Chronic rhinosinusitis (CRS) is the single most common self-reported chronic health condition in the United States and is estimated to affect 16% of the adult population annually. Despite the prevalence of this disease, there still exists an incomplete understanding of CRS pathophysiology. In this review, the authors highlight technological advances in rhinology: real-time polymerase chain reaction, epithelial cell culture, flow cytometry, genomics/single-nucleotide polymorphism detection, microarrays, and genetic/nongenetic animal models of sinusitis. The purpose of this review is to describe these methodologies and their contributions toward achieving a better understanding of CRS.
Chronic rhinosinusitis (CRS) is the single most common self-reported chronic health condition in the United States and is estimated to affect 16% of the adult population annually. Despite the prevalence of this disease, there still exists an incomplete understanding of CRS pathophysiology. Due to the lack of effective therapies directed at CRS refractory to medical and surgical therapy, it is critical that we develop a more mature molecular understanding of CRS and CRS with nasal polyps. Research advances over the years have shifted from CRS emerging from sinus ostial obstruction and persistent bacterial infection to newer mechanisms focusing on inadequate host immune responses, persistent bacterial biofilms, and fungal colonization. Technological advances have helped to embrace many of these new mechanisms. In this review, the authors highlight technological advances in rhinology: real-time polymerase chain reaction, epithelial cell culture, flow cytometry, genomics/single-nucleotide polymorphism detection, microarrays, and genetic/nongenetic animal models of sinusitis. The purpose of this review is to describe these methodologies and their contributions toward achieving a better understanding of CRS.
Real-time PCR
The polymerase chain reaction (PCR) has revolutionized molecular research over the past several decades. PCR is a method that allows exponential amplification of short DNA sequences within a longer double-stranded DNA (dsDNA) and requires the use of primers that are complementary to a defined sequence on each of the 2 strands of DNA. These primers are then extended by a heat stable DNA polymerase (Taq polymerase) to complete the sequence, leading to logarithmic amplification. Historically, PCR products were run on an agarose gel and stained with an ethidium bromide stain because the reaction was qualitative, determining the presence or absence of a product. Another variation of this technique is reverse transcriptase PCR (RT-PCR), which uses the enzyme reverse transcriptase to convert mRNA to cDNA for use in a PCR reaction. Real-time PCR was developed in efforts to quantitatively assess DNA copy number.
Real-Time PCR: the Technique
Real-time PCR has the ability to monitor the progress of PCR as it occurs “in real time.” In this technique, reactions are characterized by the point in time during cycling when amplification of a target is first detected, rather than the amount of target accumulated after a fixed number of cycles. The higher the starting copy number of the gene or nucleotide sequence, the sooner a significant increase in fluorescence is detected. In contrast, conventional PCR and RT-PCR measure the amount of accumulated PCR product at the end of the cycle.
Two primary fluorescence-based sequence detection systems are used: Taqman and SYBR Green chemistry. Taqman chemistry uses fluorogenic labeled probes to enable the detection of PCR product as it accumulates. In the Taqman system, specific hybridization between the probe and target is required to generate a fluorescent signal. Probes can also be labeled with different distinguishable reporter dyes, which can allow for amplification of 2 distinct sequences in 1 tube. The primary disadvantage of this system is that specific probes need to be designed for each reaction. SYBR Green dye is a highly specific dye that binds to all dsDNA. As amplicons are created during PCR, SYBR Green binds to all dsDNA causing an increase in fluorescence proportional to the amount of PCR product created ( Fig. 1 ). The primary advantage of the SYBR Green system is that no probe is required, which reduces assay time. This system, however, may generate false-positive signals as SYBR Green dye will bind to any dsDNA.
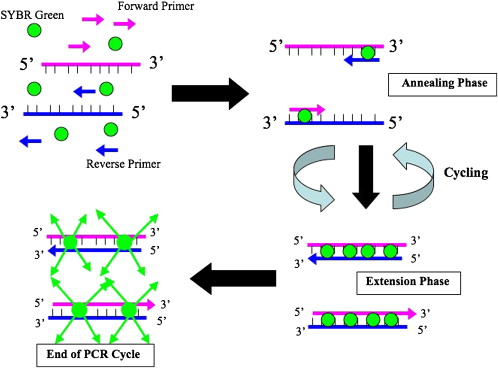
Real-time PCR data are acquired by a computer and can be analyzed in various ways. Cycle thresholds (C T ) reflect the fractional cycle number of a gene or sequence at which the fluorescence passes the fixed threshold, which is a sample that does not contain a template. The ΔC T method uses the difference in C T value obtained between a normalizing housekeeping gene (18 S ribosomal RNA, GAPDH) and the target gene to calculate relative quantification (ΔC T = the difference in threshold cycles for target and housekeeping gene).
Real-Time PCR Applications in Rhinology
Although conventional PCR and RT-PCR have been used for decades in rhinologic research to evaluate for the qualitative presence of markers/genes in nasal tissue, quantitative real-time PCR has largely grown to replace this technique over the past 5 years. Claeys and colleagues and Lane and colleagues initially demonstrated the expression of toll-like receptors (TLRs), human beta defensins, and costimulatory molecules from nasal epithelial cells using real-time PCR. Richer and colleagues demonstrated marked reductions in the level of expression of several genes involved in epithelial barrier maintenance and repair in the inflammatory state of CRS. Furthermore, numerous studies have shown altered sinonasal innate immune epithelial gene expression including TLRs, interleukin 22 (IL22), TLRs, and lactoferrin in patients with CRS compared with normal patients. Although this literature review is incomplete, it serves to demonstrate that real-time PCR is a powerful rapid method to screen for alterations in genes/markers between control populations and CRS.
In vitro cell culture systems
The hallmark of patient-oriented basic science research is studying the differences between diseased and nondiseased tissue samples. Harvesting sinonasal mucosal biopsies in the operating room is critical to better understanding the cellular and molecular differences in CRS. These tissue biopsies are primarily used either for immunohistochemical staining or to isolate mRNA for PCR. The limitations of these biopsies include small amounts of tissue and the inability to biologically manipulate mucosal cells. Currently, there are no immortalized nasal cell lines from either normal patients or those with CRS. There have been reports of establishing primary nasal fibroblast cultures and of groups using immortalized lower respiratory epithelial cell lines (A549 and BEAS-2B) given the similarities between the upper and lower airways.
Primary Sinonasal Epithelial Cells in Culture/Brushed Epithelial Cells
A new advance in rhinologic research has been the use of primary sinonasal epithelial cells (SNECs) grown in culture at the air-liquid interface. This model involves extracting nasal epithelial cells from mucosal biopsies and growing them in cell culture ( Fig. 2 A) as previously described. After these cells reach confluence, they are split into transwell inserts with air above the insert and medium below ( Fig. 3 ), mimicking the natural microenvironment of the nose. These cells eventually become ciliated, and histologically resemble fresh brushings of SNECs. Another novel technique to acquire sinonasal tissue is mucosal brush biopsy of the middle meatus, which can be performed in the clinic using topical anesthesia ( Fig. 2 B). The advantage of brushed epithelial cells is that they can be collected easily in the clinic with local anesthesia. This technique also facilitates longitudinal evaluation of epithelial cell function in nonoperative patients. These brushings can be used in cell culture or can be used for PCR or primary analysis by immunohistochemistry or flow cytometry (see later discussion). SNECs can be stimulated/suppressed with exogenous cytokines and their effector function can be determined by analyzing the cells for gene/protein expression or the culture medium can be analyzed for secreted proteins.
Recent Advances in Rhinology Research Using SNECs
Numerous investigators have used SNECs from CRS patients in vitro to stimulate the production of cytokines and other effectors. Lalekar and colleagues demonstrated that SNECs can be stimulated with chitin to produce the Th2-associated molecule, acidic mammalian chitinase (AMCase), and eotaxin-3. This finding was the first to suggest the possible existence of an innate immune pathway for local defense against chitin-containing organisms in the sinonasal tract. It is possible that dysregulation of this function could precipitate or exacerbate Th2 inflammation, potentially acting as an underlying factor in CRSwNP. Using SNECs, Kim and colleagues showed that the nasal epithelial costimulatory molecules, B7-H1 and B7-DC, are inducible and elevated in CRS compared with controls. In addition, through using brushed nasal epithelial cells, Saatian and colleagues demonstrated that SNECs express B7-H2 and B7-H3 and may act as antigen-presenting cells to activated mucosal T cells. Clearly, the SNEC model, which mirrors the natural mucosal epithelium of the nose, holds great promise to study the effects of various pharmacologic and environmental stimuli.
