Surgical management of posterior uveitis
Sunir J. Garg MD
Vitrectomy is important for the care of patients with uveitis.1 It plays a role in the treatment of complications that result from uveitis, including epiretinal membranes, cystoid macular edema, and retinal detachments. Vitrectomy also plays an important role in both the diagnosis and treatment of uveitis.
Diagnostic vitrectomy
Most cases of uveitis are still diagnosed through a combination of the patient’s medical and ocular history, demographic information, and clinical appearance. Serologies and other tests such as X-rays and CT scans can also help establish a diagnosis. Nonetheless, despite advances in the diagnosis of some causes of uveitis, approximately 30% to 50% of anterior uveitis, 35% of intermediate uveitis, and 25% to 50% of posterior uveitis have no identifiable underlying cause.2,3,4
Vitrectomy can be useful in the diagnosis of suspected infectious uveitis, including Whipple’s disease, suspected primary intraocular lymphoma (PIOL), and in cases in which a cataract or vitreitis precludes the ability to assess the retina clinically (Table 1).5,6,7
TABLE 1. Reasons for Diagnostic Vitrectomy | |
|---|---|
|
That said, even when an infection is suspected there is often no reason to perform a vitrectomy. In immunocompetent individuals, toxoplasmosis is the most common infectious cause of posterior uveitis.8 Because the clinical presentation usually is so characteristic, there is no diagnostic dilemma. In immunocompromised individuals, cytomegalovirus retinitis has been the most common cause of posterior uveitis.9 Again, the clinical appearance usually makes the diagnosis pretty straightforward.
Other members of the herpes family, including varicella zoster and herpes simplex, cause posterior uveitis (acute retinal necrosis and progressive outer retinal necrosis) often with characteristic appearances, but if needed, these organisms can be reliably diagnosed with a vitreous specimen and polymerase chain reaction (PCR).10 Bacteria, fungi, and parasites (Toxoplasma gondii and Toxocara) also can be reliably diagnosed with a vitreous specimen.11
If patients are worsening clinically despite aggressive therapy, consider obtaining a vitreous specimen, either through a vitreous tap or by vitrectomy. Additionally, if intraocular lymphoma or Whipple’s disease is considered, a vitreous specimen is critical to establish the diagnosis.
Methods of obtaining a vitreous sample
Informed consent is important for all patients. The patient should be told of possible risks including cataract formation, occurrence of a retinal tear or detachment, and development of infectious endophthalmitis. They should also be counseled that even after vitrectomy, no definitive diagnosis may be found.
Periocular, retrobulbar, or general anesthesia can be used. It can be difficult to achieve good anesthesia in inflamed eyes, especially those with acute infectious endophthalmitis. In such cases, consider heavily sedating the patient as the eye is initially entered.
For some cases of uveitis, an in-office diagnostic tap is all that is needed (postcataract infectious endophthalmitis is a good example).12 A 5/8″ 25-gauge needle on a syringe (I prefer a 3 cc syringe, others use a TB syringe) can be used to enter the eye 4 mm from the limbus in phakic eyes, and 3.5 mm from the limbus in pseudophakic/aphakic eyes. Usually no more than 0.5 cc can be obtained, but for many routine studies, this is plenty. PCR requires only a small specimen (approximately 100 μL). There is a handheld vitrectomy device (Visitrec, Becton Dickson Ophthalmics) that can be used to obtain a larger specimen that a vitreous tap. This system has the advantage of being readily accessible and can be performed in the office.13 I generally prefer a three-port pars plana vitrectomy rather than this device because intraoperative control of the eye is better, a more complete vitrectomy is possible, additional therapeutic interventions can be performed, and visualization is much better.
For cases that are taken to the operating room, the 20-, 23-, or 25-gauge systems can be used. For most cases, the smaller gauges are great, especially for cases of endophthalmitis.14 I prefer 20-gauge vitrectomy if extensive membrane dissection is required. If the vitrectomy is being performed for cytology, consider reducing the vitrectomy cut rate to approximately 100 CPM, especially when using the 23- or 25-gauge systems as the smaller gauge systems can chop up the cells (J. William Harbour, MD, personal communication).
Once in a while, the area of interest is deeper than the vitreous. In order to diagnose certain conditions (suspected sub-RPE lymphoma, retinal granulomata) and infections (atypical herpetic retinitis, toxoplasmosis, fungi), a retinal, subretinal, or choroidal biopsy may be needed. The technique for this has been well described elsewhere.15,16,17,18,19 It is important to obtain a specimen that encompasses both healthy and involved tissue, especially for noninfectious cases, because this greatly increases the diagnostic yield. Usually a sufficient specimen should be obtained to send a portion to microbiology, part for immunopathology, and another portion placed in formalin/glutaraldehyde for histopathology. Both patient and surgeon should be prepared for intra- and postoperative hemorrhage, as well as the need for silicone oil as an intraocular tamponade. There is also an increased incidence of proliferative vitreoretinopathy (PVR).
