytes and giant cells indicative of chronic granulomatous inflammation surround an empty lipid vacuole. The lipid was dissolved by fat solvents during processing. Empty lipid vacuoles are required for the diagnosis of lipogranulomatous inflammation. The inflammatory infiltrate also contains many plasma cells and lymphocytes. (B. H&E ×100)
Sebaceous carcinoma, which also arises in Zeis and meibomian glands, can be confused with chronic chalazia clinically. It is imperative that recurrent or atypical chalazia be submitted for histopathologic examination to exclude the possibility of this potentially fatal eyelid malignancy. The author personally has seen several cases of sebaceous carcinoma that were initially misdiagnosed as chalazia. These lesions were treated with incision and drainage and were not examined histopathologically. When the correct diagnosis was finally made, extensive pagetoid involvement of the conjunctiva by tumor cells necessitated orbital exenteration. Other unusual neoplasms such as sweat gland carcinomas, Merkel cell tumors, and carcinoma metastatic to the eyelid have been misdiagnosed clinically as chalazia.
VIRAL LESIONS
Verruca Vulgaris
Verruca vulgaris, a benign papillomatous skin lesion caused by human papillomavirus type 2 (HPV-2), a DNA papovavirus, occasionally occurs on the eyelid. These viral papillomas are distinguished by elongated spire-shaped papillary fronds and rete ridges that bend inwardly toward the center of the lesion (Fig. 13-5A). Vertical tiers of parakeratosis occur on the crests of papillomatous elevations and overlie foci of vacuolated cells that contain eosinophilic intranuclear and intracytoplasmic viral inclusions (Fig. 13-5B). The viral inclusions, which are necessary for definitive diagnosis, are only found in early lesions. Verruca vulgaris is diagnosed infrequently in the ophthalmic pathology laboratory.
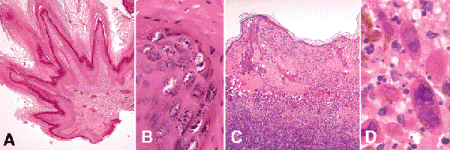
Fig. 13-5. Viral lesions. A. Verruca vulgaris. Viral papilloma is composed of spire-shaped fronds of hyperkeratotic epidermis. B. Parakeratosis and vacuolated cells containing viral inclusions are seen on crest of frond. C. Herpes simplex infection of the skin. Serous fluid and inflammatory cells fill herpetic vesicle that has formed within the acantholytic epidermis. The underlying dermis is intensely inflamed. D. Multinucleated giant cells with Cowdry type A intranuclear inclusions are present. (A. H&E ×25, B. H&E ×250, C. H&E ×25, D. H&E ×250)
Molluscum Contagiosum
Molluscum contagiosum is a fairly common viral tumor with a distinctive histopathologic appearance (Fig. 13-6). Caused by a pox virus, molluscum contagiosum often is associated with poor hygiene, and lesions may occur in crops on the face and eyelids of children as the result of autoinoculation. Massive involvement of eyelids and adnexal skin has been reported in patients with AIDS. Clinically, molluscum contagiosum appears as either an elevated smooth nodule with a central umbilication, or a larger pore enclosing multiple flattened papillae. Histopathologically, molluscum contagiosum typically has an elevated cup- or crateriform configuration and is composed of lobules of markedly acanthotic benign epithelium (Fig. 13-6A). The epithelial cells contain large intracytoplasmic inclusion bodies called Henderson Patterson corpuscles (Fig. 13-6B). The inclusions are eosinophilic near the base of the epithelium where they form, and they become denser and more basophilic as they mature and migrate toward the surface where they discharge virus particles. The individual virus particles are quite large and can be resolved with the oil immersion objective of the light microscope in one micron thick plastic-embedded sections. Molluscum contagiosum on the eyelid margin can incite unilateral chronic follicular conjunctivitis by dispersing viral particles into the conjunctival sac. The lids of any patient who has chronic follicular conjunctivitis should be inspected carefully for molluscum contagiosum (or the presence of pubic or “crab” lice whose waste can incite a similar reaction).
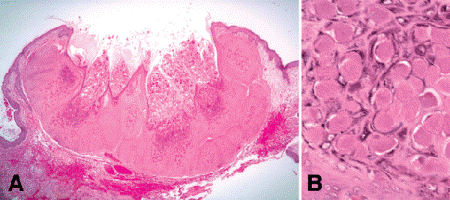
Fig. 13-6. Molluscum contagiosum. A. This viral tumor has a typical crateriform configuration. Inclusions of pox virus shed by the infected acanthotic epithelium fill the crater. B. Henderson-Patterson corpuscles. The pox virus infection causes lobular acanthosis of the epidermis. The thickened epidermis contains large oval intracytoplasmic viral inclusions called Henderson-Patterson corpuscles. The inclusions become increasingly basophilic as they mature. (A. H&E ×10, B. H&E ×100)
Molluscum contagiosum is one of three cup-shaped or crateriform lesions commonly found on eyelid skin. Basal cell carcinoma and keratoacanthoma also are often umbilicated or have a central area of ulceration. Both tend to be larger than molluscum contagiosum.
Other viral lesions that affect eyelid skin include the vesicular eruptions caused by the herpes simplex and varicella-zoster viruses. Herpes simplex infection causes fluid-filled intraepidermal vesicles marked by profound epidermal degeneration and acantholysis (Fig. 13-5C,D). Round acantholytic balloon cells and multinucleated giant cells that contain eosinophilic intranuclear viral inclusions are found.
Benign Cystic Lesions
Eyelid cysts are commonly accessioned by ocular pathology laboratories. Most are epidermal inclusion cysts or sweat ductal cysts.
Epidermal inclusion cysts are round or oval and unilocular (have a single lumen) and are filled with cheesy, foul-smelling keratin debris. They are lined by keratinizing stratified squamous epithelium that resembles normal epidermis (Fig. 13-7). By definition, the epithelial lining has no epidermal appendages such as pilosebaceous units or sweat glands. (If epidermal appendages are present, and hair shafts are found mixed with the keratin filling the lumen, the cyst probably should be classified as a cystic dermoid, a developmental lesion caused by entrapment of epidermis in bony sutures.) Many epidermal inclusion cysts are caused by the cystic dilatation of hair follicles by keratin debris. Essentially, they result from the cystic dilatation of a single epidermal appendage. Follicular cyst, infundibular type is the dermatopathologic term applied to this type of cyst. In such cases, the lining of the cyst may communicate with the skin through a pore. Clinically, epidermal inclusion cysts are called sebaceous cysts, an inappropriate term since they have nothing to do with sebaceous glands or sebum.
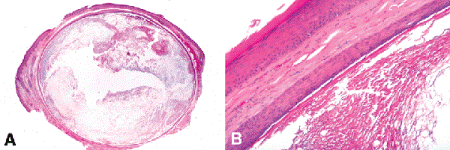
Fig. 13-7. Epidermal inclusion cyst. A. Epidermal inclusion cysts typically are round or oval and unilocular. The lumen of the cyst contains laminated eosinophilic keratin, which had a cheesy appearance grossly. The cyst is lined by keratinized squamous epithelium that resembles skin but lacks epidermal appendages. B. The surface of the skin is seen at top left. The cyst is lined by keratinized stratified squamous epithelium resembling epidermis. Laminated keratin fills lumen. (A. H&E ×5, B. H&E ×50)
Sweat ductal cysts, sudoriferous cysts, or hidrocystomas are caused by the blockage of a sweat duct. These cysts are soft, smooth, and fluctuant and are filled with watery fluid. Some are transparent or translucent, and they readily transilluminate. Some patients have multiple cysts that often involve that skin of both medial or lateral canthi (Fig. 13-8).
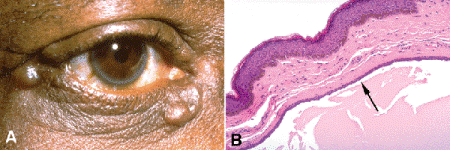
Fig. 13-8. Eccrine hidrocystoma. A. Cysts caused by the blockage of eccrine sweat glands often involve the canthal skin. Multiple cysts occur in some patients. They are filled with watery fluid. Eccrine hidrocystomas also are called sweat ductal or sudoriferous cysts. B. The eccrine hidrocystoma is lined by a dual layer of epithelial cells (arrow) resembling the epithelium of an eccrine sweat gland duct. The lumen is filled with eosinophilic granular debris consistent with serous fluid. The lumen is often branching and multilocular. (B. H&E ×100)
Histopathologically, sweat ductal cysts are often multilocular, and their lumens appear empty or contain scant amounts of granular eosinophilic material consistent with serous fluid. The epithelial lining, which is often attenuated, is composed of a dual layer of cells that resembles the normal lining of an eccrine sweat duct (Fig. 13-8B). Although most sudoriferous cysts are eccrine hidrocystomas, the epithelial lining occasionally shows apocrine differentiation. Apocrine hidrocystomas (Fig. 13-9) are recognized by their epithelial lining, which is composed of tall cells with eosinophilic cytoplasm and apical snouts of decapitation secretion (Fig. 13-9D). The cytoplasm of the apocrine cells may contain golden-brown granules of lipofuscin pigment that are periodic acid-Schiff-positive. Apocrine hidrocystomas often contain brown fluid stained by this pigment and may be misdiagnosed preoperatively as primary melanocytic lesions (Fig. 13-9A,B).
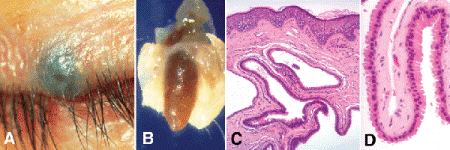
Fig. 13-9. Apocrine hidrocystoma. A. Bluish lesion near lid margin was thought to be pigmented melanocytic nevus preoperatively. B. Macrophoto of another case shows brownish fluid filling lumen of cyst. C. Branching lumen of multilocular Moll gland cyst appears empty. D. The cells comprising the epithelial lining are tall and show apical snouts of decapitation secretion indicative of apocrine differentiation. (C. H&E ×25, D. H&E ×100)
Benign Epidermal Lesions
Benign tumors that arise from the epidermis generally are located anterior to the plane of the surrounding normal skin because they are composed of cells that are unable to invade or infiltrate the dermis or deeper structures of the lid as malignant lesions can. The redundant epidermis formed by the proliferation of these benign cells typically forms a branching tree-shaped lesion composed of fronds or fingerlike projections that have cores of fibrovascular tissue analogous to dermis. A lesion that exhibits this growth pattern is called a papilloma (Fig. 13-10). Papillomas can be elevated and exophytic or relatively squat and sessile. A variety of lesions show a papillomatous growth pattern including some malignant tumors, for example, papillary squamous cell carcinoma of the conjunctiva.
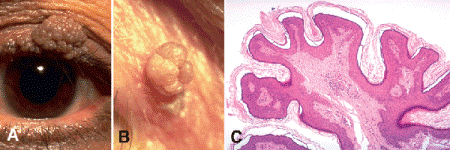
Fig. 13-10. Squamous papillomas. A. Squamous papillomas are very common benign epidermal tumors of the eyelid. They are branching tree-shaped lesions composed of multiple fronds of benign epidermis. A large papilloma is present on upper eyelid. B. Smaller pedunculated papillomas are called skin tags, acrochordons, or fibroepithelial polyps. C. Multiple fronds or fingerlike projections of epidermis comprise the benign epidermal tumor. The epidermal fronds contain a central core of fibrovascular tissue. Hyperkeratosis is present on the surface of this lesion. (C. H&E ×25)
Benign papillomatous lesions of eyelid skin such as seborrheic keratoses and squamous cell papillomas frequently are submitted for histopathologic evaluation. In most instances, the correct diagnosis is suspected preoperatively, and cosmesis is a major indication for excisional biopsy.
Seborrheic Keratosis
Seborrheic keratoses usually occur in elderly patients. They are sharply demarcated, superficial lesions that sit anterior to the plane of surrounding epidermis like a button on the surface of the skin (Fig. 13-11). They typically have a greasy or scaly appearance caused by hyperkeratosis and are often pigmented. Approximately one third of black adults have multiple, small, pigmented seborrheic keratoses on their facial skin, a condition called dermatosis papulosa nigra (Fig. 13-11A).
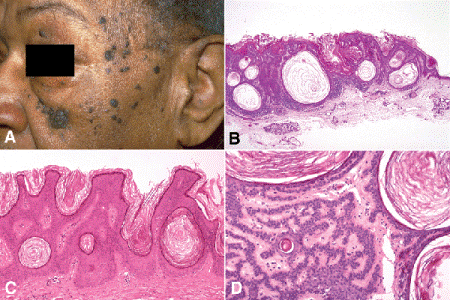
Fig. 13-11. Seborrheic keratosis. A. Dermatosis papulosa nigra. Multiple seborrheic keratoses are seen on the face of this elderly African American woman. B. Benign sessile papilloma is situated anterior to the plane of the surrounding skin. Lesion contains many characteristic pseudohorn cysts. C. Thick layer of hyperkeratosis covers sessile papilloma. Characteristic pseudohorn cysts filled with keratin are present. D. Seborrheic keratosis, adenoid type. The dermis contains interweaving bands of benign epithelial cells. Several keratin-filled pseudohorn cysts are present. (B. H&E ×10, C. H&E ×25, D. H&E ×50)
Seborrheic keratoses are an upward papillomatous proliferation of basaloid cells that resemble normal epidermal basal cells. Therefore, under low magnification, seborrheic keratoses often appear “blue” and are situated “above” the plane of the surrounding skin (signifying a benign lesion of basal cells) (Fig. 13-11B). In contrast to “garden variety” squamous papillomas, acrochordons, or skin tags, which are often exophytic and pedunculated (Fig. 13-10A,B), seborrheic keratoses are often sessile. A thick surface layer of keratin usually is present, and circular spaces filled with keratin called “horn cysts” or “pseudo-horn cysts” are found within the acanthotic epithelium. Most probably represent invaginations of surface keratin (Fig. 13-11B–D).
Interweaving bands composed of benign cells are found in the dermis in the adenoid variant of seborrheic keratosis (Fig. 13-11D), which should not be confused with the highly infiltrative morpheaform variant of basal cell carcinoma. Inverted follicular keratosis (IFK) is considered by some to be an irritated form of seborrheic keratosis. IFK typically has an inverted cup-shaped configuration and surface invaginations that have been interpreted as hair follicles (Fig. 13-12). Other characteristic histopathologic features include acantholysis (widening of intercellular spaces between squamous cells) and small foci of squamous differentiation called squamous eddies.
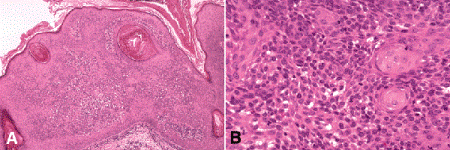
Fig. 13-12. Inverted follicular keratosis. A. IFK is thought to be an irritated variant of seborrheic keratosis. Acantholysis and circular foci of squamous cells (squamous eddies) are characteristic histologic features. B. Several oval foci of squamous differentiation called squamous cells are seen at right. The smaller basaloid cells show mild acantholysis. (A. H&E ×25, B. H&E ×100)
Many benign eyelid papillomas do not fulfill all of the diagnostic criteria for seborrheic keratosis. Such lesions tend to be small and pedunculated, and their epidermal component is not particularly basaloid and resembles normal or only slightly acanthotic skin with minimal hyperkeratosis (Fig. 13-10C). The dermal component often contains a sparse to moderate infiltrate of chronic inflammatory cells. Such lesions usually are diagnosed as squamous papillomas and are also called skin tags or acrochordons. Some may be viral in origin.
Keratoacanthoma
Keratoacanthoma is a crater-shaped squamous cell lesion that classically arises rapidly in elderly patients and then undergoes spontaneous involution (Fig. 13-13A). Although keratoacanthoma was classified as a benign pseudoepitheliomatous hyperplasia in the past, some authorities now believe that it actually may be a “deficient squamous cell carcinoma” that tends to involute spontaneously. Rare cases invade deeply like squamous cell carcinomas.
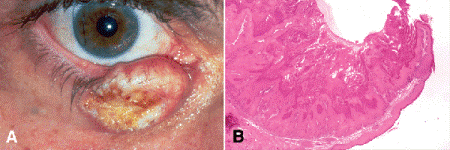
Fig. 13-13. Keratoacanthoma. A. Keratin fills irregular crater in large biopsy-confirmed keratoacanthoma of the lower eyelid. The tumor developed rapidly. Some authorities now believe that keratoacanthoma is a “deficient squamous cell carcinoma” that tends to involute spontaneously. B. A mass of keratin fills the crater-shaped tumor, which is composed of squamous cells with eosinophilic cytoplasm. A lateral buttress of normal skin is seen at right. The base of the lesion has a smooth “pushing” margin. The general configuration of the lesion is important in histopathologic diagnosis. It usually is impossible to distinguish keratoacanthoma from squamous cell carcinoma in a small incisional biopsy. (B. H&E ×5)
Histopathology discloses a central crater filled with a mass of keratin that is enclosed by markedly acanthotic squamous epithelium that typically extends like a lip or buttress over the side of the crater (Fig. 13-13B). The base of a well-developed lesion appears regular and well demarcated, and the margin is relatively smooth and “pushing ” rather than infiltrative. A intense band of inflammation usually is present in the underlying dermis. The overall configuration of the lesion is quite important from a diagnostic standpoint. It is usually impossible to differentiate keratoacanthoma from squamous cell carcinoma in a small incisional biopsy because keratoacanthoma frequently harbors atypical cells and mitotic figures. Keratoacanthomas should be totally excised with frozen section control of margins.
MALIGNANT TUMORS OF THE EYELID
Basal Cell Carcinoma
Basal cell carcinoma is the most common malignant tumor affecting the periocular skin. Basal cell carcinomas tend to affect fair skinned adults but can occur in younger persons. They arise in sun-exposed skin and are thought to be caused by actinic damage.
Several variants of basal cell carcinoma are recognized clinically. Nodulo-ulcerative basal cell carcinomas are firm, elevated, pearly nodules whose surface initially may be marked by telangiectatic vessels (Fig. 13-14A). Ulceration often develops centrally as the nodules increase in size. More advanced lesions appear as a slowly enlarging ulcers with prominent rolled pearly borders. Nodulo-ulcerative basal cell carcinomas usually have relatively distinct margins.
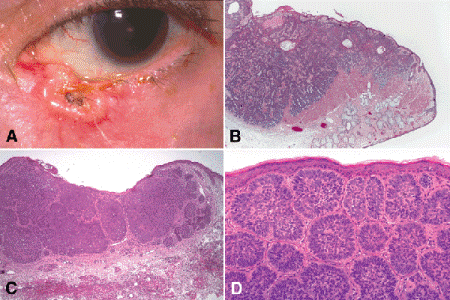
Fig. 13-14. Basal cell carcinoma. A. Nodulo-ulcerative basal cell carcinoma of lower lid has pearly elevated margins. B. Basal cell carcinoma of lower eyelid appears “blue and below.” Its cells are basophilic and are located deep to the plane of the epidermis. C. The surface of this tumor is ulcerated. Basophilic lobules invade dermis. D. Well-differentiated basal cell carcinoma is composed of nests of basaloid cells that show peripheral palisading of nuclei. Retraction artifact is not present. No connection with epidermis is evident in this field. (D. H&E ×100)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


