Fig. 3.1
(a) Histology of the human cornea stained with hematoxylin and eosin (H&E). (b) Corneoscleral border. (c) Limbal junction. Reprinted with permission from Takacs et al. [3]
At the limbal zone, where the corneal meets the conjunctival epithelium, the corneal structure changes. The epithelium thickens and forms epithelial pegs made up of 11–12 cell layers instead of 5. Bowman’s and Descemet’s layers are missing and the undulated epithelium basement membrane lies above the limbal stroma. The collagen bundles become less organized and cells are abundant and fibroblast-like. The endothelial cells here are larger and flatter than in the central cornea. The bulbar conjunctiva becomes more adherent to Tenon’s capsule; and these tissues blend to make up radiating ridges called palisades of Vogt. This area is thought to contain corneal epithelial stem cells [1–3] (see Fig. 3.1c). Although it has been hard to definitively identify limbal epithelial stem cells, a breakthrough came with the identification of transient amplifying cells (TACs). These cells are a differentiated and rapidly proliferating epithelial cell type and are found both in the peripheral and central cornea, but not in the limbus, providing further evidence for the existence of stem cells specifically in the limbus [5].
Corneal endothelial cells (CEC) originate from the neural crest [6] and have a monolayer arrangement. The endothelial cell density increases from the center to the periphery [7]. CECs are connected by focal tight junctions that create a semi-permeable barrier, which allows water and nutrients from the aqueous humor to enter the stroma. CECs also contain ionic pumps that expel water from the stroma [8]. This fluid balance maintains corneal clarity.
Definition of Corneal Stem Cells
Adult stem cell populations are found in most tissues. These are cells that are able to maintain and regenerate a given tissue for a long time. They are characterized by three main properties. First, the cells must be capable of self-renewal: one of the dividing cells must always remain a stem cell. Second, the cells must remain in an undifferentiated state but maintain the potential for differentiation. Finally, the cells must remain in a growth-arrested state, supported by a microenvironment that provides external factors necessary for maintaining stem cell properties, until they are required to differentiate [9].
Corneal epithelial stem cells can be stimulated to divide and differentiate into transient amplifying cells. They continue to divide into terminally differentiated corneal epithelial cells and migrate centripetally for the continued replacement and regeneration of tissues following injury. This maintains a steady-state population of healthy cells.
Corneal stromal cells are stromal keratocytes, of neural cell origin. They help form an interconnected network of stromal lamellae and are responsible for the deposition of stromal extracellular matrix and for maintenance of corneal transparency. Stromal stem cells have the ability to differentiate from keratocytes into fibroblastic cells in the face of irritation such as wounds, infections, and corneal pathologies. Keratocyte-derived fibroblasts secrete fibrotic matrix components that disrupt corneal transparency but allow for wound healing and strength [10, 11].
Corneal endothelial stem cells (CESC) are particularly important because corneal endothelial cells have decreased proliferative capacity, particularly after the age of 20 years [12]. CESCs are believed to be located in the transition zone of the posterior limbus and to be active after corneal wounding [13]. However, much more research is required to understand how CESCs respond to endothelial damage.
Limbal Epithelial Stem Cells
The corneal epithelium is a continuously regenerating surface tissue. In 1971, Davanger and Evenson proposed that the limbus could be the reservoir of new epithelial cells when they first characterized the central migration of cells from the limbus [14]. Further studies described the distance of migration in a mouse model to be 94 ± 14 μm in 7 days [15]. In support of the capability of self-renewal, Cotsarelis’ group noted that a population of slow cycling cells labeled with tritiated thymidine retained an easily detectable amount of thymidine while the amount of label per cell quickly decays in more frequently dividing cells [16] (see Fig. 3.2). From the percentage of thymidine retaining cells in the limbal zone, it has been concluded that stem cells may represent less than 10 % of the total limbal basal cell population [17]. Dua and Forrester were able to demonstrate that limbal cells were involved in wound healing, proving that these limbal cells met all criteria of stem cells [18]. Furthermore, central cornea cells are biochemically different than limbal cells based on keratin expression providing further evidence that they are a distinct population of cells [19].