In vitro cell culture systems
The hallmark of patient-oriented basic science research is studying the differences between diseased and nondiseased tissue samples. Harvesting sinonasal mucosal biopsies in the operating room is critical to better understanding the cellular and molecular differences in CRS. These tissue biopsies are primarily used either for immunohistochemical staining or to isolate mRNA for PCR. The limitations of these biopsies include small amounts of tissue and the inability to biologically manipulate mucosal cells. Currently, there are no immortalized nasal cell lines from either normal patients or those with CRS. There have been reports of establishing primary nasal fibroblast cultures and of groups using immortalized lower respiratory epithelial cell lines (A549 and BEAS-2B) given the similarities between the upper and lower airways.
Primary Sinonasal Epithelial Cells in Culture/Brushed Epithelial Cells
A new advance in rhinologic research has been the use of primary sinonasal epithelial cells (SNECs) grown in culture at the air-liquid interface. This model involves extracting nasal epithelial cells from mucosal biopsies and growing them in cell culture ( Fig. 2 A) as previously described. After these cells reach confluence, they are split into transwell inserts with air above the insert and medium below ( Fig. 3 ), mimicking the natural microenvironment of the nose. These cells eventually become ciliated, and histologically resemble fresh brushings of SNECs. Another novel technique to acquire sinonasal tissue is mucosal brush biopsy of the middle meatus, which can be performed in the clinic using topical anesthesia ( Fig. 2 B). The advantage of brushed epithelial cells is that they can be collected easily in the clinic with local anesthesia. This technique also facilitates longitudinal evaluation of epithelial cell function in nonoperative patients. These brushings can be used in cell culture or can be used for PCR or primary analysis by immunohistochemistry or flow cytometry (see later discussion). SNECs can be stimulated/suppressed with exogenous cytokines and their effector function can be determined by analyzing the cells for gene/protein expression or the culture medium can be analyzed for secreted proteins.
Recent Advances in Rhinology Research Using SNECs
Numerous investigators have used SNECs from CRS patients in vitro to stimulate the production of cytokines and other effectors. Lalekar and colleagues demonstrated that SNECs can be stimulated with chitin to produce the Th2-associated molecule, acidic mammalian chitinase (AMCase), and eotaxin-3. This finding was the first to suggest the possible existence of an innate immune pathway for local defense against chitin-containing organisms in the sinonasal tract. It is possible that dysregulation of this function could precipitate or exacerbate Th2 inflammation, potentially acting as an underlying factor in CRSwNP. Using SNECs, Kim and colleagues showed that the nasal epithelial costimulatory molecules, B7-H1 and B7-DC, are inducible and elevated in CRS compared with controls. In addition, through using brushed nasal epithelial cells, Saatian and colleagues demonstrated that SNECs express B7-H2 and B7-H3 and may act as antigen-presenting cells to activated mucosal T cells. Clearly, the SNEC model, which mirrors the natural mucosal epithelium of the nose, holds great promise to study the effects of various pharmacologic and environmental stimuli.
Flow cytometry
The technique of flow cytometry or fluorescence-activated cell sorting (FACS) has revolutionized single-cell protein analysis and sorting based on cellular protein markers, surface and intracellular. Traditionally, Western blots have been used to measure specific protein expression in cellular or mucosal lysates. The enzyme-linked immunosorbent assay (ELISA) is another technique to measure soluble secreted protein. Unless the tissue source is homogenous, like cell lines, Western blots or ELISA cannot determine which cell type is expressing or secreting a certain protein. Flow cytometry, however, has the powerful ability to sort individual cell populations based on multiple fluorescent markers specific for various receptors or proteins.
