Surgical Management of Neovascular and Exudative Macular Choroidal Disease
Allen Chiang
Matthew A. Thomas
James F. Vander
Exudative maculopathy arising from choroidal neovascularization (CNV) secondary to exudative age-related macular degeneration (AMD) is a leading cause of decreased vision. Other disease processes that attenuate or compromise the Bruch membrane may also lead to CNV, including but not limited to presumed ocular histoplasmosis syndrome (POHS), high myopia, angioid streaks, multifocal choroiditis, and trauma. The advent and efficacy of anti–vascular endothelial growth factor (VEGF) therapy has brought about a transformative shift in the management of exudative maculopathy, with surgical intervention for subfoveal CNV now used on a rare and highly select basis. However, the management of submacular hemorrhage in particular still represents a therapeutic challenge for which surgical intervention remains a relevant treatment option.
CHOROIDAL NEOVASCULARIZATION (CNV)
In the normal eye, the Bruch membrane serves as a physiologic barrier between choroidal vessels and the overlying retinal pigment epithelium (RPE) and neurosensory retina.1 Various ocular disorders can weaken this barrier, allowing the ingrowth of capillary tufts and the subsequent proliferation of fibrovascular tissue beneath the retina. In age-related macular degeneration (ARMD), the Bruch membrane demonstrates diffuse abnormalities, with the accumulation of drusen that predispose to vascular invasion.2 In the presumed ocular histoplasmosis syndrome (POHS) and other inflammatory conditions, the defects in the Bruch membrane are usually more discrete and focal.2 Other disease states such as myopic degeneration and angioid streaks may also have extensive defects or cracks in the Bruch membrane. Regardless of the underlying cause, visual function usually declines when exudative fluid, hemorrhage, or the neovascular tissue itself extends beneath the central macula, disrupting its normal anatomy and interfering with the normal metabolic interactions between the photoreceptors and the RPE.
In the past, the mainstay of management of choroidal neovascularization was ablation of the tissue with light energy via laser or xenon photocoagulation. A series of randomized, controlled, prospective studies conducted by the Macular Photocoagulation Study (MPS) group established the superiority of laser photocoagulation over observation in various settings.3,4,5,6,7,8,9,10 However, such conventional laser treatment induced vision loss, especially following application to subfoveal membranes. Even with juxtafoveal CNV, decreased vision at times would result from the spread of laser scars into the subfoveal region. This prompted evaluation of alternative means of eradicating subfoveal neovascularization.6,7
THE SURGICAL APPROACH TO TREATMENT OF SUBFOVEAL CNV
Macular Translocation
Macular translocation is a procedure in which the neurosensory retina overlying the fovea is shifted away from subfoveal CNV to another area with healthy RPE. Various methods of macular translocation have been described, encompassing two broad categories. One category involved large retinal incisions with further variation based on whether or not a 360° retinotomy was performed. The other major category (limited macular translocation) involved only punctate retinotomies with variations based on the absence or presence of chorioscleral shortening, either infolding (imbrication) or outfolding (outpouching).11
Regardless of the method, the goal of this relocation was the same: preserve photoreceptor function and displace CNV to an extrafoveal location for photocoagulation. Historically, most patients who underwent macular translocation were those with exudative AMD. The size and location of the underlying CNV determined the particular method that would most effectively achieve the necessary degree of translocation. The details of each method and their variable results have been reported in detail elsewhere.12,13,14,15,16,17,18
Although macular translocation was considered to be an innovative approach to the treatment dilemma posed by subfoveal disease, it is mentioned here mainly for historical reasons. Variable results from concurrent investigation of the direct excision of CNV in the Submacular Surgery Trials (SST) along with the emergence of treatment modalities such as photodynamic therapy (PDT) and anti-VEGF therapy impeded its widespread adoption.
Submacular Excision Of CNV
In 1988, De Juan and Machemer19 described vitrectomy techniques for the removal of blood and disciform scars in four patients with end-stage ARMD. Vision improved in three, but the best acuity achieved was only 5/200 (1.5/60), and two patients developed severe proliferative vitreoretinopathy. Given these results, the technique of creating a large flap retinotomy to expose the macular scar was not widely used. Blinder et al20 also performed large flap retinotomies on eyes with extensive subfoveal neovascular scars with similarly discouraging results. They speculated that the postsurgical absence of the RPE was the cause of poor vision, and thus they attempted to replace subfoveal RPE with an autologous pedicle graft or with an homologous patch of RPE removed from a blind donor eye undergoing enucleation.21 Yet the surgical transplantation of RPE failed to improve vision.
In spite of this, encouraging results from the implementation of a novel small retinotomy technique initially reported by Thomas and Kaplan22 for POHS cases prompted further refinement of the surgical instrumentation and technique, thereby expanding its application to a wider assortment of cases.22,23 This approach used a small retinal hole through which instruments were introduced into the subretinal space in an effort to dislodge, grasp, and extract the CNV (Figs. 62-1,62-2,62-3,62-4,62-5).
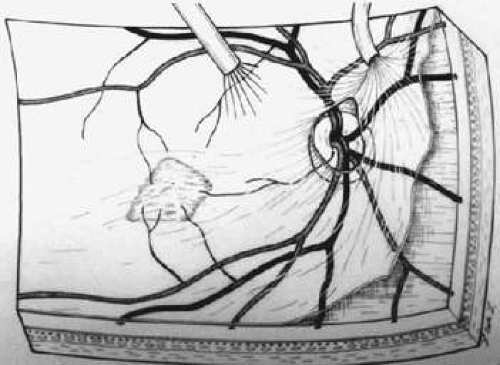 Figure 62-1. The posterior hyaloid is engaged with a silicone tip extrusion cannula near the optic disc. |
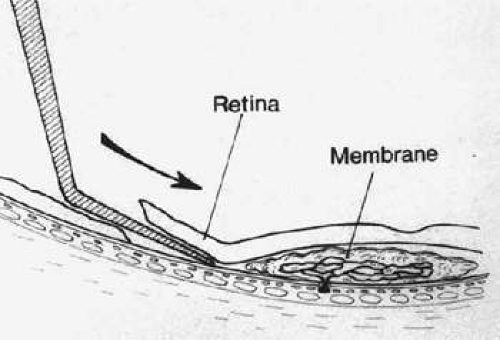 Figure 62-2. A small retinotomy is created with a 120°-angled, sharp, 36-gauge pick. Arrow indicates direction of pick motion tangential to underlying RPE. |
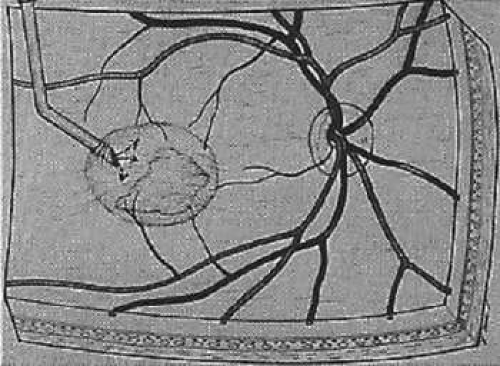 Figure 62-3. The neurosensory retina is gently elevated by the slow infusion of balanced salt solution through a 120°-angled, bevel-up 33-gauge subretinal cannula (arrow). |
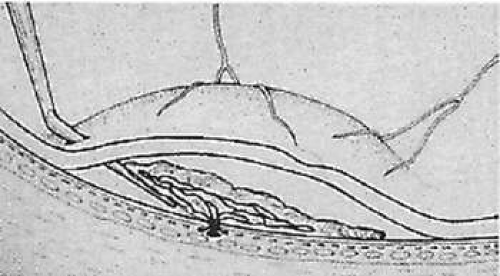 Figure 62-4. The neovascular complex is elevated from the underlying retinal pigment epithelium with the sharp, 36-gauge subretinal pick. |
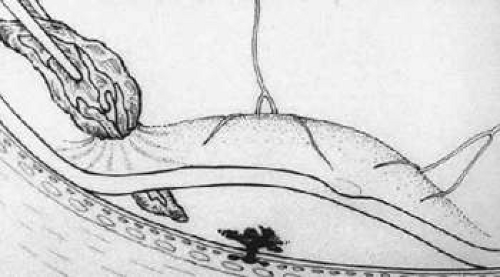 Figure 62-5. The neovascular complex is grasped with horizontal subretinal forceps and removed from the eye. |
Following several reports on the surgical excision of subfoveal membranes in AMD, it became clear that unless subfoveal RPE is preserved postoperatively, central acuity is not regained.24,25,26,27,28 Therefore, the intraoperative preservation of foveal RPE and choriocapillaris became the single most important predictor of good visual outcome. The focus of surgical excision of CNV then shifted from refining operative techniques to improving preoperative case selection. Eyes with neovascular complexes that existed anterior to the native RPE, involved focal defects in the Bruch membrane, or possessed ingrowth sites eccentric to the center of the fovea tended to have better visual outcomes.29
In 1994, Gass30 proposed classifying CNV into type 1 and type 2 membranes to aid preoperative selection. Type 1 CNV is seen in AMD and occurs underneath or enmeshed with RPE. The defects in the Bruch membrane and areas of attenuated RPE are diffuse. Removal of CNV of this type is, therefore, more difficult and often results in damage to the supportive cellular structures underlying the fovea. This damage is associated with poor visual recovery.31 Type 2 CNV grows from a small focal defect in the Bruch membrane. Because this type of CNV grows over the RPE, it may be possible to remove it with little collateral damage to RPE. Conditions such as POHS, high myopia, and idiopathic etiology predispose individuals to this type of CNV.31 The value of extracting subfoveal CNV, regardless of type, was investigated prospectively in the pivotal SST.
The Submacular Surgery Trials (SST)
The SST was a collection of multicenter randomized trials designed to formally evaluate the effectiveness and safety of the surgical excision of subfoveal CNV. It began when the only established modality of treatment was conventional laser photocoagulation. Indeed, in the first SST Pilot Study report, surgery was compared to laser photocoagulation for recurrent CNV in AMD. Fewer surgery eyes (50%) than laser-treated eyes (65%) had better visual acuity or no more than one line worse compared to baseline.32
Surgery for subfoveal CNV in AMD was examined in the SST Group N trial, which revealed no significant difference in outcomes between eyes randomized to surgery compared to observation.33
Similarly, the SST Group B trial investigated surgery for hemorrhagic CNV lesions due to AMD but failed to demonstrate any benefit to surgery over observation in terms of stability or improved visual acuity. Loss of eight or more letters of vision occurred in 56% of surgery eyes versus 59% of observation eyes at 24 months. Although fewer eyes in the surgery arm of the study experienced severe vision loss (defined as more than six lines), this was not a primary study outcome of interest.34
In contrast, the SST Group H trial indicated that surgery should be considered in eyes with subfoveal CNV associated with POHS or idiopathic subfoveal CNV. With 2 years of follow-up, 55% of surgery eyes versus 46% of eyes in the observation arm had vision better than or within one line of baseline visual acuity. In a subgroup of eyes with vision worse than 20/100 at baseline, 76% of surgery eyes compared to 50% of observation eyes had visual acuity better than or within one line of baseline vision, a slightly larger benefit.35 Consequently, the SST Group H results suggest that patients with CNV associated with POHS or idiopathic membranes may derive some benefit from surgical removal of CNV.
Aside from the SST Group H findings, submacular surgery as performed in the SST did not increase the chance of stable or improved visual acuity. In light of newer, more effective treatments for exudative maculopathies such as AMD, submacular surgery for the removal of CNV is now rarely used.
SUBMACULAR SURGERY IN THE ERA OF PHOTODYNAMIC THERAPY (PDT)
PDT induces rapid occlusion of a choroidal neovascular membrane within 24 hours, with subsequent closure of adjacent choroidal vessels over the course of a week.36 The results of the Treatment of Age-related Macular Degeneration with Photodynamic Therapy (TAP) study group trial and the Verteporfin in Photodynamic Therapy (VIP) study found photodynamic therapy (PDT) to be an effective treatment for predominantly classic or purely occult subfoveal CNV in AMD, respectively, although visual improvement remained rare.37,38 Thus PDT provided ophthalmologists with a new, less invasive alternative to treatment with submacular surgery. By extrapolation, PDT later became accepted as a treatment option for CNV arising from POHS or idiopathic causes as well, given that such CNV is often predominately classic.
Nevertheless, it was observed that in select cases submacular surgery could be complementary to PDT and thus retain some relevance. For instance, if the foveal RPE and choriocapillaris remain intact following successful PDT, using submacular surgery to remove inactive fibrovascular tissue may be beneficial and result in an improvement in visual acuity. Furthermore, in nonresponders to PDT or those with low preoperative visual acuity due to fibrotic membranes, submacular surgery still seemed to be appropriate based on a meta-analysis.39
SUBMACULAR SURGERY IN THE ERA OF ANTI-VEGF THERAPY
The advent of anti-VEGF therapy created a dramatic shift in the management of CNV in exudative maculopathies such as AMD. Unlike conventional laser, PDT, or surgery, VEGF inhibition has introduced a modality with which to treat CNV without inducing tissue disruption or destruction. In distinction to PDT, which rarely improved vision in patients with subfoveal CNV due to AMD, anti-VEGF therapy has demonstrated its effectiveness not only to stabilize visual acuity but, for the first time, to achieve unprecedented improvements in vision in a considerable proportion of cases.
Ranibizumab, a recombinant, humanized, monoclonal antibody Fab (fragment antigen binding) that binds to the VEGF-A isoform, is FDA approved for the treatment of exudative AMD. Two landmark, multicenter, double-blind, randomized, controlled clinical trials, the ANCHOR and MARINA studies, investigated the efficacy of ranibizumab for the treatment of predominantly classic CNV and minimally classic or occult with no classic CNV, respectively, associated with AMD over 2 years.40,41 In ANCHOR, ranibizumab proved superior to PDT with verteporfin, with greater than 94% of ranibizumab patients having fewer than 15 letters lost compared with 64% of patients given PDT. Although only 5.6% of PDT patients experienced an improvement in vision of more than 15 letters, 35.7% of patients given 0.3 mg ranibizumab and 40.3% of those in the 0.5 mg ranibizumab group achieved this study end point. The results were quite similar in MARINA, in which ranibizumab was compared to sham injection. In both studies, ranibizumab demonstrated an acceptable safety profile.
Preceding ranibizumab was bevacizumab, a full-length monoclonal antibody against VEGF-A and a common off-label treatment for CNV secondary to AMD or other etiologies, in addition to various VEGF-mediated ocular disorders.42,43,44,45 Although data from prospective randomized clinical trials are lacking, multiple case series have not identified any significant ocular or systemic morbidity with its use. Intravitreal injection of an anti-VEGF agent carries the low risks of hemorrhage, cataract, retinal detachment, and noninfectious or infectious intraocular inflammation, which is a particularly concerning complication.46,47,48 However, the reported rate of infectious endophthalmitis following intravitreal injection of bevacizumab is very low, approximately 0.1% per injection.49
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


