20

The Diagnosis of Allergic
Fungal Sinusitis
Appropriate medical therapy for allergic fungal rhinosinusitis is predicated on recognition of the disease process. Up until 1981, patients with allergic fungal sinusitis (AFS) were mistakenly diagnosed as “immunocompetent hosts” with invasive fungal sinusitis, or the presence of the fungus was unappreciated and the diagnosis of chronic inflammatory sinusitis was given. In 1981, Millar and colleagues1 published an abstract that highlighted the similarity of the sinus histopathology to allergic bronchopulmonary aspergillosis (ABPA). Because this similarity was the basis for the original recognition of AFS, early authors assumed the causative fungus in the rhinosinusitis form of the disease was Aspergillus, just as in the pulmonary form of the disease. No fungal cultures were obtained to support this diagnosis. Nevertheless, for over half a decade, AFS was known as allergic Aspergillus sinusitis. Katzenstein, a pathologist in St. Louis, Missouri, a location that turned out to be a hotbed for AFS, also noted the distinctive sinus histopathology and its similarity to ABPA. Along with her colleagues, she published the first papers on the unique histopathology of the entity.2 Even after the recognition of the similarity of the disease process pathologically to ABPA, many authors continued to mistake this entity as infectious.3
 Spectrum of Fungal Manifestations within the Paranasal Sinuses
Spectrum of Fungal Manifestations within the Paranasal Sinuses
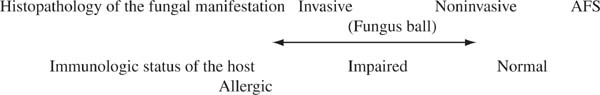
The various manifestations of fungal sinusitis depend on the immunologic competence of the host. Prognosis and treatment vary dramatically depending on whether the fungal sinusitis is invasive, noninvasive, or secondary to a hypersensitivity reaction.4
At one end of the spectrum are immunosuppressed patients, who suffer invasive fungal sinusitis from a variety of organisms, most commonly the Aspergillus species, and mucormycosis, frequently from Rhizopus species. Invasive fungal rhinosinusitis is often relentlessly fatal unless the underlying cause of immunocompromise can be reversed. Appropriate therapy includes systemic anti-fungal therapy and surgery.
Rarely, immunocompetent patients develop invasive fungal rhinosinusitis. In these individuals, the disease is chronic, persisting for months or years, and in some situations it is fatal. It is exceedingly rare in the United States. The largest series was reported in Sudan.5
Fungus balls may grow in any sinus. They occur in immunocompetent patients, and the treatment is surgical removal. They seldom recur. They differ from AFS histologically in the presence of matted hyphae without mucin, rather than scattered hyphae in allergic mucin.
At the allergic end of the immunocompetency spectrum lies AFS. An IgE-mediated hypersensitivity to the fungus or fungi is the presumed cause of the disorder. AFS is frequently recurrent, chronic, almost never life threatening, and best treated with surgery, steroids, and probably postoperative immunotherapy. Because the same fungus can cause any of these disease manifestations, one sees how patients in whom the diagnosis of AFS is unappreciated may be mistakenly treated as if they have an invasive form of the disease and inappropriately subjected to the toxicities of systemic antifungal therapy and radical exenterative surgery. Figure 20–1 details the variety of fungal manifestations along an immunologic spectrum.
FIGURE 20–1 The spectrum of fungal manifestations in the paranasal sinuses depends on the immunologic status of the host.
 Histopathology
Histopathology
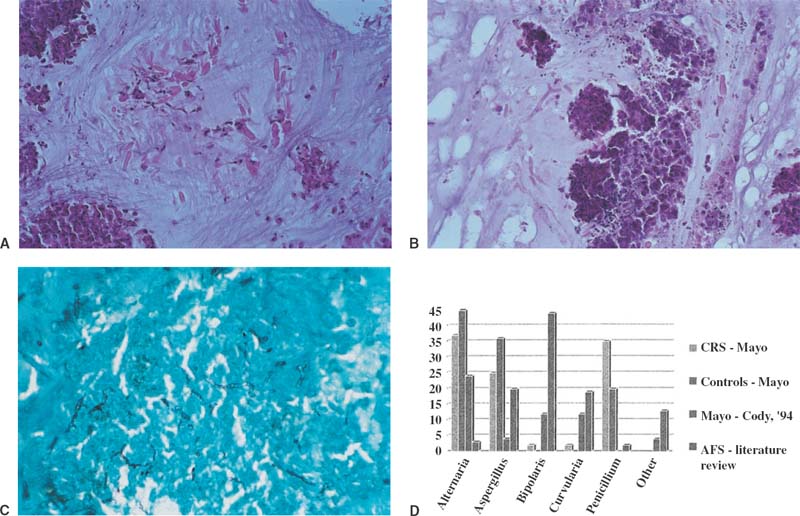
The distinctive histopathology of AFS was most likely overlooked for many years because it lies not in the polypoid tissue but rather in the tenacious inspissated mucin. This allergic mucin is often characterized as being “peanut buttery.”6 I find it more like rubber cement in its thick elastic properties that often cast the shape of the nasal or sinus cavity from which it is extracted. Under the microscope, this mucin reveals necrotic eosinophils, frequently in wavelike concentric layers. On higher power, Charcot-Leyden crystals are seen. On cross section, they may appear hexagonal, whereas on longitudinal section they are thick splinters with tapered ends. They are thought to be a product of eosinophil degranulation2 (Fig. 20–2A) Most important diagnostically is the finding of hyphal fragments scattered throughout the eosinophilic mucin. Special stains for fungi such as Gomori methenamine–silver are frequently required to see these hyphae, although they may occasionally be appreciated on hematoxylin and eosin stain (Fig. 20–2B,C). One cannot differentiate among the many fungi that cause AFS based on histopathological appearance. Most cases are not caused by the Aspergillus species, in contradistinction to the early assumptions by Millar et al1 and Katzenstein et al,2 who initially designated AFS as allergic Aspergillus sinusitis. Rather, several types of fungi, including Aspergillus, are responsible. Ultimate attribution to a particular fungal species depends on fungal culture results.7
FIGURE 20–2 (A) Histopathology of allergic mucin showing necrotic eosinophils and abundant Charcot-Leyden crystals on hematoxylin and eosin stain. (B) Histologic pathology of allergic fungal mucin showing dense inflammatory infiltrate, rare Charcot-Leyden crystals, and hyphae present. Note branching of hyphae and length, which helps distinguish hyphae from a Charcot-Leyden crystal on hematoxylin and eosin stain. (C) Gomori methenamine–silver nitrate stain of mucin showing hyphae. (D) Graph of fungal species incidence in four patient groups: CRS,35 normals,35 AFS at the Mayo Clinic reported by Cody et al (1994),16 and AFS reported in the literature.36
Preoperative steroids may sometimes obscure the diagnosis, as detailed in one case of a man with characteristic clinical findings for AFS treated with 5 days of prednisone 60 mg daily preoperatively. Histologic examination revealed fungal hyphae, but no allergic mucin or eosinophils. The patient was diagnosed as having a sphenoid fungus ball, but 2 weeks later, his symptoms returned, and endoscopic removal of sphenoid mucin now showed characteristic allergic mucin with eosinophils, Charcot-Leyden crystals, and fungal forms.8
Eosinophilic Mucin Sinusitis without Fungus: A Difficulty in the Histopathological Diagnosis of AFS
Occasionally, patients with clinical characteristics of AFS are noted to have characteristic eosinophilic mucin but no evidence of hyphae, despite special stains. In the initial report of nine patients by Katzenstein et al,2 two cases lacked fungi. One went on to have fungi noted on subsequent surgery. Allphin et al9 reported on 13 patients with this polypoid histopathology and eosinophilic mucin but absent fungi and negative fungal cultures. Interestingly, all patients in both reports had moderate to severe asthma and were aspirin sensitive. These aspirin sensitive asthmatics with nasal polyposis are sometimes referred to eponymously as having Samter’s triad. Aspirin-sensitive asthmatics frequently have severe asthma, unlike most patients with AFS, who either do not have asthma or the asthma is mild.10,11,35 This group of patients, with eosinophilic mucin sinusitis without hyphae and severe asthma that is often aspirin sensitive, represent a group of patients with a different pathophysiologic basis for their sinus disease than patients with AFS. The most probable cause of AFS as already stated is a hypersensitivity reaction to fungi. That is why the fungi are present in the mucin. They are the driving force for the inflammatory reaction. Those with eosinophilic mucin sinusitis without fungus probably have a systemic immunologic abnormality, which would account for their asthma and the involvement of all sinuses. In a review of 431 AFS patients compared with 69 eosinophilic mucin rhinosinusitis (EMRS) patients, significant differences were present between the two groups. The EMRS patients were statistically older (mean age 48 compared with 30), more likely to have asthma (93% vs. 41%), more likely to have aspirin sensitivity, also known as Samter’s triad, (54% vs. 13%), and, most significantly, never had unilateral disease. In AFS, unilateral sinus disease is present ~50% of the time.35 Patients with AFS may show unilateral disease, because the fungal stimulus is only present unilaterally, thus sparing the remaining sinuses. Patients with eosinophilic mucin sinusitis without fungus have bilateral disease because the driving force is a more systemic abnormality that accounts for their asthma and upper airway polyposis.
