|
Note: These structures are connected and regulated by tear fluid, hormones, blood, nerves, cytokines, light, etc.
The tear film, the cornea, the conjunctiva, the lacrimal glands, and the lids work together in a close anatomic and functional relationship as a functional unit to provide an efficient system recognized as the ocular surface (Table 6-1).1–3 The components of the ocular surface are in anatomic continuity and share feedback mechanisms, which result in simultaneous reactions to a single stimulus. Any impairment to any of the components will reflect on the well being of the whole system. For example, a minor epithelial wound to the cornea causes increased tear flow, change in the content of the tear fluid, increased epithelial movement at the corneal surface, increased limbal mitosis, conjunctival capillary dilatation, increased vessel permeability, and changes in blink rate.
Sensory (trigeminal) and motor (facial) nervous connections, as well as cytokines and systemic hormones, guarantee the traffic of information among the different structures, which can either maintain homeostasis or permit fast reactions to new stimuli. Pathogenic events that disturb such a homeostasis and are not promptly neutralized by appropriate reactions of the ocular surface system, will, with time, create a vicious circle of events and will lead to the appearance of disease.1,2
As a part of such a system, the eyelids have important functions toward the protection and homeostasis of the ocular surface and the whole globe, both voluntary and involuntary, including formation, preservation, spreading, distribution, and drainage of the tear film (Fig. 6-1). These activities are dependent on the following factors:
- Proper frequency and completeness of lid movement
- Appropriate appositional tension of the eyelids against the globes
- An exact anatomical orientation of the eyelid margins and lacrimal puncta to the globe, plica semilunaris, and caruncle
- Lipid secretory activity that is quantitatively and qualitatively normal
- Patency of and proper pressure relationships within the lacrimal drainage pathways
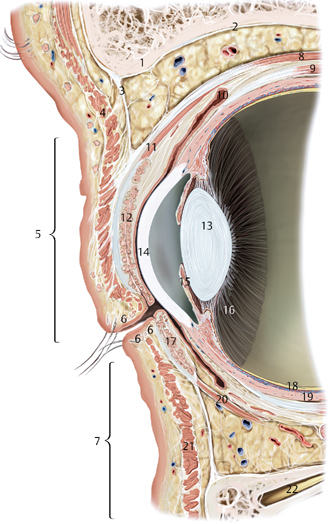
Figure 6-1 Anatomy of the eyelid and forepart of the eye.
1. Periorbital fat
2. Periorbit
3. Orbital septum
4. Orbicularis oculi muscle (orbital part)
5. Upper eyelid
6. Glands of Zeis and Moll’s glands
7. Lower eyelid
8. Levator palpebrae superioris muscle
9. Superior rectus muscle
10. Superior fornix of conjunctiva
11. Superior tarsal muscle
12. Superior tarsus with meibomian glands
13. Lens
14. Cornea
15. Iris
16. Ciliary body
17. Inferior tarsal glands
18. Retina
19. Sclera
20. Inferior tarsus muscle
21. Orbicularis oculi muscle (palpebral part)
22. Intraorbital nerve
♦ Lid Position and Movement
Blinking
The blinking mechanism protects the ocular surface against any external unwanted stimuli and allows the even spreading of the tears contained in the menisci over the surface of the eye, and is the result of the simultaneous contraction and release of two muscles, the levator palpebrae superioris and orbicularis oculi, which act antagonistically. Blinking movements, even if there are negligible variations in speed and amplitude, are paired and symmetrical between the two eyes. Neuronally controlled eyelid blinking, which occurs approximately every 5 to 10 seconds, can be an involuntary spontaneous reflex or a voluntary reflex.
The motion of the eyelids during a blink is a combination of rotation and translation.4 The movement of upper and lower lid is different during a normal complete unforced blink. While the superior lid goes down to meet the inferior, the lower lid moves almost horizontally toward the caruncula, this has a significant impact in tear distribution over the ocular surface and on corneal epithelial cell movement as well as damage distribution leading to clinical signs in ocular surface disease.
The involuntary blink reflex that occurs in response to external stimuli without physical contact can be elicited by fright, sound, and sight, and is mediated by the optic nerve or cranial nerve VIII (auditory) as the afferent arc and primarily by cranial nerve VII (facial) as the efferent arc.
