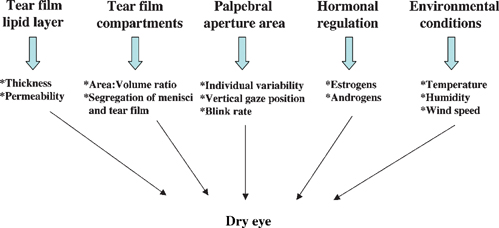
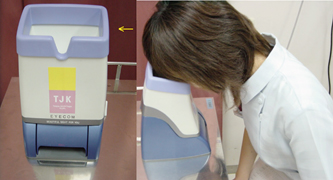
7 Kazuo Tsubota and Murat Dogru The National Eye Institute/Industry Workshop on Clinical Trials in Dry Eyes in 1993 to 1994 sought to provide consensus definitions to assist in clinical trial development and communication and reported a global definition of dry eye as follows: “Dry eye is a disorder of the tear film due to tear deficiency or excessive tear evaporation which causes damage to the interpalpebral ocular surface and is associated with symptoms of ocular discomfort.”1 Tear-deficient dry eye and evaporative dry eye are interdependent disorders. Assuming that there is a critical osmolarity that will result in ocular surface damage, for aqueous tear deficiency, the tear flow rate at which critical osmolarity occurs depends on the evaporation rate for that particular eye; for evaporative dry eye, the rate of evaporation that results in critical osmolarity depends on the tear flow rate. In this sense, some dry eye specialists view most forms of dry eye as evaporative and believe that lid closure may prevent hyperosmolarity, leaving only those pathological changes due to primary or secondary inflammatory events.2 Yet the present definition is of great practical use and will be adhered to in this chapter. Studies by Mathers indicate that the evaporative loss from normal eyes is 14.8 ± 6 × 10-7 g/cm2/second, which is significantly increased to 49.9 ± 6 × 10-7 g/cm2/second in eyes with meibomian gland disease alone and to 59.1 ± 6 × 10-7 g/cm2/second in eyes with combined meibomian gland disease and aqueous deficiency.3 Although there is some evidence that evaporation is affected by lipid film thickness,4,5 the exact influence of lipid thickness and composition is not known. A decrease in phospholipid content of meibomian secretion has been reported in patients with dry eye.6,7 Each of these factors could have a destabilizing effect on the tear film. Craig and Tomlinson have reported a lack of confluence of the tear film lipid layer (TFLL) in mild to moderate dry eye states; the TFLL exhibited zones of thickening (lipid islands) and thinning or absence, in association with an increased rate of evaporative loss.8 Increased evaporation was attributed to a patchy absence of the lipid layer. Several investigators have observed that forced blinking increased the thickness of the TFLL, and, not unexpectedly, so does the deliberate expression of meibomian oil, a procedure that is applied therapeutically in the treatment of meibomian gland dysfunction (MGD).9–11 The size of the palpebral aperture is another important factor related to evaporative water loss from the ocular surface. Tsubota and Nakamori demonstrated, in a Japanese population, that the exposed area of the ocular surface increased by 1.8 and 2.5 times in changing from downgaze to straight ahead and from downgaze to upgaze, and that evaporation per eye increased by 2.5 and 3.4 times, respectively.12 Certain occupations or recreational activities favor a wider aperture and, hence, greater evaporative loss. Thus, it has been suggested that individuals working at video display terminals (VDTs) are at greater risk of dry eye because the eyes are more widely open while looking straight ahead at a computer screen than they would be when they are directed downward toward written work. Tsubota has recommended relocating screens to a lower position to counteract this risk.13 Blink rate has also been suggested as an important factor in ocular surface evaporation. As the blink rate falls, the fraction of time during which the eyes are covered by the blink is reduced.14 Thus, a low blink rate may precipitate dry eye. An extreme example is encountered in patients with extrapyramidal diseases, such as Parkinson’s disease, in which the blink rate may be greatly slowed.15 Blink rate is reduced in persons working at VDTs and in surgeons performing microsurgery.16 The blink rate is increased in patients with dry eye and is normalized by the use of tear substitutes and moisture-conserving spectacles.16 Humidity appears to be another important factor in evaporative ocular surface water loss. Ousler and coworkers employed low ambient humidity and high wind speed as stress factors in their controlled adverse environment studies of dry eye,17 and a high wind speed in conjunction with muscarinic blockade has been used to create a mouse model of dry eye. There is greater risk of ocular surface drying in conditions of very low humidity, such as in desert conditions or during flying at high altitudes.18 Evaporative loss is reduced in conditions of high ambient humidity. Moreover, Korb et al have observed a thickening of the TFLL in subjects wearing goggles that provide a high ambient humidity,19 concluding that the water-carrying capacity of air increases with temperature. Thus, for a given relative humidity (e.g., 30%), there would be a greater carrying capacity and, hence, evaporative loss at a higher temperature. Factors that influence evaporation from the ocular surface are summarized in Figure 7-1. Because the most common basis for increased evaporation from the ocular surface is meibomian gland disease, the diagnosis of evaporative dry eye disease starts with a careful slit lamp examination in an office setting and may include further tests such as tear evaporimetry, TFLL interferometry, meibometry, meibography, quantitative assessment of glandular dropout scores, and meibomian expression. There seems to be a trend to evaluate tear film dynamically