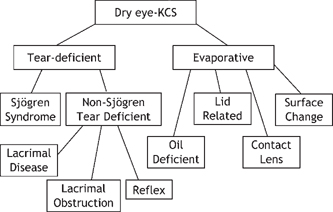
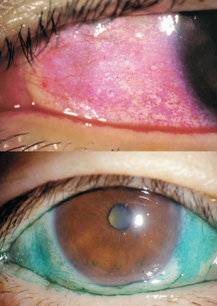
15 Shigeru Kinoshita and Norihiko Yokoi Sjögren’s syndrome is an autoimmune disease more prevalent in women than men that presents with dysfunction of the general exocrine glands, especially the lacrimal and salivary glands, due to gland destruction by infiltrating lymphocytes. It is classified into primary and secondary Sjögren’s syndrome. In the United States, the male to female ratio of Sjögren’s syndrome patients is 1:20. Secondary Sjögren’s syndrome involves other collagen diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and Hashimoto’s disease. In ophthalmology, Sjögren’s syndrome is representative of aqueous tear-deficient (ATD) dry eye diseases in which ATD causes ocular surface epithelial damage (keratoconjunctivitis sicca) that leads to ocular irritation, such as foreign body sensation and photophobia, as well as a reduction in the quality of vision. The first international classification1 categorized dry eye into two types: ATD dry eye and evaporative dry eye (Fig. 15-1). ATD dry eye is subclassified further into Sjögren’s syndrome and non-Sjögren’s syndrome. Patients with ATD dry eye but not Sjögren’s syndrome are diagnosed as having the non-Sjögren’s type of ATD dry eye; this diagnosis includes lacrimal gland disease, lacrimal duct obstruction, and dysfunction of the neural reflex loop (see later).1 In general, for a diagnosis of Sjögren’s syndrome, there must be ATD dry eye, the presence of autoantibodies (including antinuclear antibody, rheumatoid factor, anti-SS-A/R. antibody and anti-SS-B/La antibody), and a histopathological demonstration of destruction of the lacrimal or salivary glands together with infiltration of tissues by lymphocytes. A combination of examinations yields several diagnostic criteria for Sjögren’s syndrome. For example, with the aid of criteria proposed by Fox et al,2 a clinical diagnosis of ATD dry eye is made first; the subsequent verification of dry mouth leads to further examination of serum and the small salivary glands (lip biopsy). Figure 15-1 Classification of dry eye, or keratoconjunctivitis sicca (KCS).1 Sjögren’s syndrome is classified as the aqueous tear-deficient type of dry eye. Figure 15-2 Representative example of rose bengal (upper) and lissamine green (lower) staining of the conjunctiva in Sjögren’s syndrome. For a diagnosis of ATD dry eye, it is necessary to demonstrate a tear volume decrease; the Schirmer’s 1 test3 or determination of tear meniscus parameters such as meniscus height4 is appropriate. Conjunctival examination is now recognized as important because conjunctival inflammation may also be involved in the mechanism of conjunctival damage in ATD dry eye, especially in Sjögren’s syndrome.5,6 It is therefore important to identify epithelial damage in the conjunctiva by fluorescein staining using a blue-free barrier filter7 or by traditional rose bengal or alternative lissamine green8 staining (Fig. 15-2). Representative abnormal values for these examinations are summarized in Table 15-1. Although the pathogenesis of ocular surface epithelial damage and symptoms in Sjögren’s syndrome has been explained only from the point of ATD, at least five mechanisms, as described in the following sections, are involved. The ocular surface comprises the corneal and conjunctival epithelium and the tear film, which serves as a lubricant that protects the epithelium from the mechanical action of blinking as well as from desiccation while the eye is open. Therefore, when the tear volume decreases as a result of lacrimal gland dysfunction, the epithelial cells are exposed to desiccation, and superficial cells may become desquamated as a result of blink-related mechanical effects. The consequent epithelial damage may be a major factor in dry eye symptoms. For an insight into the mechanisms of ATD dry eye, the interactions between the tear film and the ocular surface epithelium must be understood (Fig. 15-3).