The blinking movement protects the ocular surface from unwanted stimuli and allows the eyelids to spread the fluid contained in the tear meniscus over the ocular surface (Table 6-2). Impairment or loss of this protective reflex mechanism may render the ocular surface more susceptible to pathological insults, as may be seen in neuroparalytic keratopathy (Table 6-3). Through blinking and contraction of eyelid muscles, tears are removed from the ocular surface and drain through the puncta into the nasolacrimal system.
|
|
Blinking occurs between visual fixations and may be timed so as not to interfere with significant visual input,5 and its rate is low when information memory is operating and is suspended during certain cognitive activities, to avoid disruption of display areas accessible to visual input.
Eyelid blinks are important for maintenance of the tear film optical performances.6 Between blinks there is progressive thinning of the tear film, which becomes nonuniform on the ocular surface and may break up. This produces an irregular air-tear interface.7,8 Any local changes in tear film thickness will result in an irregular air-tear interface, leading to the buildup of aberrations into the eye’s most powerful optical system with a reduction in image quality.9 A reduction in the blink rate has been associated with a change in the profile of the corneal tear film and up to a 6% reduction in visual acuity.10 Furthermore, a reduction in blink rate leads to a reduction in contrast visual acuity.11 The lipid layer is essential for the renewal of the aqueous phase of the tear film and the lipid layer itself is influenced by lid motion because of the blink-related secretion of oils from meibomian glands.
If the blinking frequency is reduced, the thickness of the lipid layer is reduced,12,13 and, furthermore, there is a slowdown of the rate of restoration of the thickness of the lipid layer. The longer the period between blinks, the greater the effect on the tear film due to water evaporation. Tear evaporation depends on the width of the palpebral fissure and the surrounding humidity. Evaporation increases with widening of the palpebral aperture leading to a lower tear film stability.14 If tear production cannot keep step with tear water loss, there will be a shift toward hyperosmolarity of the tear film and the progressive development of a symptomatic evaporative dry eye.2 Restoration of the tear film occurs immediately following a blink, so the ability to maintain a regular tear film depends on the blink rate.
The blink rate tends to increase initially to compensate for pathological conditions (such as blepharitis) associated with decreased tear breakup time and resulting from greater tear water evaporation. Incomplete blinking and abnormalities in eyelid congruity have similar effects. Even if most of the involuntary spontaneous blinks are complete, a small number of them are incomplete or are just a twitch (Table 6-4). The latter are particularly frequent during visual activities requiring concentration such as watching the climax of a performance, video display terminal work, or reading.15 The ratio between incomplete and complete blinks increases in ocular surface diseases such as blepharitis or dry eye. This mechanism-intended to reduce the symptoms caused by the friction between the lids and the bulbar surface or the contact between inflamed lid surfaces, in turn affecting tear distribution and clearance—will, in fact, maintain and worsen the diseases.16
| Type | Percentage of blinks |
| Complete | 80 |
| Incomplete | 17 |
| Twitch | 3 |
| Forced | 0 |
Tear Clearance
In addition to their activity in the distribution and even spreading of tear film components, the lids perform a cardinal role in the removal of the tear fluid from the ocular surface. In fact, under normal conditions, aqueous tears are secreted by the lacrimal gland, spread over the entire ocular surface by lid blinking, and then cleared from the eye into the nose through the nasolacrimal drainage system, which includes superior and inferior puncta and canaliculi, the lacrimal sac, and the nasolacrimal duct.17 It has been reported that tear turnover rate is approximately 16%/minute.18 A delayed tear clearance will result in a slowed-down turnover of fresh tears on the ocular surface. This will have an impact on the rate and quantity of the growth and regulatory factors that are constantly secreted onto the ocular surface from the lacrimal gland. At the same time possibly, proinflammatory cytokines and other toxic factors from cell metabolism are not discharged from the ocular surface and will stay longer, delivering their noxious activity.
Several image studies have confirmed that the nasolacrimal drainage system relies on lid blinking to generate a pumping effect to draw tears into the lacrimal sac in rabbits19 and in humans.20 This may explain why lax and floppy lids might contribute to the development of delayed tear clearance. Furthermore, corneal sensitivity progressively decreases with age,21 and that decreased sensitivity is the major cause of decreased blinking.22 Blinking is also known to be the key factor controlling tear clearance,23 especially during the initial rapid phase. Fluorophotometric studies have shown that older people, especially women, tend to lose the initial phase of rapid clearance and have a slower tear clearance rate.24 Several conditions and circumstances can reduce tear clearance rate, including aging (because of the structural changes of lids, lacrimal glands, and tear drainage apparatus); the use of medications that affect tear production (e.g., anticholinergics, retinoids); cosmetic eyelid surgery; corneal surgery that affects corneal nerve conditions and sensation (penetrating keratoplasty, photo reactive keratectomy, laser in situ keratomileusis); prolonged use of video display terminals; and use of contact lenses, which can have an impact on tear fluid spread, blink, and sensation.
A 1997 study suggested that patients with delayed tear clearance frequently had a significantly higher concentration of interleukin 1 in their tears.25 It is possible that once delayed tear clearance has set in, it can perpetuate the development of mucosal inflammation through a vicious circle, especially in susceptible patients. Even simple eye closing during sleep is known to generate inflammation in normal individuals.26
♦ Glands of the Lid and Lipid Secretions
The Glands of the Lids
The opposing margins of the eyelids are about 2 mm in width, are nearly flat, and have a sharp posterior margin that lies against the bulbar conjunctiva and a rounded anterior edge. The portion of the eyelid margin extending from the lacrimal punctum to the lateral canthal angle is, for obvious reasons, termed the eyelash portion of the eyelid. Each ciliary follicle contains two sebaceous glands (glands of Zeis). The sweat glands of the eyelid (Moll’s glands) lie near the cilia, which empty into the adjacent follicles.
The secretions from the tarsal glands form a greasy strip that helps prevent tears from overflowing the lid margin. Immediately posterior to the openings of the tarsal glands and their overlying hydrophobic lipid strip is the mucocutaneous junction. Here the keratinized epithelium of the eyelid skin changes its character to become nonkeratinized, stratified, squamous epithelium. The eyelid margin posterior to the lipid strip is continually wet with tears and is termed the moist portion.
A strip of pretarsal orbicularis oculi muscle, isolated from the remainder of the orbicularis muscle by the eyelash follicles, is known as the ciliary bundle of Riolan or Riolan’s muscle. This strip of muscle deep within the eyelid skin creates an optical effect known as “the gray line” that marks the anterior edge of the tarsus, and constitutes the lipid reservoir of meibomian secretions for the lipid layer of the tear film.
Human meibomian glands are embedded in the tarsal plate in a single line with 20 to 25 and 35 to 40 individual glands present along the respective lower and upper eyelid, and consist of a long, straight, primary duct into which many secondary ducts empty, forming multiple gland lobules. The histological structure of these glands is similar to that of sebaceous glands in other areas, showing bimodal differentiation in the acini with some keratin formation in the ducts. The glands are surrounded by a network of capillaries and fine cholinesterase-positive nerves, which suggest at least a partial neural control of secretion, a condition peculiar to these giant sebaceous glands. In the eyelids of older individuals, the ducts and acini of the glands tend to degenerate and disappear.
Their orifices open onto the skin of the lid margin, just anterior to the mucocutaneous junction. In this way, the meibomian secretion is delivered on the surface of the lid margin to form the marginal reservoirs. Eventually, at each blink, the meibum is spread onto the preocular tear film. The lipids produced by meibomian glands are important in the stabilization of the architecture and in guaranteeing the performance of the tear film; abnormalities in their production have an adverse effect on tear film stability. The mechanisms controlling the quality and the amounts of lipid produced are not completely clarified. The meibomian glands are subject to neuronal, hormonal, and vascular influences.
Innervation is predominantly parasympathetic, but sympathetic and sensory sources are also present. Additionally, vessels surrounding the meibomian glands are richly innervated.27,28 Meibomian gland function may, therefore, be under neuronal control through the direct innervation of the meibomian gland acini or through the indirect regulation of the vasculature to control the synthesis and excretion of meibomian gland lipids. There is also significant evidence of a hormonal androgen control of meibomian gland secretion, as this tissue contains androgen receptor mRNA and androgen receptor protein within acinar epithelial cell nuclei.29 Androgen deficiency is associated with significant and striking alterations in the neutral and polar lipid patterns of human meibomian gland secretion.30 Furthermore, androgen receptor dysfunction in women with complete androgen insensitivity syndrome is associated with meibomian gland changes and a significant increase in the signs and symptoms of dry eye.31
All meibomian glands show positive nuclear staining for antibodies for estrogen receptors. At the moment, no specific correlation has been shown between the quality of meibomian gland secretion and the level of estrogen expression in health and disease. The proportion of cells expressing estrogen receptors increases with age.32 The delivery of meibomian lipid to the lid margin, through the meibomian gland orifice, appears to involve both an active lid-blinking process and a passive excretory component.33,34
In normal subjects, clear oil can be expressed from the meibomian orifices by pressure on the glands through the lids. With age, the proportion of glands from which lipid can be expressed decreases, without apparently decreasing the thickness of the tear film lipid layer.35 Chemical analysis of expressed lipid from normal subjects shows it to consist of a mixture of nonpolar lipids (wax esters, cholesterol, and cholesterol esters), which make up about 60 to 70% of the whole, and of polar lipids, which are mainly phospholipids and glycolipids. There are also small amounts of free fatty acids and alcohols, derived from the ester fractions, and some monoglycerides and diglycerides and neutral fats; part of it might be of sebaceous origin.36
According to McCulley and Shine,37 three types of human meibum exist:
- ♦ Adult 1, with very low levels of cholesterol esters and without unsaturated cholesterol esters and wax ester fatty acids and alcohols.
- ♦ Adult 2, with high levels of cholesterol esters and with unsaturated cholesterol esters and wax ester fatty acids and alcohols, which is the most common and best known type.
- ♦ Infant, characterized by the presence of a thick lipid layer and a long breakup time.38,39
Surface-active compounds such as phospholipids, fatty acids, and free cholesterol along with the proteins and water-soluble constituents of the aqueous phase of the tear film are responsible for the foam that is often seen at the outer canthus. This is produced by the whipping action of the eyelids during blinking.40 A decrease in secretion from the meibomian glands as a result of obstruction or reduced blinking will result in a compromised lipid layer and increased evaporation of the aqueous layer, with subsequent thinning of the tear film.41
The Lipid Layer of the Tear Film
The anterior layer of the tear film is composed chiefly of lipids secreted by the meibomian glands, on two linear reservoirs along the lid margin. It forms and extends across the margin from lid to lid as a continuous sheet. Because meibomian lipid is delivered directly onto the lid skin, it is inevitable that it will mix with skin lipids of sebaceous origin. This is a potential source of contamination both of the lipid that is the source and of the tear film lipid layer.
The hydrophobic lipid at the lid margin is also a barrier to the overspill of tears in the absence of excessive reflex tearing, and it may also prevent maceration of the skin.42 With lid closure, in the down phase of the blink, the lipid of the tear film is compressed onto the lid margin and can flow only forward because surface tension forces prevent it from flowing behind the tarsal plates.43
Forced blinking increases the thickness of the tear film lipid layer.44,45 The deliberate expression of meibomian oil has been proposed as possible treatment for obstructive meibomian gland dysfunction (MGD).12,46 When the ability to express the meibomian gland is reduced by disease, lipid layer thickness is reduced,47,48 and the same happens to the tear film. Benedetto et al49 have shown by fluorometry that forced blinking, which induces a thicker tear film lipid layer, also produces a thicker precorneal tear film than light or incomplete blinking.
The melting rate of meibomian lipid has been estimated to be between 19.5 and 32.9 ± 0.9°C (mean ±SD),50 which is lower than the internal temperature of the lids and of the lid margin, allowing the fluid state of the meibomian lipid within the ducts of the meibomian glands and in the marginal reservoirs when the eyes are open. Changes of external environment temperature could influence the fluidity of lipid in the tear film lipid layer, as well as its distribution and functional performance. The lipid layer of the tear film is composed of two phases: a deeper polar-surfactant phase covered by a nonpolar phase exposed to air.
The polar phase of the lipid layer most likely is only one to three molecules thick and serves as an interface between aqueous tears and the thicker nonpolar lipid layer, to a point that the organization of the nonpolar phase depends on the underlying polar phase for its integrity.51
The polar phase of the lipid layer of the tear film has multiple functions:
- Acting as a surfactant that facilitates the interaction with the aqueous layer of the tear film
- Serving as a barrier against contamination of the nonpolar phase
- Offering structural components for the nonpolar phase
| Class of Lipids | Function in Tears | Consequence of Alteration |
| Polar lipids | Surfactant (helps the spreading of lipids) | Decrease in ability of spreading of the lipid layer and of the underlying water |
| Structure for nonpolar lipids | Submerge into water layer contaminating mucus (?); increase evaporation of water | |
| Nonpolar lipids | Barrier to water and contaminants | Increase in water evaporation and possible direct irritation by free fatty acids |
| Lubricant | Decrease in fluidity and melting of meibomian secretion cause inspissation or orifice plugging |
Adapted with permission from McCulley JP, Shine WE. Meibomian gland function and the tear lipid layer. Ocular Surface 2003;1(3):97–106.
The nonpolar phase of the lipid layer, composed predominantly of wax esters, provides the air-tear film interface and forms the barrier that retards evaporation of water.
Because polar lipids rule the structural organization of the overlying nonpolar lipids, loss or alteration of polar lipids results in uneven spreading of the tear lipid over the ocular surface with loss of efficiency of the nonpolar phase and decreased stability of the lipid tear layer52 (Table 6-5). The lipid layer in a spontaneously blinking normal eye shows a remarkable stability between blinks. The lipid film is compressed in the down phase of the blink; it is formed anew in the up phase, moving rapidly with the movement of the upper lid and then slowing down, to become entirely stationary about 1 second after opening. The mixing of lipid, either within the lipid layer or between the lipid layer and the reservoirs, is limited.53
The chief functions of the tear film lipid layer are to provide a smooth optical surface for the cornea and to retard water evaporation from the ocular surface. A normal lipid layer can prevent up to 90% of possible water evaporation from the tear film.54 Gilbard et al demonstrated directly that meibomian occlusion resulted in a rise of tear osmolarity, possibly as a consequence of an increased evaporation rate of water from the tear film.55 The addition of a tensio-active material able to disrupt the lipid layer to the tear film increases the rate of evaporation,14 the trapping of particulate matter, and, possibly, the antimicrobial function.56
Functions related to the role of the lipid layer in guaranteeing ocular clarity have been discussed previously. Preservation of aqueous fluid results from two related functions of the tear film lipid layer. First, a well-structured lipid layer prevents the overflow from the aqueous-mucin layer, such as might occur during a blink. Second, a well-structured lipid layer decreases evaporation from the ocular surface. Another function may be to aid in preventing harmful entities from injuring the cornea. A well-structured lipid layer can trap small particles such as microorganisms, pollen, or other organic matter; enfolded in the lipid layer, the foreign particle is isolated and harmless. Table 6-6 lists the main recognized functions of lid lipids and of the lipid layer of the tear film.
♦ Classification of Lid Disease
The term blepharitis refers to a family of inflammatory diseases of the eyelids. The condition most typically has a chronic course, with intermittent exacerbations of symptomatic disease. Blepharitis is poorly diagnosed in general clinical practice because the condition goes unnoticed, or, because it does not involve the eye ball, it is considered unimportant until complications or associations bring the symptoms to a level perceived by the patient.47 Patients with blepharitis present with the typical symptoms of eye irritation. In most cases, symptoms are worse in the morning, possibly because almost no tears are produced during the night, so the toxic biological products of bacteria and inflammation are not washed away from the ocular surface.
Blepharitis often is associated with systemic diseases. Dermatological diseases such as rosacea and seborrheic dermatitis, as well as ocular diseases such as dry eye disease, chalazion, trichiasis, conjunctivitis, and keratitis can affect the skin of the lids and induce blepharitis (Table 6-7).
|
|
Infectious Blepharitis
Fungal Lid Infections
Candidal infections of the eyelid are uncommon and are usually associated with candidal infections elsewhere. Usually, the infection occurs in patients who are immunosup-pressed or in those who are taking glucocorticoids or broad-spectrum antibiotics.
Viral Lid Infections
- ♦ Molluscum contagiosum infections are characterized by elevated, round, waxy, pearly white noninflammatory lesions with umbilicated centers.
- ♦ Verruca vulgaris, the common wart, frequently appears on the eyelid, especially on the lid margin. The lesion may be pedunculated or broad-based, and it is often multilobulated.
- ♦ Herpes simplex virus can infect the lid as either a primary or recurrent infection. The infection is characterized by vesicles on an erythematous base that usually progress to ulcers. Regional lymphadenitis usually occurs in the primary infection.
- ♦ Herpes zoster virus involvement of the ophthalmic branch of the fifth cranial nerve often affects the upper eyelid, whereas involvement of the maxillary branch often affects the lower eyelid.
Parasitic Lid Infections
- ♦ Phthirus pubis can infect the lid margin. The organism produces itching and erythema of the lid margin. Nits (egg cases) are characteristically found cemented to the eyelashes.
- ♦ Demodex folliculorum is present in most individuals more than 70 years old and is commonly associated with blepharitis, but its true role in infection is unknown. Diagnosis is confirmed by the presence of “sleeves”—thin, tube-like, transparent processes that extend from the skin over the proximal end of the eyelash for 0.5 to 1 mm. An extensive review could not confirm the existence of a case of Demodex-provoked blepharitis.57,58 Other data suggest a negative influence of Demodex on the risk of producing blepharitis, as gram-positive and gram-negative bacteria seem to grow more often in patients with Demodex. In a way, Demodex seems to be a mediator of chronic blepharitis.59
- ♦ Leishmaniasis, Loa loa, onchocerciasis, and Ascaris lumbricoides can affect the eyelid directly or by producing an immune reaction.
Bacterial Lid Infections
Many cases of blepharitis are caused by bacteria infection, especially staphylococcal infection, which is often associated with seborrhea and meibomian gland disorders.
Allergic Blepharitis
Atopic Dermatitis
Atopic dermatitis presents as a moist, itchy, erythematous eruption of the skin that becomes vesicular and often has crusted areas. It can be acute, subacute, or chronic. The face and lid margins are frequently involved, resulting in a blepharoconjunctivitis and keratitis. Staphylococcus aureus is frequently present as a secondary invader.
Contact Dermatitis
The thin and delicate tissues of the lid are particularly susceptible to a variety of irritants, including cosmetics, topical medications (e.g., neomycin, atropine, epinephrine), plants, and some industrial chemicals. In some cases, a mild follicular conjunctivitis is associated with the eczematoid lid inflammation.
Blepharitis Classified by Location
Based on characteristic clinical features, the location of the predominant inflammatory changes, and the lid structures primarily involved, blepharitis can be grouped into two general categories: anterior lid margin and posterior lid margin blepharitis.60 The gray line (projection of the muscle of Riolan) divides the lid into an anterior lamella (skin and muscle) and a posterior lamella (tarsus and conjunctiva; Fig. 6-2).
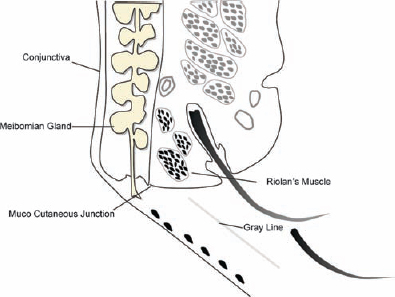
Figure 6-2 The gray line is the projection of Riolan’s muscle on the lid margin and separates the anterior from the posterior palpebral margin.
The eyelash follicles and associated glands of Zeis are part of the anterior lamella, and the meibomian glands are part of the posterior lamella. Because of the contiguity of the different anatomical structures and the functional unit response to any event that is typical of the ocular surface system, this classification, albeit useful in the first-line clinical approach, does not clarify the different pathogenic pathways leading to the lid disease. Knowledge of the pathogenic process can lead to a more accurate therapy. In this perspective, McCulley’s classification of blepharitis could represent a useful aid61:
- ♦ Type I, staphylococcal blepharitis (anterior blepharitis with lid crusting and dry scaling), is marked by acute inflammation of the lids.
- ♦ Type II

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