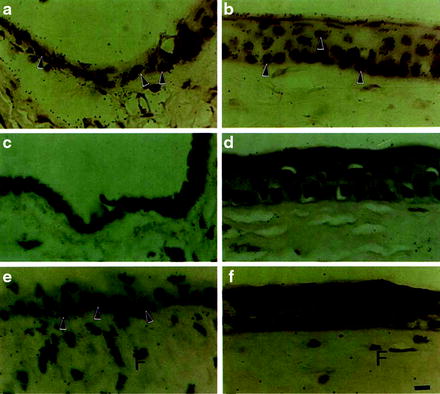

Fig. 3.2
Epithelial stem cells. Identification of label-retaining cells (LRC) in limbal epithelium. Autoradiograms demonstrate the labeling pattern of limbal (a, c, e) and central corneal (b, d, f) epithelia after long-term labeling under various conditions. After 2 weeks of continuous 3H-TdR labeling (a, b), nuclei of almost all corneal and limbal epithelial cells contain silver grains (arrowheads). Most of these labeled cells disappeared from both limbal (c) and corneal (d) epithelia after a 4-week chase. Wounding during the continuous labeling followed by a similar 4-week chase revealed a population of label-retaining limbal basal cells (arrowheads) in (e). No LRCs were observed in the corneal epithelium (f). Some fibroblasts (F) in limbal (e) and corneal stroma (f) also retain labels: these cells were probably stimulated to incorporate 3H-TdR during wounding and then returned to a normal state during chasing. Calibration bar (a–f) = 8 pm. Reprinted with permission from Cotsarelis et al. [16]
Once limbal stem cells were identified, many groups were able to culture and further identify stem cell features of these cells. Pelligrini’s group was able to demonstrate the expression of alpha enolase, alpha-6 integrin, low CD71, and no connexin 43 [20]. They also showed an increased expression of nuclear p63 by clonal analysis [21].
Since these advances were made, the location and expression of limbal epithelial stem cells (LESCs) as well as their migration have become a subject of debate. As recently as 2010, Majo et al. demonstrated that despite extensive limbal cell damage, corneal integrity and clarity were maintained for 4 months, claiming that oligopotent stem cells exist throughout the cornea surface and that the limbal, central, and conjunctival epithelium all share similar properties [22]. In addition, others have found that despite clinically evident total limbal cell deficiency, corneal epithelial regeneration is maintained, providing further evidence that corneal epithelial stem cells may be present throughout the cornea epithelium [23].
Limbal stem cells are now accepted to be of small size and have a high nucleus to cytoplasm ratio. They are normally quiescent, which is thought to reduce the possibility of DNA damage. Limbal stem cells retain the possibility of differentiating into many different and distant cell types including hair follicles. Conversely, epidermal stem cells are capable of differentiating into corneal epithelial cells and repairing a damaged corneal surface in the setting of total limbal stem cell deficiency, as shown in a goat model [24].
Limbal stem cell deficiency (LSCD) is characterized by persistent or recurrent epithelial defects, ulceration, corneal vascularization, chronic inflammation, scarring, and conjunctivalization of the cornea with loss of clear demarcation of the cornea and conjunctival epithelium at the limbus [25] (see Fig. 3.3). Causes of LSCD are both hereditary and acquired. Hereditary causes include aniridia and keratitis associated with multiple endocrine deficiency (MEN), in which stem cells may be congenitally absent or dysfunctional. Acquired causes are much more common in clinical practice and include Steven–Johnson syndrome (SJS), chemical injuries, ocular cicatricial pemphigoid (OCP), contact lens-induced trauma, neurotrophic degenerations, and peripheral ulcerative keratitis.
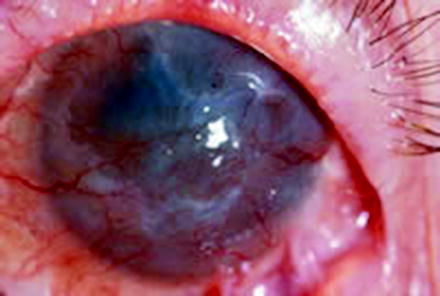

Fig. 3.3
Limbal stem cell deficiency in a 25-year-old patient with Steven–Johnson syndrome
The chronic instability of the corneal epithelium caused by LSCD leads to persistent corneal epithelial breakdown, superficial corneal vascularization, chronic discomfort, and impaired vision. This may lead to progressive melting of the cornea, and can cause subsequent perforation. This sequence has been demonstrated in animal models in which removal of the limbus resulted in insufficient re-epithelialization and conjunctival invasion of corneal surface [26]. Furthermore, during wound healing it has been found that there is an increase in the number of cells in the limbal area [27].