Systemic Issues and Dry Eye Disease
Key Points
♦ Sjögren’s Syndrome and Aqueous Tear-Deficient Dry Eye
Definition, Classification, and Diagnosis of Sjögren’s Syndrome
Pathophysiology of Sjögren’s Syndrome
Interactions between the Tear Film and Epithelium
| Test | Implication of Examination | Abnormal Values |
| Schirmer’s 1 test | Function from reflex loop to lacrimal gland | ≤5 mm in 5 minutes |
| Fluorescein breakup time | Tear film stability | ≤5 seconds |
| Fluorescein staining | Superficial corneal epithelial damage | Depends on the classification |
| Rose bengal staining (or lissamine green staining) | Condition of mucin coverage on ocular surface epithelium | Depends on the classification |
| Impression cytology | Epithelial differentiation of conjunctiva | Depends on the classification |
The tear film comprises the uppermost lipid layer and the lower aqueous layer in which goblet cell-derived mucin is dissolved. The lipid, mucin, and lipocalin reduce the surface tension of the aqueous layer9; they not only prevent tear film instability but also prevent desiccation of the ocular surface epithelium. In ATD dry eye, however, because of a decrease in the aqueous tear volume, the tear film becomes thinner and unstable, resulting in early tear film breakup and consequent epithelial damage (Fig. 15-4). Tear film breakup induces blinking, and patients with severe ATD dry eye cannot keep their eyes open for many seconds; this results in frequent blinking. Also, because of the dense distribution of sensory nerve endings on the ocular surface, epithelial damage produces severe ocular symptoms despite the diminution of corneal sensitivity in patients with severe ATD dry eye. Two types of mucin—membrane-binding and secretory mucin—are expressed in the tear film, and their interaction plays a major role in tear film stability.10 As damaged epithelium cannot express mucin, there is a decrease in epithelial moisture.11 In ATD dry eye, tear film instability and the concomitant decrease in epithelial moisture disrupt the intimate relationship between the tear film and epithelium, leading to a vicious cycle that results in chronic epithelial damage.
Feedback System for Mutual Interactions between Tear Film and Epithelium
Even if the relationship between the tear film and the ocular surface epithelium is disturbed, there is a feedback system that leads to restoration to a healthy condition (Fig. 15-3) because reflex tears are secreted by the lacrimal glands. This stimulatory tear secretion involves the reflex loop that comprises sensory nerves of the ocular surface, the brain stem, and the accessory nerves. The production of reflex tears results in an increase in the tear volume and constitutes a compensatory mechanism to resolve tear film instability and assist in the recovery of a healthy relationship between the tear film and the ocular surface epithelium. Because of the lacrimal gland dysfunction in ATD dry eye, however, the feedback system cannot halt the vicious cycle between the tear film and the ocular surface epithelium, and the consequent chronic epithelial damage persists. This is true especially in Sjögren’s syndrome, although in patients with secondary Sjögren’s syndrome or non-Sjögren’s ATD dry eye (Fig. 15-5), there may be partial recovery of epithelial damage depending on the lacrimal gland function or other accompanying collagen diseases. Therefore, the more severe the lacrimal gland destruction, the more severe the resultant corneal epithelial damage in Sjögren’s syndrome because there is less support from the feedback system. Thus, the Schirmer’s 1 test,3 which examines the quantity of reflex tears produced in response to conjunctival surface irritation, should be performed to assess the efficacy of the feedback system.
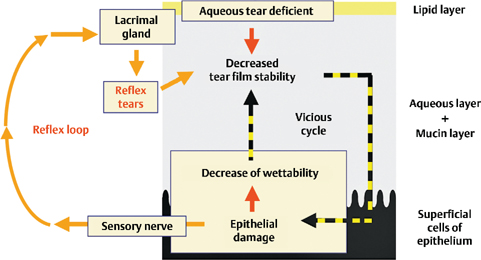
Figure 15-3 Possible mechanism underlying aqueous tear-deficient (ATD) dry eye at the interface between the tear film and the superficial epithelium of the ocular surface. In ATD dry eye, the tear film is thin and unstable due to a decrease in the aqueous tear volume. This results in early tear film breakup, epithelial damage, and a decrease in epithelial moisture. This disruption of the intimate relationship between the tear film and epithelium causes a vicious cycle. In ATD dry eye, because of the dysfunction of the lacrimal gland, the feedback system via the neural reflex loop cannot resolve the vicious cycle, and, as a consequence, there is chronic epithelial damage.
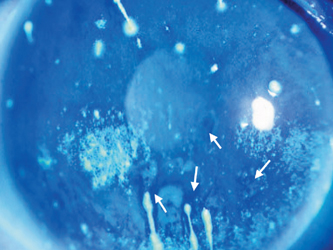
Figure 15-4 Representative example of fluorescein-stained cornea in severe Sjögren’s syndrome. Superficial punctate keratopathy, fluorescein breakup (arrows), and corneal filaments can be seen.
Coverage of the Aqueous Tear Layer by the Lipid Layer
Tear film lipid, secreted from the meibomian glands, forms a tear film lipid layer and suppresses evaporation of the aqueous layer. At the time of eye opening, the lipid layer spreads on the aqueous layer as a carrier of lipid. In the most severe form of ATD dry eye disease, however, the aqueous layer is too thin to carry the lipid layer on its surface. This results in the loss of lipid layer coverage on the aqueous layer and considerably accelerated evaporation (Fig. 15-6).12 Although Sjögren’s syndrome contains a category for tear-deficient but not evaporative dry eye (Fig. 15-1),1 in its severest form there may be loss of both the aqueous and lipid layer and the presentation may be a combination of ATD dry eye and evaporative dry eye. This explains why these patients may present with superficial punctate epitheliopathy involving the entire cornea that cannot be adequately addressed with the frequent instillation of eyedrops alone.
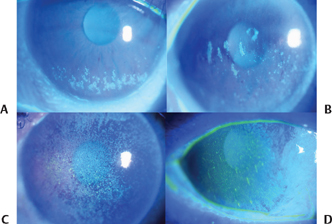
Figure 15-5 Representative examples of mild (A), moderate (B) and severe (C,D) epithelial keratopathy in patients with Sjögren’s syndrome and ATD dry eye.
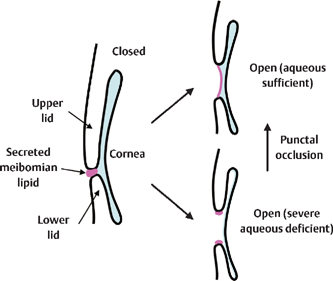
Figure 15-6 Suggested mechanisms underlying the combination of evaporative dry eye and severe aqueous tear-deficient dry eye disease. In eyes with a thicker aqueous layer, meibomian lipid can spread and form a lipid layer (the precorneal tear film). In eyes with an extremely thin aqueous layer due to severe aqueous tear deficiency, the meibomian lipid remains at the lid margin and is not available for the formation of the lipid layer. Occlusion of both upper and lower puncta can help to spread the meibomian lipid to form the lipid layer of the tear film by increasing the aqueous tear volume.
Inflammation in the Tears and the Conjunctiva
The possibility of a connection between immunological inflammation and the pathophysiology of dry eye has been suggested. In Sjögren’s syndrome, there is lymphocyte infiltration into the conjunctiva13 and various cytokines (e.g., interleukin [IL]−6, IL−8, IL−1α, and tumor necrosis factor α [TNF]-α have been found in the tears of sufferers.5 Moreover, the conjunctival epithelium in Sjögren’s syndrome expresses abnormal keratin (keratins 6, 16, 17) and overexpresses a gene induced by interferon-γ (IFN-γ Th−1—related inflammation).14 Preservative-free cyclosporin emulsion (0.05%) has been used to suppress the inflammation of the tears and conjunctiva in this disease.15 In patients with Sjögren’s syndrome with mild ATD, the conjunctival epithelium frequently is more seriously damaged than the corneal epithelium. This suggests that conjunctival damage is not the result of aqueous deficiency but rather of inflammation because inflammation interferes with normal conjunctival epithelial differentiation, resulting in squamous metaplasia and the decreased expression of mucin. The former can be demonstrated by impression cytology and the latter by rose bengal and lissamine green (Fig. 15-2), which reportedly stain ocular surface epithelium that lacks mucin coverage.16 It is therefore possible that both stains detect the inflammatory aspect of Sjögren’s syndrome.
Delayed Tear Clearance
Theoretically, the tear flow decreases with the tear volume, and this results in delayed tear clearance.17 Depending on the severity of the decrease in the tear volume, clearance of the aqueous component of tears is delayed and results in the accumulation of mucus, which in turn results in the development of mucus-related corneal complications such as corneal filaments or corneal mucous plaques (Fig. 15-7) that produce the most annoying irritation symptoms in Sjögren’s syndrome patients. Corneal filaments consist of a mucous core that is surrounded by regenerated corneal epithelium, and they are attached to the desquamated epithelial surface epithelium. These filaments connected to the highly sensitive corneal surface are pulled forward during blinking and cause severe irritation. On the other hand, corneal mucous plaques, seen in patients with relatively severe ATD dry eye, consist of dried mucus attached to the corneal surface epithelium; their formation is related to a delayed mucus turnover. The most severe form of Sjögren’s syndrome is often accompanied by corneal filaments and corneal mucous plaques, and these patients may require treatment with punctal plugs.
Treatment of Sjögren’s Syndrome Based on Pathophysiology
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


