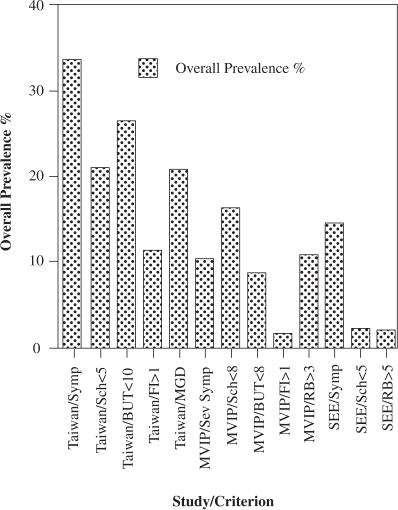
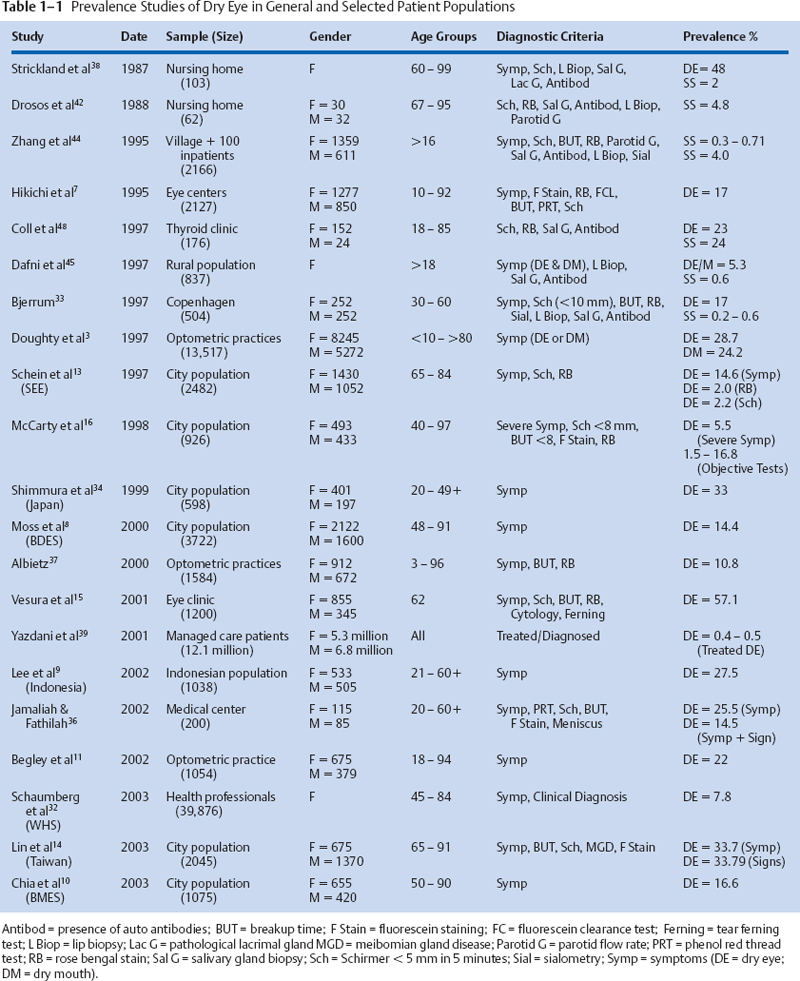
1 Alan Tomlinson A moist film over the ocular surface is necessary for the health and normal function of the eye and visual system. Essential for a complete tear film are adequate and continuous tear production and an appropriate elimination of tears through evaporation, absorption, and drainage. If anything interferes with the balance between these factors, a condition of dry eye or keratoconjunctivitis sicca (KCS) can result. The syndrome that makes up dry eye disease represents a heterogeneous group of conditions with multifactorial etiologies.1 These disorders have features in common that are embraced by a single definition developed by the National Eye Institute(NEI)/Industry Workshop on Clinical Trials in Dry Eyes2: “Dry eye is a disorder of the tear film due to tear deficiency or excessive tear evaporation which causes damage to the interpalpebral ocular surface and is associated with symptoms of ocular discomfort.” There is no cure for dry eye, and it is one of the leading causes of patient visits to ophthalmologists and optometrists in the United States.1 The potential size of the problem is great, with more than 4 million elderly Americans having frequent symptoms and signs of the condition; in the population as a whole, close to 33% report having occasional symptoms of dry eye.3 Given the scale of the problem, several studies have attempted to determine the prevalence of dry eye. In recent years significant information has emerged on the prevalence in the general population, the types of dry eye, and the potential risk factors for the condition. A problem emerging from these studies of dry eye disease is the lack of a single definition for the condition and the differing criteria employed in the various prevalence studies. Dry eye, or KCS, is a rubric for several clinical diseases characterized by a disturbance in the preocular tear film resulting in ocular surface disease.4 It can occur in many different eye disorders that disturb tear volume, composition, and hydrodynamic factors. The consequent tear instability that results from deficiencies of one or more of the three layers of the tear film needs to be identified so that effective therapy, specific for each tear deficiency state, can be initiated.5 Therefore, accurate determination of the prevalence of dry eye and its successful treatment depend on making an accurate diagnosis of the condition. Several subjective assessments and objective diagnostic tests are used clinically to measure the integrity and stability of the preocular tear film. The utility of these techniques is hampered by the lack of widely accepted criteria for each type of dry eye and the lack of correlation between the diagnostic tests, the reported symptoms, and the conditions.6 Several studies of dry eye prevalence rely only on reports of symptoms for the definition of the condition.3,7–12 It is appropriate to diagnose dry eye from symptoms as the condition rarely progresses to cause damage to the ocular surface in the absence of symptoms.13 But others have argued5 that symptoms alone are not sufficient for differential diagnosis of dry eye as patients with a range of different tear film disorders have symptoms of ocular irritation, and the same symptoms are often experienced by patients with different types of dry eye.5 Ideally, symptoms and objective signs of dry eye are needed for diagnosis and measurement of prevalence rates, but this is where the problem arises when comparing prevalence studies. Dependence on symptoms or signs, or both, as criteria for dry eye leads to apparent variability in prevalence (Fig. 1-1; Table 1-1). As an example, in the Salisbury Eye Evaluation (SEE),13 it was found that of the symptomatic patients, only one in six had a low Schirmer’s 1 test readings, one in seven had rose bengal staining, and only one in four had both these clinical signs of dry eye. In the group symptomatic of dry eye measured by Lin et al,14 80% had low tear breakup times, 63% had low Schirmer’s readings, and 63% had meibomian gland disease (MGD). In the symptomatic patients observed by Versura,15 only 57% had objective signs of dry eye. But perhaps the best illustrations of the difficulty of applying both symptoms and signs to identify dry eye are given by McCarty,16 who found variable and limited relationships within clinical signs and between signs and the reports of severe symptoms. The poor agreement between objective tests for dry eye is shown by the four clinical tests (Schirmer’s 1, tear breakup time, and rose bengal and fluorescein staining) performed on this population study in Melbourne. Out of the 635 patients in the study, only four showed agreement between three of the tests and only one between all four tests. The poorest correlate with severe symptoms was Schirmer’s 1 test, with only 12% of patients with low Schirmer’s 1 test results complaining of severe symptoms; the best correlate was fluorescein staining with 33% of patients with staining complaining of severe symptoms.16 Figure 1-1 The prevalence rates found for dry eye in three general population studies depend on the criteria for diagnosis (symptoms or clinical measurements). The studies are the Melbourne Visual Impairment Project (MVIP),16 Salisbury Eye Evaluation (SEE),13 and the Shihpai Eye Study (Taiwan).14 BUT, breakup time; Fl, fluorescein stain; MGD, meibomian gland disease; RB, rose bengal stain; Sch, Schirmer; Symp, symptoms. The poor correlation between symptoms and signs of dry eye has been frequently reported in the literature.2,16–18 There are several reasons for this finding. First, symptoms may precede signs of dry eye. Second, dry eye is a heterogeneous condition differing in etiology and pathophysiology,4 and different objective tests measure different aspects of tear physiology. Also, symptoms of dry eye are not specific to a particular disorder,16 but a single objective test for dry eye is of limited value without a report of symptoms.13,16 The sensitivity and specificity of all tests of dry eye vary with the characteristics and biases of the study population.17 There is also high test/retest variability in both questionnaires and objective tests for dry eye.19 In addition, it should be remembered that the tear system is extremely volatile and the components are interdependent,20 so the ideal measurements of tear physiology in dry eye diagnosis would be “simultaneous” (with all measurements taken at the same instant in time). This is obviously impossible. Finally, because many of the objective tests for tear physiology are to some degree invasive, one measurement may affect subsequent tests, and care must be exercised in the order in which measurements are performed (i.e., least to most invasive). In view of the difficulties of diagnosis of dry eye in prevalence studies, the use of a test battery (or panel) of measurements for dry eye has been suggested. The importance of establishing diagnostic criteria (based on accepted batteries of tests) was first identified by the NEI/Industry Workshop on Clinical Trials in Dry Eyes.2 The dry eye workshop proposed four global tests for dry eye: The first three tests are readily available: several validated questionnaires of symptoms have been devised,3,11,21,22 ocular surface damage can be measured by rose bengal staining,21,24 and tear instability can be tested by breakup time.25,26 But tear osmolarity is much more difficult to measure, currently requiring laboratory techniques involving high technical expertise.27 Several different sets of criteria for the definition of dry eye (KCS), usually including Sjögren’s syndrome, have been developed.4, 28,29 The need for a comprehensive and agreed-on set of tests to define the array of dry eye conditions is illustrated by Table 1-1 and Figure 1-1. The table illustrates the various levels of prevalence found for dry eye in different studies. This variability is due, in part, to the different criteria applied in definition. It is anticipated that new criteria based on designated subjective and objective tests for dry eye will emerge from an ongoing series of meetings of the reconvened NEI/Industry Workshop on Dry Eye that began in 2004. There was no information available until recently on the incidence of dry eye; incidence is the proportion or percentage of new cases within a given time (usually an annualized incidence rate). But Moss et al in 2004 reported a 5-year incidence rate for symptomatic dry eye disease in the Beaver Dam Eye Study (BDES).30 Because of the difficulties of carrying out longitudinal studies of dry eye in sufficiently large population groups, cross-sectional prevalence, or the proportion or percentage of persons with dry eye disease (including old and new cases of persons with the condition) at a specific time, is more frequently reported in the literature. In considering the occurrence of dry eye disease, the information on prevalence will be discussed first. The prevalence of dry eye disease depends on the type, its method of diagnosis, and the population under study. In recent years several significant population-based studies of the prevalence of dry eye disease have been performed8,9,13,14,16,31,32 (Table 1-1). A population-based prevalence survey (the SEE project) included 2520 residents of Salisbury, MD, aged 65 years and older in September of 1993.13,31 Dry eye was determined by a standardized questionnaire, Schirmer’s 1 test, rose bengal staining, and anatomic assessment of meibomian glands. In this population, 14.6% had one ore more reports of symptoms often or all the time, 2.2% had symptoms and a low Schirmer’s 1 test result (≤5 mm) of strip wetting, and 2% were symptomatic and had high rose bengal test scores (s≥5). Furthermore, 3.5% had symptoms and either a low Schirmer’s 1or a high rose bengal score, and 0.7% were symptomatic and had both a low Schirmer’s 1 and a high rose bengal score. Anatomic features of meibomianitis were occasionally associated with symptoms, but 76% of individuals with these features had no symptoms.13 When the symptom of dry mouth was combined with a report of dry eye often or all the time, the incidence of “dryness” rose from 14.6 to 27.4%, 4.4% of the sample reporting both dry eye and dry mouth symptoms.31 Bjerrum33 reported the presence of dry eye (KCS) in the Danish population aged 30 to 60 years. The study covered 514 patients recruited from the Copenhagen City Heart Study whose reported symptoms were recorded together with results from Schirmer’s 1 test, tear breakup times, and rose bengal staining of the ocular surface. Sialometry and autoantibody status were also recorded for the diagnosis of primary Sjögren’s syndrome. The prevalence of dry eye (KCS) was found to be 11% according to the Copenhagen criteria and 8% according to the preliminary European criteria.4 In the BDES cohort of 3722 patients aged from 48 to 91 years, a prevalence of dry eye by self-reported history at the 5-year follow-up visit was found to be 14.4%. The Melbourne Visual Impairment Project (MVIP)16 of age-related eye disease in people 40 years and older had a total of 926 patients participating. Dry eye was diagnosed in 10.8% by rose bengal staining, 16.3% by Schirmer’s 1 test, 8.6% by tear breakup time, 1.5% by fluorescein staining, 7.4% with two or more signs of the condition, and 5.5% with any severe symptom. In an epidemiological study of dry eye in elderly Chinese in Taiwan,14 Lin et al found that of the 1361 participating in the study, 33.7% had symptoms of the condition (reporting one or more dry eye symptom often or all the time). Among those with symptoms, 78.9% had a low tear breakup time (≤10 seconds), 62.5% had a low Schirmer’s 1 test (≤5 mm), 61.7% had abnormal anatomic features of the meibomian glands, and 85.4% had symptoms and either a low Schirmer’s 1 score or abnormal meibomian gland