Stromal Stem Cells
The main cell type of the stroma is the keratocyte. In response to wound healing, keratocytes proliferate and become fibroblastic. This enables them to secrete fibrotic matrix components creating a more structurally stable interface at the expense of corneal clarity. Early work with keratocytes aimed to elucidate environmental influences on morphological changes; however, multiple groups found evidence of stromal stem cells or, at very least, stromal progenitor cells in both human cell lines and animal models. These progenitor cells are differentiated from primary keratocytes in a few key ways. They have the ability to form spontaneous aggregations of attachment independent spheroids. These spheroids are solid balls of viable cells that express high levels of keratocyte-specific genes. They express a number of genes known to be up-regulated in multiple stem cells types, such as Notch-1, Bmi1, SCF, FHLIM1, ABCG2, CD73, CD90, and CD166. They also express high levels of the genes PAX6, Six3, Six2, all of which are associated with eye development [28, 29]. Further support for these stromal stem cells comes from work where the progenitor stromal cells of C57BL6/J mice were cultured and conditioned to differentiate into either stromal fibroblasts or a-SMA-positive myofibroblasts, or to maintain primary keratocyte properties if cultured in a different serum media [30] (see Fig. 3.4).
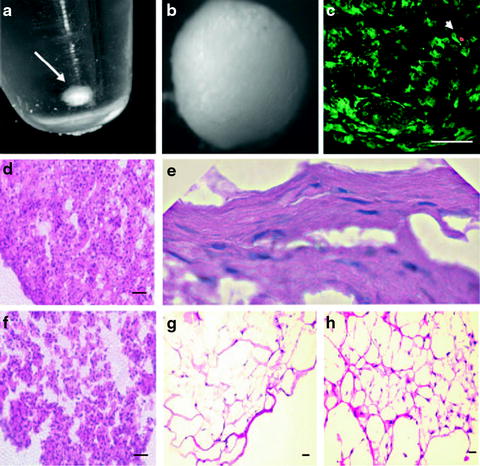

Fig. 3.4
Stromal stem cells. Three-dimensional cultures of human corneal stromal stem cells (hCSSC). (a) Formation of a free-floating pellet after centrifugation of 2 × 105 hCSSC in a 15-mL conical polystyrene tube (arrow). (b) Pellet has formed a smooth sphere after 1 week of culture. (c) Identification of viable cells after staining with calcein AM (green) and dead cells using propidium iodide (red, arrowhead) after 3 weeks of culture. (d) H&E staining of hCSSC cultured 3 weeks as a pellet. (e) Flattened cells near the periphery of an hCSSC pellet. (f) H&E of a pellet formed by fibroblasts cultured for 3 weeks. (g) Stained section of hCSSC cultured in a fibrin gel for 3 weeks. (h) Fibroblasts cultured in fibrin gel. Scale bars, 50 μm. Reprinted with permission from Du et al. [31]
Though the field of stromal stem cell research is in its earliest stages, it provides an optimistic outlook for providing bio-prosthetic stromal material. The stromal progenitor cells have been shown to exhibit multipotent differentiation potential. Perhaps most importantly, corneal stromal stem cells have been shown to be capable of secreting an organized tissue-like extracellular matrix resembling that found in the corneal stroma. This matrix can be populated with keratocyte-like cells, with significant biomedical engineering implications [31].
Endothelial Stem Cells
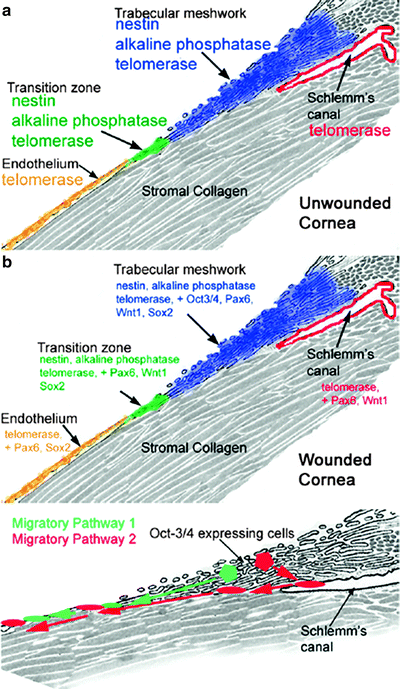
In 2005, Whitehart first demonstrated the existence of corneal endothelial stems cells (CESC). His group showed increased telomerase activity and incorporation of bromodeoxyuridine into cellular DNA after endothelial damage, which is suggestive of cellular division [32]. This work was expanded upon by McGowan in 2007, who found the stem cell markers nestin, alkaline phosphatase, Oct-3/4, Pax-6, Sox-2 and Wnt-1 in addition to telomerase in the trabecular meshwork and the transition zone of the posterior limbus, with several of these markers appearing in the endothelial periphery only after corneal wounding [13]. These studies formed the foundation for demonstrating the existence of corneal endothelial stem cells (see Fig. 3.5).
Corneal endothelial cells in general have a comparatively reduced ability to divide [12], which has prompted research into the factors governing CESC division. Though the governing mechanisms of CESC proliferation are not yet fully understood, the most studied inhibiting factor of CESC proliferation is transforming growth factor-beta2 (TGF-beta2), which is present in the aqueous humor [33]. In multiple animal studies, TGF-beta2 has been shown to modulate endothelial wound healing. The endogenous presence of TGF-beta2 has been shown to be necessary for growth, while its exogenous addition may inhibit division [34, 35]. Regulators of CESC division will require further investigation to obtain a more complete understanding.
CESCs are of particular importance since the cells of the corneal endothelium lose their proliferative capacity with age, and may lose all proliferative capacity by the age of 20 years old [12]. Due to endothelial cells’ decreased ability to proliferate, damage will often lead to a decreased endothelial cell density. Additionally, endothelial cells are susceptible to damage during common surgeries, such as cataract removal, and in the course of degenerative and dystrophic diseases. Though CESCs seem to respond to endothelial damage [13], the endothelium’s primary response is endothelial flattening and expansion by approximately 25 % in order to cover the damaged area [36]. This change in cellular form may compromise the ability of the endothelial layer to retain its barrier function, making research and understanding of CESC proliferation and methods of transplantation [37] to be of particular therapeutic importance.
Current Treatments of Corneal Stem Cell Deficiencies
The potential for regeneration of the corneal epithelial surface has stimulated the creation of numerous techniques to replace limbal stem cells. These can be classified as one of the following procedures: conjunctival autograft (CAU), conjunctival allograft (CAL), conjunctival limbal autograft (CLAU), cadaveric conjunctival limbal allograft (c-CLAL), living related conjunctival limbal allograft (lr-CLAL), or keratolimbal allograft (KLAL).
In 1977, Thoft first described epithelial transplantation for severe ocular surface disease when he reported conjunctival transplantation for monocular chemical bums. By using the fellow eye he was able to avoid a major problem facing the allograft procedure, namely, immunologic rejection. Next Kenyon and Tseng modified the above procedure, naming it the conjunctival limbal autograft (CLAU) in 1989 [38]. This technique takes grafts of bulbar conjunctiva that extended approximately 0.5 mm onto the clear cornea centrally, thus containing limbal corneal cells. These techniques are now widely used by many corneal surgeons. However, the use of the technique is limited to cases where the donor eye is healthy, which subsequently puts it at risk for stem cell deficiency itself [39]. Another drawback includes the inability to use these techniques for ocular surface disease patients with the greatest need, namely, those affected severely bilaterally. A further modification came when Kenyon and Rapoza described a technique they called limbal allograft transplantation, giving rise to the lr-CLAL, in which they transplanted limbal tissue with a conjunctival carrier from a living-related donor. This technique was similar to the previously described technique of CLAU, except that the donor tissue was obtained from a living relative as opposed to the fellow eye. Postoperative management included topical corticosteroids [39, 40–42].
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree