(1)
University of Sydney, Sydney, Australia
The Cornea
Overview
The cornea is a transparent structure at the front of the eye.
It is a powerful refractive surface and a robust barrier that protects the ocular contents.
1.
2.
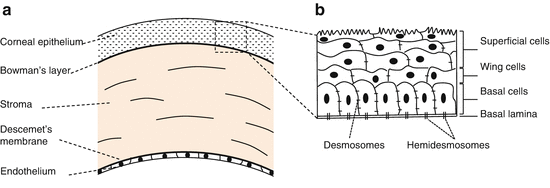
Structure (Fig. 3.1) [4]
(i)
The epithelium
The epithelium is a continuously renewed superficial layer of cells.
It interacts with the tear film to provide a smooth optical surface.
(ii)
The stroma
The stroma, a predominantly extracellular matrix, makes up the bulk of the corneal volume.
It determines the structural and optical properties of cornea.
(iii)
The endothelium
The endothelium, the innermost portion, is a highly metabolically active single-cell layer.
It allows entry of nutrients from the aqueous into the stroma and removal of water from the stroma.
3.
Optical properties
The cornea has a higher refractive index than air (1.376 vs. 1.0).
Together with the tear film, the cornea is the major refractive component of the eye.
4.
Corneal transparency is achieved through:
The highly ordered arrangement of the corneal collagen lamellae
The uniform length, diameter, and spacing of collagen fibrils within the lamellae
The glycosaminoglycan matrix that maintains the regular crystalline arrangement of fibrils
The endothelial pump that removes fluid from the cornea, maintaining stromal dehydration
Epithelium (Fig. 3.1b)
1.
Cell types
There are three cell types (from surface to basement membrane): superficial, wing, and basal cells [4].
(i)
Superficial cells
These form three to four layers.
They are terminally differentiated cells that degenerate and slough from the surface.
They have apical surface projections (microvilli) that express an adherent glycocalyx that anchors the tear film (See Chap. 2, The Ocular Surface) [12].
They include small light cells (recently arrived) and superficial large dark cells (soon to be sloughed) [13].
(ii)
Wing cells
These form the intermediate one to three layers of the epithelium [14].
They are partially differentiated with characteristic wing-shaped processes.
(iii)
Basal cells
These form a single layer of cuboidal cells adherent to a basement membrane.
Mitotic activity for epithelial cells occurs in the basal layer [14].
They originate from stem cells in the basal layer of the limbal (peripheral corneal) epithelium.
Each basal cell divides into two wing cells which subsequently differentiate into superficial cells [11].
As cell division occurs, daughter cells move toward the corneal surface and begin to differentiate.
Basal cells rest on a basement membrane of type IV collagen, laminin, fibronectin and fibrin [15].
3.
Cell basement membrane adhesion
Junction | Cytoskeletal proteins | Function |
|---|---|---|
Desmosomes (macula adherens) | Intermediate filaments, cadherins | Anchor cell membranes of adjacent cells to each other |
Hemidesmosomes | Intermediate filaments, integrins | Anchor cell membranes to their basement membrane |
Adherens junctions | Actin filaments, cadherins, integrins | Transmembrane anchors similar to desmosomes and hemidesmosomes |
Gap junctions | Connexons | A low-resistance intercellular passage allowing direct chemical communication between adjacent cells through diffusion |
Tight junctions (zonula occludens) | Transmembrane proteins | The fusion of lipid bilayers of adjacent cells, forming a low permeability paracellular barrier |
4.
Corneal epithelial migration
Corneal epithelium is maintained by a constant cycle of shedding of superficial cells and proliferation of mitotically active basal cells [11].
Basal cells proliferate and migrate superficially and centrally; most proliferate at the limbus (palisades of Vogt) from where there is a centripetal migration of cells.
This is known as the X, Y, Z hypothesis of corneal epithelial maintenance (Fig. 3.2) [21].
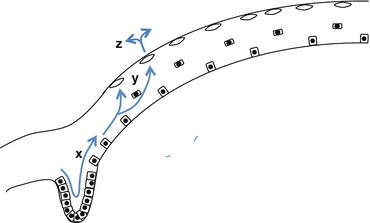
Fig. 3.2
The X, Y, Z hypothesis of corneal epithelial cell migration: centripetal migration (x), superficial migration (y), and then sloughing off the surface (z) [21]
A similar pattern of proliferation and migration occurs after epithelial injury [22].
5.
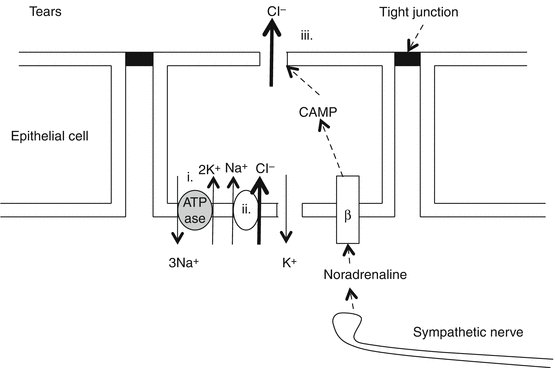
Control of transepithelial flow of solutes
The corneal epithelium acts as a barrier to preserve stromal homeostasis.
The epithelial cell membranes are joined by tight junctions that prevent water and solutes entering from the tear film [17].
Stroma
The stroma makes up 90 % of corneal thickness.
1.
Bowman’s layer
2.
Lamellar structure
The stroma is composed of 200–250 highly organized lamellae that run parallel to the corneal surface (Fig. 3.4).
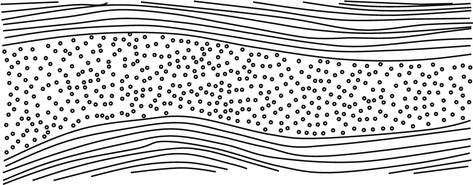
Fig. 3.4
Orthogonal arrangement of corneal lamellae
These are bundles of colinear collagen fibrils approximately 2.0 um thick and 9–260 um long [32, 33].
The lamellae lie oblique to one another anteriorly and orthogonally posteriorly.
At the limbus they form an annulus 1.5–2.0 mm in diameter; this maintains corneal curvature [34].
3.
Collagen fibrils
4.
Proteoglycan matrix
The side chains (chondroitin sulfate, dermatan sulfate and keratin sulfate) are perpendicular to the protein backbone and highly negatively charged.
The electrostatic forces help maintain the collagen fibril lattice arrangement [40].
5.
Keratocytes (stromal fibroblasts)
6.
Stromal hydration
The stroma has an inherent tendency to imbibe water and to swell, with negatively charged proteoglycans that cause excess Na+ to accumulate [44].
Excess hydration can degrade light transmission.
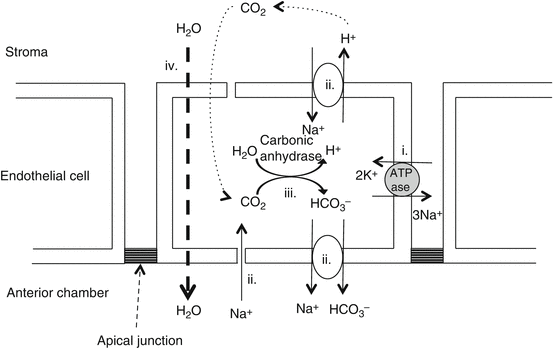
Endothelial cells continuously pump water from the cornea to prevent overhydration (see below).
7.
Pre-Descemet’s layer
Pre-Descemet’s layer is a well-defined acellular layer located between Descemet’s membrane and the deepest row of stromal keratocytes [45].
It provides a natural strong cleavage plane between Descemet’s membrane and the stroma.
Descemet’s Membrane
Descemet’s membrane is a 10–15 um thick basement membrane of the corneal endothelium [4].
It is composed of type IV collagen, laminin, and fibronectin [46].
It is secreted by endothelial cells and increases in thickness throughout life [47].
It is tough and relatively resistant to proteolytic enzymes; it may remain intact despite severe overlying stromal destruction in corneal inflammatory disease [48].
Endothelium
The corneal endothelium consists of a single layer of mostly hexagonal cuboidal cells.
1.
2.
Endothelial function: aqueous metabolic pump
3.
Endothelial cell count and morphometry
Endothelial cells generally do not replicate; however, a limbal stem cell source has been identified with limited replicative ability in response to injury [56].
Newborns have 5500 cells/mm2, while adults have 2500–3000 cells/mm2.
A minimum of 400–700 cells/mm2 is required for normal corneal function; however endothelial decompensation can occur at higher counts [59–61].
Stable endothelium has a uniform size and shape. Stressed or unstable endothelium demonstrates polymegathism (cells of varying size) and pleomorphism (cells of varying shape) [62].
Corneal Innervation
1.
2.
Neural structure
The cornea is innervated by the anterior ciliary nerves, branches of the ophthalmic nerve (V1).
Bare nerves enter limbally in the mid-stroma and run anteriorly and radially toward the center, forming a stromal plexus [67].
Branches perforate Bowman’s layer to form a subepithelial plexus that innervates basal cells [14, 64].
Corneal sensitivity is greater centrally than peripherally; it is greater superiorly than inferiorly.
3.
Neurotransmitters
Substance P and calcitonin gene–related peptide are involved in pain transmission and modulation of pain and inflammation. They are important epithelial cell trophic factors [14, 65, 67, 70].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree