Purpose
To describe retinal and choroidal morphology before and after surgery for epiretinal membranes (ERM) in swept-source OCT (SS-OCT). Additionally, to evaluate factors responsible for visibility of the suprachoroidal layer (SCL) and suprachoroidal space (SCS).
Design
Prospective consecutive case series.
Methods
Twenty-nine eyes of 29 patients with symptomatic, idiopathic ERM were included. Pars plana vitrectomy with ERM removal and ILM peeling was performed. We examined patients with SS-OCT twice preoperatively (9–12 months and 1 week before surgery), then postoperatively at 1 week and 6 and 12 months.
Results
Twelve months after surgery visual acuity improved to 20/50 (0.48 logMAR), statistically significantly higher as compared to 1 week preoperatively ( P < .001). Preoperative loss of visual acuity was commonly associated with progression of deformation of the plexiform layers, as central retinal thickness (CRT) did not decrease in this period, nor did photoreceptor defects increase. Choroidal thickness decreased 6 months after surgery ( P = .02) and remained stable until 12 months postoperatively ( P = .2). The outer choroidoscleral boundary was irregular in 16 eyes preoperatively but only in 4 eyes 12 months post surgery. SCS and SCL were visible in 15 eyes.
Conclusion
During the natural course of idiopathic ERM, deformation of the outer plexiform layer progresses and is associated with decreased visual acuity. Eyes with an initially irregular outer choroidoscleral boundary (CSB) recover visual acuity faster after vitrectomy with ILM peeling for ERM. Three factors are independently associated with the visibility of the SCS: disarrangement of plexiform layers, CRT, and multiple adhesion points between retina and ERM.
Earlier studies put under debate the predictive value of various morphologic details of the retina, concluding that retinal thickness and photoreceptor layer and external limiting membrane integrity are the most important in estimating the outcome of idiopathic epiretinal membrane (ERM) surgery. However, a recent meta-analysis revealed that many discrepancies exist between studies, and the need for a good predictive model still remains.
Swept-source optical coherence tomography (SS-OCT) is an emerging technology that offers simultaneous, high-resolution, wide-field cross-sectional imaging of the vitreous, retina, and choroid. In a recent study we confirmed that in addition to the retina being altered in idiopathic ERM, the choroid is affected as well. We confirmed that choroidal thickness was decreased 3 months after surgery. Further study is required to determine whether this decrease is long lasting.
Furthermore, we have previously noted several novel findings of the choroidal architecture, such as the suprachoroidal layer, suprachoroidal space, and possible irregularities of the outer choroidoscleral boundary. The meaning of those findings was not exclusively studied.
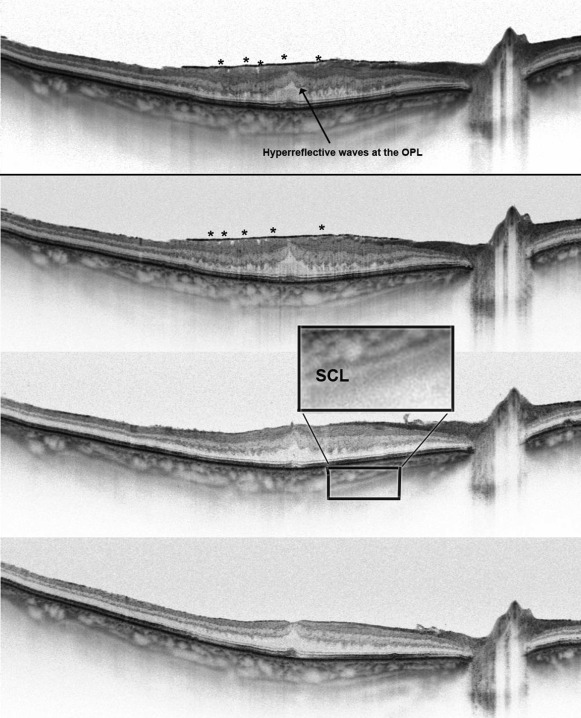
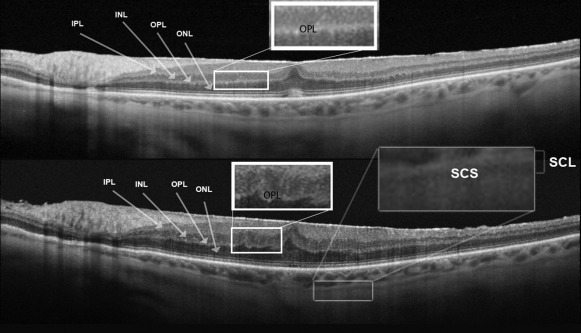
The suprachoroidal layer consists of 2 bands, an inner hyperreflective band and an outer hyporeflective band. The hyporeflective band of the suprachoroidal layer probably corresponds to the suprachoroidal space. The suprachoroidal space was historically observed only in choroidal effusion syndrome. Recently, this layer was reported to be more frequently visible in hyperopia. Furthermore, we confirmed this layer to be more often visible in macular diseases than in healthy eyes. In patients with ERM without metamorphopsia and without vision complaints, we did not observe this layer in any of 20 analyzed eyes. In another retrospective study of 21 eyes, examined before and after vitrectomy for idiopathic ERM, we noted this layer in 4 cases. It was also observed that the suprachoroidal layer is even more often visible in eyes with full-thickness macular holes and vitreomacular traction syndrome. The factors correlating with the visibility of this layer are still not precisely understood. With the advent of new technology enabling the introduction of drugs to the suprachoroidal space, the role of the visualization of the suprachoroidal layer and suprachoroidal space might be of growing importance.
The aim of this prospective analysis was to estimate if there are factors associated with the visibility of the suprachoroidal layer in eyes with idiopathic ERM. An additional intention was to describe long-term changes in retinal and choroidal morphology, visualized with SS-OCT, before and after vitrectomy for idiopathic ERM.
Methods
This is a prospective consecutive case series of 29 eyes of 29 patients with idiopathic ERM operated on in the year 2013. Inclusion criteria were symptomatic ERM scheduled for vitrectomy. We excluded eyes with membranes secondary to other pathologies such as retinal detachment, laser photocoagulation, or vascular occlusion and also excluded patients with previous ocular surgery (except noncomplicated cataract surgery performed at least 12 months before the beginning of the study), coexisting age-related macular degeneration, nicotine abuse, diabetes or glaucoma, or refractive errors above +2 and −2 diopter before cataract surgery. We obtained institutional ethics committee approval (Centrum Zdrowia Matki Polki, Lodz, Poland) and the study complied with the Declaration of Helsinki. Patients were recruited from the authors’ outpatient department and signed informed consent for the study.
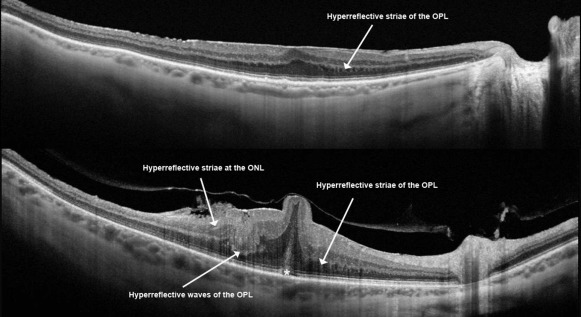
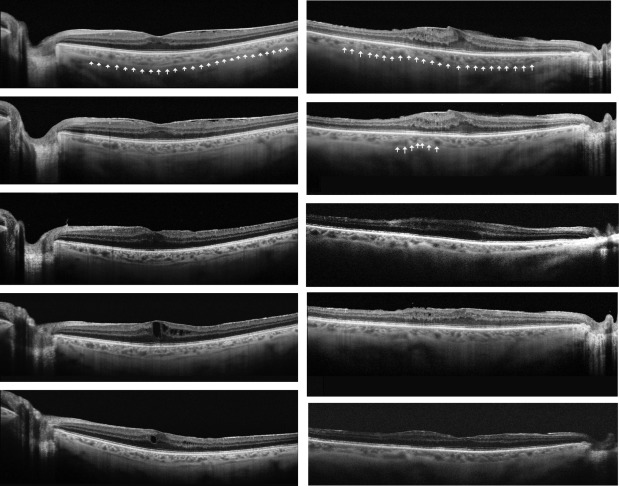
In all eyes we measured visual acuity (Snellen charts) and performed a complete ophthalmic examination and SS-OCT (DRI-Atlantis; Topcon, Tokyo, Japan) before surgery at 2 time points: 9–12 months and then 1 week before vitrectomy. Postoperatively, we examined the patients at 1 week, 6 months, and 12 months. In all cases, we performed a single line scan with a resolution of 1 μm, with a length of 12 mm. Focal central retinal thickness was measured manually as the distance between the internal limiting membrane (ILM) and the retinal pigment epithelium (RPE). Choroidal thickness was measured manually (by Z.M. and K.O.) on black-and-white images, between the line representing RPE (the outermost hyperreflective retinal layer) and the outer choroidoscleral boundary (the outer hyperreflective line of the choroid). The software included a caliper for manual measurements. Irregularities of the inner and outer plexiform layers were categorized as either striae or waves ( Figure 1 ). The extent of adhesion between ERM and the retina surface was categorized as either multiple adhesion points to the retina ( Figure 1 , Top; Figure 2 , Center top) or flat adhesion without evident anteroposterior traction ( Figure 1 , Top; Figure 3 ). Additionally, we analyzed the visibility of the suprachoroidal space ( Figure 3 ), the regularity of the outer choroidoscleral boundary (regular vs irregular) ( Figure 4 ), defects of the photoreceptors, and central hyperreflective deposits ( Figure 1 , Bottom, star). We used SigmaStat (Systat, London, UK) for Windows for statistical analysis.
Surgical Technique
Two experienced surgeons (J.N., Z.M.) performed all of the operations. Core vitrectomy and Membrane Blue Dual staining (0.06% solution for 1 minute) were performed. ERM peeling within the vascular arcades followed. Then, a second staining was performed for 30 seconds. If present, the second layer of the ERM was then peeled. If not, the ILM was grasped with ILM forceps and peeled off in a circular fashion ( Supplemental Video , available at AJO.com ). In cases of multilayered membranes, staining was applied until the ILM was peeled. No tamponade was used in any case.
Results
Twenty-nine patients (23 women and 6 men) with a mean age of 68 years were included in this study. Four eyes were pseudophakic at the beginning of the study; 6 other eyes had phacoemulsification performed in the first year after vitrectomy.
Visual Acuity
There was a statistically significant decrease in the preoperative mean visual acuity from 20/63 to 20/160 (from 0.48 to 0.85 logMAR; 0.42 to 0.17 Snellen) ( P < .001) during the 9–12 months preoperative observation period. There was a statistically significant improvement in the mean visual acuity from 20/125 (0.81 logMAR; 0.2 Snellen) 1 week after surgery ( P = .69) to 20/63 (0.54 logMAR; 0.34 Snellen) 6 months later ( P < .001) and 20/63 (0.48 logMAR; 0.41 Snellen) 12 months after vitrectomy ( P < .001). Final visual acuity improved statistically significantly from the values obtained 1 week before surgery ( P = .01) but did not differ from visual acuity noted 9–12 months preoperatively. There was a trend toward correlation between final visual acuity and central retinal thickness 1 week preoperatively, but without strong statistical significance ( P = .08) ( Table 1 ).
| Visual Acuity (Snellen) | Visual Acuity (logMAR) | Central Retinal Thickness (μm) | Maximum Retinal Thickness (μm) | Central Choroidal Thickness (μm) | |
|---|---|---|---|---|---|
| 6–12 months before surgery | 0.42 | 0.48 | 475 | 514 | 221 |
| 1 week before surgery | 0.17 | 0.85 | 468 | 522 | 222.7 |
| 1 week after surgery | 0.2 | 0.81 | 434 | 502 | 218 |
| 6 months after surgery | 0.34 | 0.54 | 376 | 442 | 213 |
| 12 months after surgery | 0.41 | 0.48 | 357 | 430 | 209 |
Details of Retinal Morphology
Central retinal thickness did not increase significantly during the preoperative period ( P = .4) ( Table 1 ). Photoreceptor defects did not increase in any case, nor did the hyperreflective deposits under the fovea. We observed that the outer plexiform layer and the inner plexiform layer are disarranged in all cases of ERM. However, the stage of disarrangement differed. At the initial visit we observed irregularities in the form of hyperreflective striae at the outer border of the inner plexiform layer and outer plexiform layer in all eyes ( Figure 1 , Top). In 12 eyes, besides visible striae, at some spots the disarrangement was more pronounced and wavy. Thus we called this form of disarrangement “hyperreflective waves” at the outer border of the inner plexiform layer and outer plexiform layer ( Figure 1 , Bottom). Central retinal thickness and mean retinal thickness were significantly higher ( P = .007, P = .008, respectively), and visual acuity was significantly lower ( P = .049), in eyes with these hyperreflective waves when compared to eyes with hyperreflective striae only. The irregularities of the plexiform layers always increased during the preoperative follow-up and 7 eyes with hyperreflective striae developed additional hyperreflective waves ( Figure 3 , upper box) developed additional hyperreflective waves ( Figure 3 , lower left box). After surgery, those irregularities of the outer plexiform layer tended to normalize, and the outer plexiform layer was completely regular in most eyes 12 months after surgery ( Table 2 ).
| 6–12 Months Before Surgery | 1 Week Before Surgery | 1 Week After Surgery | 6 Months After Surgery | 12 Months After Surgery | |
|---|---|---|---|---|---|
| Hyperreflective striae at the outer plexiform layer | 100% | 100% | 100% | 16/29 | 0/29 |
| Hyperreflective dots in outer nuclear layer | 0 | 0 | 8/29 | 6/29 | 3/29 |
| Wavy outer plexiform layer | 12/29 | 19/29 | 0/29 | 0/29 | 0/29 |
| Visible suprachoroidal layer and space | 15/29 | 15/29 | 15/29 | 15/29 | 15/29 |
| Irregular choroidoscleral boundary | 8/29 | 16/29 | 16/29 | 10/29 | 4/29 |
| Multiple adhesion points between retina and epiretinal membrane | 11/29 | 11/29 | – | – | – |
| Photoreceptor defects | 3/29 | 3/29 | 3/29 | 2/29 | 2/29 |
| Highly reflective foveal region | 4/29 | 4/29 | – | – | – |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