Even though one sends the specimen for testing postoperatively, most of the tests and the types of specimens that are needed for these tests should be decided upon preoperatively. It can be very helpful to talk to the pathologist, microbiologist, and immunochemist before going to the operating room. For most cases, the initial portion of the vitrectomy should be performed with the infusion turned off and the vitrector connected to a syringe. This undiluted specimen can then be aliquoted for microbiology, PCR, cytology and flow cytometry as needed. I have had good luck increasing the amount of undiluted vitrectomy specimen by infusing air rather than balanced salt solution (BSS). After this, you can turn on the BSS infusion and send the cassette to the appropriate places.
Diagnostic tools
Cytology
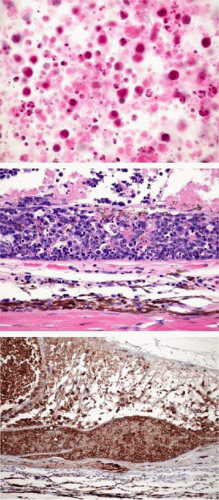
Before going to the operating room, the surgeon should communicate with the respective laboratories to ensure that the lab receives a useful amount of the appropriate specimen in the appropriate manner and in time. This is especially true for cytology. Primary intraocular lymphoma is hard to diagnose on a good day, and few things will burst the surgeon’s bubble faster than the pathologist saying that the specimen is inadequate (when the pathologist really wants to says that the surgeon is inadequate). These specimens, especially suspected cases of intraocular lymphoma, need immediate attention; otherwise, the cells start to degrade. Depending on how long it takes to get the specimen to the lab, it may be worthwhile putting the specimen into a tissue culture medium such as RPMI-1640S or formaldehyde (talk to the cytopathologist first). Having the specimen read by an experienced cytopathologist is still the gold standard to establish the diagnosis of intraocular lymphoma, but the sensitivity leaves room for improvement.20 (Figs. 1A-C). Because of this, additional testing for IgH,21 cytokine levels,22 flow cytometry,23 and T-cell alterations24 may increase the diagnostic yield.25
Intraocular antibody analysis
One can assume that in many cases, exposure to an antigen will lead to local antibody production. Analysis of this antibody production as a measure of the host response to an antigen is performed by the Witmer coefficient method, which calculates the quotient between the local and serum IgG (or IgA) levels using radioimmunoassays or enzyme-linked immunosorbent assays (ELISA)26. This technique is helpful in the diagnosis of toxoplasmosis,27,28 toxocariasis,29 and acute retinal necrosis, but is not useful in the diagnosis of cytomegalovirus.10
Cytokine analysis
Malignant B cells in patients with primary intraocular lymphoma express increased levels of interleukin-10 (IL-10). In other cases of noninfectious posterior uveitis, there are higher levels of IL-6. If there is a high IL-10:IL-6 ratio, this suggests lymphoma.22 In one study, an elevated IL-10 to IL-6 ratio was found in 24 of 31 patients with intraocular lymphoma.21
Culture
Microbial cultures are still the gold standard of diagnosing intraocular infections. The laboratory should be told what to look for as certain organisms (e.g., fungi) may take a long time to grow out, and others, such as Propionibacterium acnes, may need to be cultured in a specific manner. It can be hard to culture viruses so polymerase chain reaction (PCR) is usually a better test.
PCR
PCR plays an increasingly important role in the diagnosis of uveitis.10,30,31,32,33 The main strengths of PCR are the rapidity and specificity of the result; however, in order to utilize these strengths, the clinician still has to tell the lab which tests are most needed. Some of the newer PCR machines can run a large battery of tests on a small specimen, but these instruments are not yet in widespread clinical use. Many times, an anterior chamber tap provides an adequate specimen. PCR has successfully been used to diagnosis viral, fungal and bacterial infections, including Tropheryma whippelii and P. acnes34 (Table 2). Depending on the lab, the rapidity of diagnosis can meet or exceed that of culture or gram stain. While PCR is very specific, it can give both false positive and false negative results.35,36 False-positive results occur with the herpes viruses. One study found seven patients were PCR positive for varicella zoster although clinically they did not have zoster-related disease.37 It remains unclear whether these specimens were contaminated from the tear film or elsewhere, or if varicella is actually present, but clinically silent, in the anterior chamber in a number of individuals. A more recent study suggests that using PCR in combination with another test, such as the Witmer coefficient, may increase the diagnostic yield.38
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree