Chapter 11 The Evaluation and Management of Thyroid Nodules
Introduction
Thyroid nodules are common. The prevalence of nodularity increases with age, and population surveys suggest nearly 5% to 15% of the adult population may harbor a clinically significant nodule requiring evaluation.1 Thyroid nodule prevalence appears to have increased in the since the 1990s. Presently, no known environmental factor has been identified to explain this increase in prevalence. Rather, it has been largely attributed to two factors. First, thyroid nodular disease is an age-related illness.2 Also, recent population projections suggest that the median age of most industrialized populations is increasing. Together, these facts lead to an absolute increase in thyroid nodules. Increasing detection of thyroid nodules, often incidentally, appears equally important. Computer assisted tomography (CAT) and magnetic resonance imaging (MRI) of the head, spine, or chest frequently detect otherwise unidentified thyroid nodules. It is estimated that the number of CAT and MRI scans performed annually in the United States and Europe has more than tripled since the early 2000s. The detection of thyroid nodules has similarly increased.
Though often asymptomatic, thyroid nodules require evaluation primarily because of their risk of thyroid carcinoma and its potential dangers. As patients become aware of the risk for possible cancer, the anxiety associated with this disorder should not be underemphasized. In most large-scale reports, approximately 8% to 15% of thyroid nodules > 1 cm in diameter prove to be cancerous.3,4 This incidence appears to have increased since the 1990s, attributed primarily to improved nodule detection and more frequent nodule evaluation.5 Most well-differentiated thyroid malignancy is an indolent process effectively treated with surgery and, when needed, adjunctive radioactive iodine therapy.6 In contrast, untreated thyroid cancers can rarely metastasize and threaten life. Therefore, patients with newly identified thyroid nodules should seek diagnostic evaluation. Even if benign, large nodules may also cause obstructive symptoms, such as difficulty swallowing or tightness throughout the anterior neck. Cystic nodules can spontaneously hemorrhage. When this happens, rapid enlargement may occur, usually associated with pain and discomfort prompting urgent evaluation. If evaluation is delayed, resolution and reduction in nodule size will often occur. This is in contrast to most solid nodules, which often are persistent. Though benign solid thyroid nodules grow with time, the rate of expected growth is modest. One report suggested a growth rate of 1 to 2 mm in each dimension per year can be predicted.7 However, widespread interindividual variability exists and should be expected. Rarely, thyroid nodules will decrease in size over time. Such nodules are usually cystic in nature, and the reduction in nodule size often reflects resorption of internal fluid.
Thyroid nodule evaluation and subsequent assessment are usually performed by an endocrinologist, though a multidisciplinary team is often required for optimal care of the affected patients.8 Radiologists, pathologists, and surgeons are integral members of the care team. The evaluation of thyroid nodular disease is nearly always performed in the ambulatory setting and with minimal patient discomfort. Patients with benign nodules of average size usually require no further evaluation, and conservative management is recommended. Large nodules, however, and those with suspicion or evidence of malignancy usually require further management and are the focus of separate chapters in this text (see Chapter 7, Surgery of Cervical and Substernal Goiter).
The fundamental goal during the evaluation of a patient with a thyroid nodule is to optimally assess risk, inclusive of the disease, the diagnostic tests, and the possible treatments. With the use of various testing modalities, physicians are increasingly able to determine the probability of cancer or benign disease in any given nodule. Even if thyroid carcinoma is likely, well-differentiated malignancy may ultimately pose minimal risk to many patients even if untreated.9 Furthermore, surgery carries a small but consistent rate of complication (see Chapters 45, Pathophysiology of Recurrent Laryngeal Nerve Injury, 46, Management of Recurrent Laryngeal Nerve Paralysis, and 47, Non-Neural Complications of Thyroid and Parathyroid Surgery). Ultimately the goal in thyroid nodule evaluation and management is to provide robust cancer screening, and offer surgery to those few patients in whom it is required.