appearance. In a Japanese population study of dry eye disease, a high proportion (33%) was found to have symptoms of the condition.34 In an Indonesian study of dry eye, Lee et al9 found that 27.5% of the 1058 participants aged 21 years and over reported one or more dry eye symptoms often or all the time. In a later Australian study, Chia et al10 found that of the 1174 patients in the Blue Mountains Eye Study (BMES) aged 50 years and older, at least one dry eye symptom was reported by 57.5% of participants, with 16.6% reporting moderate to severe symptoms. In a large 2003 study of 39,876 women in the United States participating in the Women’s Health Study, dry eye disease was reported in 5.7% of women younger than 50 years of age and in 9.8% of women older than 75 years. An age-adjusted prevalence of dry eye was calculated at 7.8% for women older than 50 years in the U.S. population.32 In preliminary findings from the Physicians’ Health Study.1 the age-standardized prevalence for clinically diagnosed dry eye was 2.3%, 1.9% for severe symptoms, and 3.5% for the prevalence of symptomatic dry eye in men 55 years and older. The prevalence of dry eye found by studies of specific patient populations is generally higher than those obtained from general population samples as a result of substantial participation biases17 (Table 1-1). This is particularly the case for populations from eye centers. Such populations are those for which the biases favor inclusion of patients with more-severe disease and with systemic conditions such as Sjögren’s syndrome.17 A study of 2127 consecutive new outpatients seen in Japanese eye centers7 found dry eye in 17% of patients by objective measurements, including vital staining, tear breakup time, and phenol red cotton thread,35 Schirmer’s 1, and fluorescein clearance tests. In an Italian eye center, of 1200 patients reporting dry eye symptoms, 57.1% had dry eye diagnosed by objective techniques, including Schirmer’s 1 test, tear ferning, tear breakup time, vital dye staining, and impression cytology. At a Malaysian eye center, 200 outpatients aged 20 years and older and without any ocular surface disorder were tested by cotton thread and Schirmer’s 1 tests, tear meniscus assessment, fluorescein corneal staining, and tear breakup time measurements. The prevalence of dry eye in this sample population, as defined by the presence of symptoms and an abnormal test result, was found to be 14.5%.36 In optometric practices, the Canada Dry Eye Epidemiological Study (CANDEES)3 found that for a sample aged 10 to older than 80 years, 20.7% reported dry eye symptoms; concurrent dry mouth was reported by 24.2%. Of the 3716 patients reporting symptoms, 1.6% of symptoms were said to be severe and 7.8% constant but moderate. In optometric practices in Australia, a sample of 1584 subjects showed an overall prevalence of 10.8% for dry eye by the criteria of symptoms, tear film instability, and ocular surface staining.37 Strickland et al38 in a study of 103 elderly Caucasian women older than aged 63 years living in a Washington retirement home found 39% had symptoms of dry eye and 24% abnormal Schirmer’s tear test. A large study of managed care populations39 reported a prevalence for dry eye, identified by a dry eye diagnosis or treatment by punctual occlusion. The prevalence of treated dry eye in these populations was between 0.4 and 0.5% of the more than 10 million patients from 22 managed care facilities studied. Patients in the over 65 age group were found to have a four times greater risk of requiring treatment for dry eye. Several studies have attempted to determine the prevalence of different subcategories of dry eye, the subclassification being based on the cause as determined by tear film pathophysiology. Most of these studies have concentrated on the prevalence of Sjögren’s syndrome.40 Estimates of population prevalences vary from 0.05 to 4.8% (Table 1-1 ). In the study of elderly women in a Washington state retirement home, Strickland et al37 found 2% satisfied the criteria for Sjögren’s syndrome and 12% were suggestive of the syndrome. This compares with Sjögren’s own estimate of an incidence of 1 in 2000 individuals (0.05%) in the general population37 and to Shearn, who estimated it at 1 in 525 (0.2%).41 An autopsy series of 900 sequential individuals’ parotid glands revealed four with classic histological findings of Sjögren’s syndrome, suggesting a prevalence of 1 in 225 subjects (0.44%).41 Drosos42 examined 62 elderly patients in a public nursing home in Greece for evidence of primary Sjögren’s syndrome and found that three had primary Sjögren’s syndrome from labial salivary gland biopsies together with other objective criteria. This suggested a higher incidence of the ocular condition, which he concluded is relatively common in elderly people but may be subclinical and benign. A British study reported a 3.3% incidence of Sjögren’s syndrome in a geriatric inpatient population of 122 individuals.43 In a study of the prevalence of primary Sjögren’s syndrome in China, Zhang et al44 found an incidence of primary Sjögren’s syndrome of 0.77% by the Copenhagen criteria33 and 0.33% by the modified San Diego criteria.4 The Chinese sample of 2066 persons were age 16 years and older and had less of the elderly population reported by the previous researchers Among a group of 100 inpatients reported by Zhang,44 four were found to have primary Sjögren’s syndrome. Dafni et al,45 in the study of the prevalence of Sjögren’s syndrome in a rural community in Greece using the European criteria, found a prevalence of 0.6% in patients meeting all the criteria and probable primary Sjögren’s syndrome in 2.99%.45 A study in Denmark by Bjerrum33 of 504 persons aged 30 to 60 years in Copenhagen diagnosed primary Sjögren’s syndrome in only one person according to the Copenhagen criteria but three persons according to the preliminary European criteria. The frequency of primary Sjögren’s syndrome in this group was estimated at between 0.2 and 0.8% according to the Copenhagen criteria and between 0.6 and 2.1% according to the preliminary European criteria.33 The MVIP found 0.2% of the population had Sjögren’s syndrome.16 As might be anticipated, the proportion of Sjögren’s patients seeking corneal services at tertiary referral centers is higher than reported for general population studies. Mathers observed that some 10% of dry eye patients at the University of Iowa Hospitals had Sjögren’s syndrome,46 and Zhang et al47 found 11.3% of clinic-based dry eye patients to have Sjögren’s syndrome. A study of patients with autoimmune thyroid diseases48 found 23% had dry eye (KCS) defined by Schirmer’s 1 test results and rose bengal staining. Twenty-four percent of the autoimmune thyroid disease patients had features of Sjögren’s syndrome; similar features were found in 20% of those with Graves’ disease subgroup in the former, in 27% of those with Hashimoto’s thyroiditis, and in 29% of those with primary myxedema. Dry eye, independent of its subclassification into Sjögren’s syndrome, can be classified on the basis of its severity and frequency. Studies of the prevalence of symptomatic dry eye in particular would suggest that the condition is relatively common but that in most cases it should be described as mild or mild to moderate. The CANDEES3 study found dry eye symptoms were reported by 28.7% in a group aged from 10 to 80 years; of this group 7.8% were classified as having moderate symptoms and 1.6% as having severe symptoms. The severity of the symptoms was more pronounced in the morning than in the evening. This time course of severity differs from that reported by Begley et al,11 who found that dry eye symptoms, particularly in contact lens wearers, were more common in the late afternoon. The Melbourne study16 found that 7.4% of their subjects had two or more symptoms and 5.5% reported severe symptoms, not attributable to hay fever. But