Ramadan and Quraishi13 noted that their four cases of such patients all had asthma and were significantly older than the AFS population. Rather than terming this entity allergic mucin sinusitis without fungus, as they did, I prefer the term eosinophilic mucin sinusitis without fungi, thus avoiding reference to the word allergic for an entity in which the underlying pathophysiology is unknown. Further investigation regarding possible differences between these two groups of patients is required, because they may respond differently to antifungal therapy and recurrence rates may differ. It certainly is true that steroid therapy is effective in both groups of patients because of its anti-inflammatory activity. Most likely steroids act by down-regulating the eosinophilic activity common to both groups. Steroids act to induce apoptosis in eosinophils. Eosinophil survival is prolonged in specimens from bronchoalveolar lavages of Samter’s triad patients.14
Further Controversies in the Diagnosis of AFS: The Mayo Clinic Experience
In 1999, the Mayo Clinic reported the presence of eosinophilic histopathology with fungi present by culture or histology in 94 of 101 (93%) patients undergoing surgery for any form of chronic rhinosinusitis (CRS). Histologic evidence of fungi, using the criterion fungal elements, further defined as hyphae, destroyed hyphae, conidia, and spores, was found in 81% of this group. The high yield of positive fungal cultures (96%) in patients with CRS in this series is difficult to interpret, because 14 of 14 normal volunteers all had fungus present when undergoing this fungal nasal/sinus collection technique. The collection technique is a nasal wash with 20 mL of saline, which is plated for mold. Those patients with eosinophilic CRS and fungi present by culture or histology in the Mayo Clinic series had a much-reduced incidence of allergy, including mold allergy, compared with other series of AFS.15 Thus, Ponikau et al15 believed that IgE-mediated inflammation is not crucial to the development of AFS, and that eosinophilic chemo-taxis and activation may result from a T lymphocyte–mediated inflammatory cascade triggered by certain fungi, especially Alternaria.
In an analysis of frequency of species of fungi found in four populations, differences and similarities are apparent. The four populations illustrated in Figure 20–2D are
1. Patients with CRS with eosinophilia undergoing nasal washes by Ponikau et al15
2. Normals undergoing nasal washes by Ponikau et al15
3. Patients with AFS reported by Cody et al 5 years earlier also at the Mayo Clinic16
4. Fungi reported in the literature associated with AFS
Differences can be seen even within the same institution. Cody et al6 reported that a far larger number of patients with AFS grow Bipolaris or Curvularia, two fungi frequently implicated in other series of AFS, but rarely seen in the more recent Ponikau et al15 series. Distribution of other fungal species, such as Alternaria and Aspergillus, for example, are fairly high in both of Ponikau’s groups, CRS and normals, with no pattern emerging. In the literature, Bipolaris is the most frequently reported mold associated with AFS. This could reflect the greater publication on AFS from authors in Texas, where Bipolaris is the most common cause, than Bipolaris truly being responsible for most cases of AFS worldwide.
Saprophytic Fungal Colonization in EMRS Non-AFS patients
It is possible that the EMRS patients previously discussed can become colonized with a small amount of fungus, but the fungus is not driving the disease process. As Ponikau et al15 have shown, normals also have mold in their nose. For AFS to exist, it may be important that the fungi germinate and grow in the mucin, further fueling an IgE-mediated allergic reaction. Green and colleagues17 showed that IgE-mediated response to fungi is increased with germination. This same antigenic stimulation may not be present if just a few ungerminated mold spores are present in the nose.
 Fungal Cultures
Fungal Cultures
Fungal cultures are positive in ~70 to 80% of patients diagnosed with AFS.18 Unlike all other areas of the country, the Mayo Clinic reports virtually 100% positive fungal cultures on all patients, irrespective of pathology.15 This points out the possibility of false positives if one relies on fungal cultures, given the ubiquity of fungal spores. Why all patients with histopathologic evidence of AFS do not grow a fungus on fungal cultures has been attributed to overhomogenization of the specimen prior to plating, which disrupts the fragile hyphal walls, a delay in plating and incubating the culture, and inappropriate culture techniques to separate the fungi from the thick mucin in which they grow. Some advocate immediate culture to Sabouraud’s agar at the time of harvesting with incubation.19 Even if the fungus grows, it may be impossible to speciate it. Speciation requires the production of spores, and if the fungus remains in an asexual form, this may be impossible.
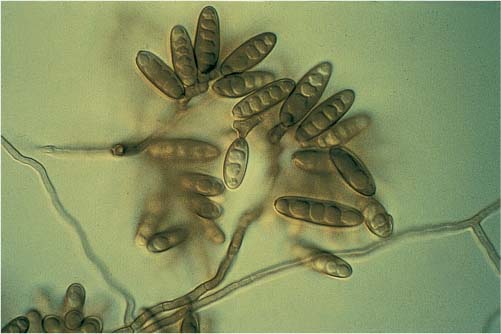
FIGURE 20–3 Photomicrograph of Bipolaris.
Dematiaceous fungi are the most common based on culture data and account for 84% of the total positive cultures.7,20 Dematiaceous fungi is a term referencing the darkened color of these fungi on histopathologic examination in tissue. It does not refer to the color of the fungi when growing in vitro. The most common fungi were Bipolaris species, followed by Curvularia, Alternaria, and Exserohilum (Fig. 20–3). Aspergillus species accounted for 13% of all fungal cultures (Table 20–1).
Unusual causes of AFS include Epicoccum nigrum20 and Schizophillum commune,21 as well as a Nodulisporium species.22 The clinical manifestations, response to therapy, and histopathology are similar no matter which fungus is associated with the process. If immunotherapy directed to the fungus causing the hypersensitivity response is important, then culture information will be increasingly important. Unfortunately, no commercial antigens to Bipolaris or to many of these unusual fungi are available.
Organism | Positve Cultures (n) | % of Total* |