in recent studies. The DR-1® tear interference camera (Kowa Co., Nagoya, Japan) for measuring TFLL interferometry can assist in analyzing the changes in the composition, thickness, and structure of the tear lipid layer.20,21 Reflective meniscometry measuring tear meniscus curvature, which is decreased in keratoconjunctivitis sicca (KCS) and correlates well with ocular surface staining, is expected to find wide applications.22 A new Tear Stability Analysis System, which measures tear stability as functions of a serial surface regularity index (SRI) and surface asymmetry index (SAI), topographical indices assessed with 1-second intervals within 10 seconds, has shown a decrease in stability with an increase in SRI and SAI in dry eye associated with Sjögren’s syndrome. The same system has been useful to evaluate the effects of punctal plug treatment in KCS.23 It is our belief that time-wise kinetic evaluations of tear film and ocular surface will become the trend in the near future. The newly developed functional visual acuity meter, which presents Landolt optotypes every second within a defined time frame, has also shown that functional visual acuity continued to decline in Sjögren’s syndrome dry eyes over 30 seconds despite normal acuity values measured by the conventional Landolt chart.24 Patients with KCS with punctal plugs displayed better acuity results compared with untreated patients with this device. We believe that both devices will change our stance and influence not only our understanding of pathogenesis of many dry eye syndromes, including the evaporative variety, but also our assessment of treatment outcomes as well. Figure 7-1 Factors influencing ocular surface water loss. The role of the ophthalmologist should not focus only on the ophthalmological aspects of the disease but should also ensure increased quality of life of the patient. The alleviation of symptoms is the major aim of currently available treatments. The mainstay of current dry eye treatment regimens is the use of artificial tear solutions and punctal plugs.25 In addition, because noninflamed obstructive MGD is a major cause of lipid tear deficiency or evaporative dry eye, an underlying MGD process should be diagnosed and treated efficiently. MGD alters or reduces lipid secretions, leading to increased tear evaporation, decreased tear stability, loss of lubrication, and increased tear osmolarity, which in turn damage the ocular surface epithelium, resulting in dry eye symptoms. The conventional treatment of MGD consists of warm compresses, lid hygiene, topical and systemic antibiotics, topical steroids, artificial tears, and castor oil eye drops as well as systemic androgen therapies.9,25 Details of MGD and its treatment are found in Chapter 6. To increase the quality of life of the patient, management of dry eye problem should include not only clinical treatment of the condition but also consultation about working habits and the work environment.25 Recent years have seen a widespread use of personal computers and portable information terminals and a dramatic increase in the amount of work done using VDTs. It has been estimated that some 28 million U.S. households now have home networks.26 A 2002 survey concluded that 66% of children aged 8 to 17 years in the United States prefer the Internet if they can have only one medium of entertainment.27 There were 150 million registered U.S. America Online (AOL) users as of September 2002, and 12 million of them were reportedly office users of VDTs.28 Prolonged VDT work has been reported to cause general and musculoskeletal symptoms, such as physical fatigue, back or shoulder pain, and pain in the arms and wrists. VDT work has also been reported to cause ocular symptoms, including asthenopia, reduced visual acuity, ocular pain, irritation, corneal epithelial damage, and dry eyes resulting from increased tear evaporation and decreased rate of blinking.13,16,29 The prevalence of eye symptoms among computer users ranges from 25 to 93%.30,31 Surveys by optometrists show that approximately one of six primary care eye examinations given in the United States is due primarily to vision- and eye-related problems caused by computer use. The cost of these eye examinations is nearly 2 billion U.S. dollars, which makes it a significant health care issue and a significant part of most eye care practices.30,32 To alleviate VDT-related symptoms, it is important not only to prevent dry eyes but also to improve the VDT environment. Simple recommendations for VDT-related dry eyes include the following30,33: It has been reported that use of small moistened sponges in special side panels of modified eye glasses (Fig. 7-2) provides steady evaporation from the sponges, increasing the ambient moisture level in front of the eyes. Although Schirmer’s test and cotton thread test results did not improve with these side panels, the rose bengal scores and fluorescein scores improved significantly during the observation period. The most bothersome aspect of treating dry eye is the need to frequently instill artificial tears. In our clinical study, patients reported a reduced frequency of eyedrop use with moist inserts, which simplified their daily therapeutic regimen. Lightweight lenses, silicone pads, correctly sized ear wire, and properly cut side panels were found to be important in increasing compliance with the spectacle side panels.34 Figure 7-2 Spectacle side panels and moist inserts. Periocular warming by an infrared warm compression device or a disposable eyelid warming device for 10 minutes effectively reduced fatigue symptoms and improved tear stability in patients with dry eye and in office workers.6,7 The eye warmer device (Ururun®; Kao, Tokyo, Japan) provided heat through a chemical reaction of the iron powders inside it on exposure to air. The disposable device also provided moisture for about 20 minutes through the presence of a transparent sheet made of synthetic paper that generated moisture with a chemical reaction. This wet-heat warming device was reported to be effective because it provided a constant temperature as well as a constant amount of generated steam with repeatability.35,36 It has also been reported that the temperature of the periocular skin remained at 40°C with the application of the warmer device, which generated steam at a rate of 80 to 200 mg H2O/10 minutes for 20 minutes.7 We believe that these devices increase the tear stability by providing expression of meibomian lipids and decreasing tear evaporation. The steamer device is simply composed of an insulated steel tub with a plastic exterior and a maximum water capacity of 200 mL (Fig. 7-3). A plastic sea goggle-like eyepiece with a rubber cushion at the top is fitted on the main tub body. The steel tub has a thermostat at the bottom that heats the water to provide continuous steam. The water temperature can be controlled by a dial knob on the device. The thermostat automatically goes on and off to keep the water temperature at a constant level of 58.5 ± 0.5°C when the water temperature is set at 60°C. MGD subjects were asked to place their eyes on the goggle piece for 10 minutes and were requested to blink freely during steam application. Ten repeated temperature measurements with 1-minute intervals made from the water and within the eyepiece showed that the temperatures in the water and within the eyepiece were 58.5 ± 0.5°C and 46.3 ± 1.9°C, respectively, and they remained constant throughout 10 minutes. Figure 7-3 shows application of the device in a representative subject. We evaluated the safety and effects of the eye steamer device on tear function and ocular surface of patients with noninflamed obstructive MGD through tear film breakup time, Schirmer’s 1 test, TFLL thickness, vital staining scores, and Visual Analogue Scale scores before and 10 minutes after treatment. We compared the outcome measures with those of healthy control subjects. We showed that the steamer device provided warming of the eyelids, expression of lipids with a concomitant increase of TFLL thickness, increased tear stability, and decreased dry eye symptoms in patients with MGD. We believe that the expression of meibomian lipids by this device helped to stabilize the tear film by decreasing the ocular surface evaporation. The eye steamer device seems to be a safe and promising alternative in the treatment of MGD and may also be useful in VDT-related evaporative dry eyes in addition to the use of warm or hot towel applications.37 Figure 7-3 Eye steamer device, and its application in a patient with dry eye. Traditional therapies for dry eye are palliative, in that their purpose is to replace or conserve the patient’s tears without correcting the underlying disease process. Tear replacement by topical artificial tears and lubricants is currently the most widely used therapy for dry eye, and a variety of components are used to formulate a considerable number of commercially available preparations.38–43 The goal of using tear substitutes is to increase humidity at the ocular surface and to improve lubrication while decreasing evaporation. Most artificial tear preparations contain cellulose ethers, carbomers, polyvinyl alcohol, polyvinyl pyrrolidones, or sodium hyaluronate as their main component.44 Among them, cellulose ethers (e.g., hypromellose, hydroxyethyl cellulose, methylcellulose, carboxymethyl cellulose) are viscoelastic polysaccharides that increase the viscosity of tears not influenced by blinking, have a good retention time on the ocular surface, and mix well with other ophthalmic products. They are sometimes co-formulated with electrolytes as hypotonic solutions and are also available as sustained-release tear inserts. Yet, hypromellose can cause crusting of lids and mimic blepharitis.44 Carbomers are synthetic polymers with high viscosity and a good retention time on the ocular surface but tend to blur vision and may be uncomfortable for patients.44 Polyvinyl alcohols are also synthetic polymers with low viscosity and optimal wetting properties at a concentration of 1.4%. They are water soluble and do not cause visual blur. They are beneficial in aqueous, lipid, and mucin layer deficiencies. However, they have a short ocular surface retention time and do not mix well with other ophthalmic products.44
Increasing Tear Quantity with Artificial Tears and Reducing Evaporation
Key Points
♦ Evaporation from the Ocular Surface
♦ Diagnosis of Evaporative Dry Eye: A Brief Insight
♦ Treatment Modalities Targeting Reduced Ocular Surface Evaporation
Increasing Ocular and Environmental Moisture with Simple Measures
New Insights into the Treatment of VDT Work-Related and Meibomian Gland Disease-Related Evaporative Dry Eye
Spectacle Side Panels and Moist Inserts

Warm Compression Devices and Eye Warmer Sheets
Eye Steamer Device
♦ The Importance and Provision of Tears and Tear Components in the Treatment of Dry Eye Disease
Artificial Tear Solutions and Ocular Surface Effects: Composition of Solutions and Viscosity
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