this Australian study considered the group aged from 40 to 97 years (with a mean of 59 years), the age of the study population being an important factor affecting severity and prevalence. In another Australian study, Albietz37 found the overall prevalence of dry eye in patients aged 3 to 96 years to be 18.1%, but further subclassification revealed 10.8% in the frank dry eye category and 7.3% in a marginal classification. This study presents some interesting data on the etiological subtypes of dry eye, classifying them into groups of lipid anomaly dry eye (LADE), aqueous tear deficiency (ATD), primary mucin abnormalities, allergic/toxic dry eye (ADE), primary epitheliopathies, and lid surfacing/blinking anomalies (LSADE). On the basis of data from objective assessments of the tear film and lid structure, the prevalence of dry eye subtypes was found to be 4% for LADE, 3.1% for ADE, 1.8% for LSADE, and 1.7% for ATD.37 No cases of primary mucin anomaly dry eye caused by cicatricial disease were diagnosed in this sample. Two cases of primary epitheliopathy were recorded. The very small proportion of ATD dry eye in this study indicates the importance of other aspects of tear physiology in producing dry eye, specifically those related to the lipid and mucin aspects of tears and the resurfacing factors related to the blink. This would suggest that the evaporative category of dry eye2 is significantly more important in the prevalence of dry eye overall than is tear deficiency. Mathers et al,49 in an extensive clinical analysis of 156 consecutive patients presenting to the corneal service of the University of Iowa Hospitals and Clinics, found 70% of the total had some form of MGD. These were broken down into obstructive MGD (17% of the total), obstructive MGD with dry eye (18%), infection (1.3%), rosacea dry eye (5.8%), seborrhea (4.5%), seborrhea/dry eye (14.7%), seborrhea/obstruc-tive MGD (3.8%), and seborrhea/obstructive MGD/dry eye (5.1%). Of the other 47 patients in the series, 10 had allergic disease (6.4%), and 37 (23.7%) dry eye. The dry eye group, although showing no MGD and reduced tear production, also showed an increased evaporative rate (compared with normal participants). This series illustrates the importance of increased evaporation as a cause of dry eye across a range of subclassifications. Bron and Tiffany,50 in a survey of the contribution of meibomian disease to dry eye, have reviewed the prevalence of MGD, evaporative dry eye, and the combination of tear deficiency with evaporative dry eye. The general prevalence of MGD in the population is difficult to assess. Hom et al51 estimated the prevalence in 398 individuals presenting for routine vision examinations as 38.9%, with the prevalence increasing markedly with age (0% at 10, to 67.2% at older than 60 years). Among groups of dry eye patients, the prevalence of MGD is high. Lin et al14 in the study of Chinese in Taiwan with dry eye symptoms found 61.7% had abnormal meibomian features. Horwath-Winter et al52 reported that in a group of symptomatic dry eye patients with reduced tear breakup time, 87% had MGD with or without tear deficiency. In Zhang et al’s47 study of clinic-based dry eye patients, tear deficiency was found in 48.7% and evaporative dry eye in 34%, with 13.9% having a mixed type. Shamazaki et al53 diagnosed MGD in 75% of patients with symptoms of dry eye, but they also found it to be apparent by expressibility of excreta and gland dropout in 61% of patients with no symptoms. The first information on the incidence of dry eye was reported on the 1998 to 2000 follow-up of the BDES.30
Epidemiology of Dry Eye Disease
Key Points
♦ Requirements for Determining Prevalence of Dry Eye
♦ Diagnostic Criteria in Dry Eye Studies
♦ Occurrence of Dry Eye
Prevalence of Dry Eye
Population-Based Studies
Studies of Selected Patient Populations
Prevalence of Subcategories of Dry Eye
Incidence of Dry Eye
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