Identifying Thyroid Nodules for Evaluation
Thyroid nodules are heterogeneous in character, structure, and size. A 3-cm purely cystic nodule should be viewed differently in comparison to a 1.5-cm solid nodule with microcalcifications and irregular borders. As part of the evaluation for any nodule suspected on physical examination, a thyroid ultrasound is required. Thyroid ultrasound allows for optimal radiologic visualization of the thyroid and associated nodules. Data confirm that many nodules suspected on the basis of physical examination are not confirmed upon ultrasound examination. A study of 156 patients referred for ultrasound evaluation after clinical suspicion of a thyroid nodule or asymmetry demonstrated that ultrasound altered the clinical management for 63% of patients.10 Of 114 patients referred for clinical suspicion of a solitary nodule, ultrasound detected additional nonpalpable nodules > 1 cm in 50. Conversely, of 59 patients referred for a diffuse goiter or multinodular gland, ultrasonography determined that aspiration was unnecessary in 20. These data confirm that ultrasound frequently changes the management of patients with known or presumed thyroid nodules and support its use once a nodule (or thyroid asymmetry) is confirmed clinically.11
Additionally, many nodules > 1 cm in diameter are not palpable on examination. A prospective series of 72 patients compared clinical and ultrasonographic examination of the thyroid in detail. Nine of 22 patients (41%) with no thyroid nodule on clinical examination were found to have a thyroid nodule > 1 cm in diameter on ultrasound imaging; 62% of patients with multiple thyroid nodules > 1 cm in diameter were misclassified as having solitary or no nodules based on clinical examination; 42% of thyroid nodules > 2 cm in diameter were not detected via palpation. These data support the use of ultrasound to evaluate the thyroid when abnormal physical examination findings are detected.11
Following ultrasound examination of the thyroid, serum thyrotropin (TSH) should also be assessed.6 A minority of patients will have a suppressed TSH below the reference range. If detected, this may suggest the possibility of a functional (or toxic) adenoma. 123I thyroid scintigraphy can be performed in patients with thyroid nodules in whom serum TSH concentrations are suppressed (suggesting autonomous thyroid hormone production). If a functional (“hot”) nodule is identified, the risk of malignancy is exceedingly small. A “hot” nodule does not require fine-needle aspiration (FNA). 123I thyroid scintigraphy can also be performed on patients with multiple thyroid nodules each > 1 cm in diameter. Nonfunctional (“cold”) nodules should undergo fine-needle aspiration. Functional nodules that are part of a multinodular gland do not require FNA. Though considered reasonable based on expert opinion, the benefit or cost-effectiveness of this approach has not been demonstrated
Importantly, not all sonographically detected nodules in euthyroid patients require evaluation. In low-risk patients, most nodules < 1 to 1.5 cm should be followed without further intervention.6 This recommendation is supported by data that confirm minimal risk from well-differentiated thyroid cancer smaller than 1 to 1.5 cm. Retrospective data from 1355 patients with well-differentiated thyroid cancer evaluated in the United States confirm that tumor size < 1.5 cm (stage 1 cancer) was associated with a significant reduction in disease recurrence. Only one death (0.4%) was attributed to a thyroid cancer < 1.5 cm in diameter.12 Separate data demonstrate that malignant histologic properties such as extrathyroidal extension rarely occur in carcinomas < 1 cm in diameter.13 For these reasons, it is recommended that thyroid nodules < 1 to 1.5 cm in diameter be followed without further intervention in most situations. Exceptions to this recommendation, however, do occur. Importantly, those patients at moderate to high risk for thyroid cancer based on historical or examination findings should be considered for evaluation at smaller sizes. Among others, these groups include patients exposed to ionizing radiation during childhood (< 16 years old) and those with unique familial cancer syndromes such as Cowden’s syndrome or multiple endocrine neoplasia (MEN) 2a or 2b. In these unique circumstances, consideration should be given to evaluating thyroid nodules > 5 mm in diameter, especially if solid (noncystic) in nature.
Fine-Needle Aspiration
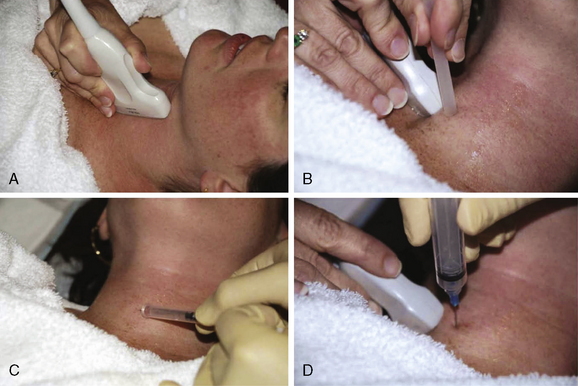
Ultrasound guidance is generally recommended for thyroid nodule FNA. Two studies have documented a significant reduction in false-negative aspirates using ultrasound guidance for FNA compared to palpation methods. One retrospective analysis of 497 patients found a lower rate of unsuccessful (nondiagnostic) aspiration with ultrasound-guided guided aspiration, as well as improved sensitivity, specificity, and cancer yield at the time of surgery. In both studies, false negative rates were < 1% using ultrasound guidance.14,15 A separate analysis of 9683 patients demonstrated 8.7% of aspirates guided via palpation were inadequate, whereas only 3.5% were inadequate for those obtained with ultrasound-guidance.15 These and other data have led to widespread adoption of ultrasound-guidance in most clinical practices. It is important to note that some physicians will aspirate thyroid nodules without ultrasound guidance (i.e., via palpation guidance) in unique circumstances. Expert consensus considers palpation-guidance reasonable for certain patients in whom ultrasound has validated the presence of a true thyroid nodule. Importantly, the nodule must also be easily identified via palpation and should only be performed on solid nodules (< 25% cystic) in patients with normal neck anatomy. Similarly, if the patient has multiple nodules or a prior thyroid ultrasound was nondiagnostic, only ultrasound should be employed for guidance. Though not demonstrated in any prospective investigation, it is also believed that ultrasound-guidance of needle aspiration reduces the risk of complications, such as bruising and pain.
The FNA procedure is usually performed with a simple 5-cc or 10-cc syringe, attached to a 25- to 27-gauge needle. Following lidocaine administration, a 5-second undulation of the needle is performed while visualizing the tip in the thyroid nodule. Between 2 and 5 separate needle samples are combined and constitute a fine-needle aspiration for one nodule (Figure 11-1). The primary adverse risk is simply localized bruising. When performed by a trained endocrinologist with ultrasound-guidance, the risk of tracheal, carotid or jugular puncture is exceedingly low. Though some patients may request a sedative to reduce symptoms of anxiety, there is no need for general anesthesia or conscious sedation during the evaluation of a typical thyroid nodule. Finalizing a diagnosis from FNA cytology usually requires 5 to 7 days. Nondiagnostic aspirations occur in 5% to 10% of cases and imply insufficient cellularity in the sample. Increasing risk of a nondiagnostic aspiration is directly related to increasing cystic content in the nodule itself. Although removal of cystic fluid from a nodule can reduce symptoms and nodule size, there is no role for the cytologic analysis of cystic fluid. Studies have confirmed that nondiagnostic thyroid nodule aspirations should be repeated a second time following healing, as up to 50% may produce a diagnostic sample during the subsequent attempt.16
When the specimen is adequate, thyroid nodule FNA cytology should be reported utilizing one of five diagnostic terms defined by the 2010 Bethesda Criteria.17 In most series, nearly 60% to 70% of samples will prove benign. This finding is highly accurate and allows for a conservative approach to future management given the lack of malignancy. Approximately 5% of samples will return positive for papillary thyroid carcinoma. This result is also highly predictive. Unfortunately, 15% to 25% of aspirations are adequate though diagnostically indeterminate. In such cases, malignancy risk is increased, though not conclusive. Three separate diagnostic categories are utilized to convey indeterminate cytology results. Cytology that is suspicious for papillary carcinoma generally implies a 60% to 75% risk of malignancy. Cytology that is suspicious for a follicular (or Hurthle cell) neoplasm implies a 20% to 30% risk of malignancy. Finally, cytology labeled “atypical of an undetermined significance” implies a 5% to 15% cancer risk. Surgery, typically a hemithyroidectomy (without frozen section), is recommended for follicular neoplasm, hemithyroidectomy (with frozen section and intraoperative touch preparation) is recommended for cytology read as suspicious for papillary cancer, and repeat FNA is recommended for cytology read as atypia of undetermined significance. With the standardization of terminology, FNA cytology results more effectively convey risk while also allowing for improved epidemiologic evaluation and comparison. At present, there is no role for core-needle biopsy in the routine evaluation of low-risk thyroid nodules.
The importance of thyroid nodule FNA in the clinical management of patients with thyroid nodules has been demonstrated in numerous investigations. In one case series performed at the Mayo Clinic, widespread adoption of fine-needle aspiration resulted in nearly a 50% reduction in thyroid surgery while still detecting similar rates of thyroid cancer.18 A meta-analysis of seven retrospective studies confirms that FNA can decrease the need for surgical thyroidectomy by 30% to 50% while increasing the diagnostic yield of surgery (the ratio of the total number of patients with carcinoma to the total number of cases operated). On the basis of clinical examination and thyroid scintigraphy, the diagnostic yield of surgery was estimated at 10% to 15%. With the introduction of fine-needle aspiration, 3144 of 18,183 patients with thyroid nodules were referred to surgery; 995 patients proved to have cancer, confirming a 32% yield. These data are among the first to support the benefits of fine-needle aspiration of thyroid nodules. A recently published experience of 4703 aspirates confirms improvement on these findings,19 demonstrating a 53% yield in the current clinical environment