Flow Cytometry: the Technique
Flow cytometry uses the principles of light scattering, light excitation, and emission of fluorochrome molecules to generate data from particles and cells. Individual cells and particles are hydrodynamically focused in a sheath of saline before intercepting a focused laser light source. Cell samples are first stained with fluorochrome-conjugated monoclonal antibodies. These stained cells then intercept the light source and scatter light, and fluorochromes are excited to a higher energy state. This energy is then released as a photon of light with specific properties unique to different fluorochromes. Ultimately, flow cytometry measures fluorescence per cell or particle. The analyzed cells can be sorted and collected for further analysis or the data can be stored in the form of a computer file, which can be analyzed as a histogram or quadrant. Most commonly, flow cytometry data are depicted as a 4-quadrant grid with an x and y axis, depicting different fluorochromes ( Fig. 4 ). Each cell is depicted as a dot on the plot and its position reflects the fluorochrome intensity. A sample dot plot is shown in Fig. 4 ; 60% of the cells in this brushed cell sample are epithelial cells expressing TLR9.
Recent Rhinologic Advances Using Flow Cytometry
Numerous studies have used flow cytometry to elucidate the pathogenesis of CRS. In 1996, Bernstein and colleagues analyzed different lymphocyte populations in nasal polyps from patients with CRS. This study found that nasal polyp lymphocyte subpopulations may be derived from the local mucosal immune system as well as from random migration of peripheral blood lymphocytes secondary to adhesion molecules and chemokines, which are known to be present in nasal polyps. Conley and colleagues used flow cytometry to measure superantigen-specific T cell receptor domains in the peripheral blood of patients with nasal polyps. This study revealed that 7/12 patients with nasal polyps had a local superantigen effect on T cells. Our group has used flow cytometry extensively to analyze SNECs in control patients and those with CRS with polyps and has shown decreased Toll-like receptor 9 (TLR9) and interleukin 22 receptor (IL22RA-1) levels associated with CRSwNP.
Mucociliary clearance and ciliary beat frequency
Ciliary dysfunction is another proposed mechanism for CRS. Respiratory cilia beat in a coordinated manner with a specific frequency and pattern. It is this coordinated beating that clears debris laden mucus toward the natural sinus ostia in a well-established pattern. Studies have shown that stressors such as exercise or infection can alter ciliary beat frequency (CBF). Numerous studies have also shown a decrease in sinonasal mucociliary clearance in CRS patients. Possible reasons for impaired mucus transport include reduced basal CBF and impaired mechanical mucociliary coupling.
Measuring CBF
CBF can be measured in vivo using the saccharin transit test or scintigraphy, however these techniques do not take into account regional variations of CBF within the nasal cavity. Also, these studies do not differentiate between the role played by ciliary activity and the overlying mucus blanket. A new advance in measuring CBF has been the advent of high-speed digital microscopy. Numerous groups have developed methods of using digital microscopy and acquisition software. In 2003, Sisson and colleagues developed a fully digital imaging system to quantify CBF accurately and more efficiently compared with analog techniques. Dimova and colleagues adapted a similar system to measure CBF in primary nasal epithelial cells with fast data acquisition and calculation. One area of controversy among in vitro measurements of CBF is the effect of temperature on ciliary function. Some groups take CBF measurements at room temperature, whereas others perform these studies at physiologic body temperature.
To control for the effects of temperature on CBF, Schipor and colleagues developed a novel system using differential interference contrast (DIC) microscopy and to measure CBF at physiologic body temperature. This technique involves placing nasal epithelial cell explants into a temperature-controlled glass perfusion chamber. Microscopic images are visualized using a water immersion objective with DIC optics. Images are then captured using a high-speed monochromatic digital video camera at 250 frames per second. Video images are then analyzed using video imaging processing software on areas of epithelium containing beating cilia, and the frequency is calculated.
Recent Advances Using CBF Measurements
Several investigators have found altered CBF dynamics in CRS patients. Chen and colleagues analyzed CBF in nasal epithelial cells from controls and CRS patients before and after exogenous stimulation with adenosine triphosphate. This study found no differences in basal CBF in controls and CRS patients but found that CRS patients had a minimal increase in CBF after ATP stimulation, concluding that this patient cohort has decreased sinonasal ciliary adaptation to environmental stimuli. Furthermore, Erickson and colleagues used the same method to quantify CBF to demonstrate that retinoic acid is beneficial in ciliary regeneration in stripped maxillary sinus mucosa from rabbits. This study found that CBF was increased in retinoic acid treated mucosa compared with controls. Use of this enhanced technology to measure CBF will allow for a better understanding of how various environmental stimuli affect mucociliary clearance in CRS patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


