Surgical Management of Corneal Opacities in Children
Robert C. Arffa
Gerald W. Zaidman
To achieve optimal visual rehabilitation, corneal surgical procedures require good patient cooperation in the postoperative period. Because such cooperation cannot be obtained from young children, many corneal surgeons are reluctant to operate on them. The administration of drops, detection and removal of loose sutures, and recognition of graft rejection are more difficult in children than in adults. Also, immunologic reactions are more severe, and visual rehabilitation, including amblyopia therapy and contact lens correction, can be challenging. In spite of these problems, corneal surgery in young children is a viable option that can also be quite rewarding.
PENETRATING KERATOPLASTY
During the past three decades corneal transplantation in children has become more successful.1,2,3,4,5,6,7 What was previously viewed with considerable caution is now more widely accepted as a therapeutic option. However, the prognosis for keratoplasty in children is still not as good as it is in adults. Even when a clear graft is obtained the visual results often are poor, most commonly because of other ocular abnormalities or amblyopia. The visual results are best for older children and for eyes with acquired nontraumatic opacities and are poorer for infants with congenital opacities.
Penetrating keratoplasty is technically more difficult in infant eyes. The eyes are smaller, and the cornea and sclera are less rigid, predisposing the eye to collapse and the lens–iris diaphragm to prolapse forward once the eye is opened. In phakic eyes, spontaneous extrusion of the lens can occur. Also, dissection of anterior synechiae and corneal suturing are more difficult.
The postoperative course is complicated by the increased adhesiveness of the iris, rapid healing, and vigorous immunologic response. The child’s difficulty with comprehension and communication impairs examination and detection of problems. In young children the risk of rejection is higher: rejection episodes tend to be severe and rapid in onset, and detection is much more difficult. Even in eyes with a clear graft, astigmatism and anisometropia can cause dense amblyopia.
INDICATIONS
The indications for penetrating keratoplasty in children are the same as those in adults: optical, to remove opacity; tectonic, to repair a hole or ectasia; therapeutic, to remove disease; and cosmetic, to improve appearance. A wide range of conditions, including both congenital and acquired disease, may necessitate keratoplasty (Table 1).
Table 106-1. Indications for Penetrating Keratoplasty | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
There are many relative contraindications to penetrating keratoplasty in children. Because parental cooperation is so important, a low likelihood of such compliance is a contraindication to surgery. Corneal anesthesia, exposure, or severe tear deficiency markedly diminishes the likelihood of success. It is best to avoid operating on eyes with persistent intraocular inflammation, corneal or conjunctival infection, persistent epithelial defects, or elevated intraocular pressure.
Whether keratoplasty is indicated depends on the condition of the fellow eye, the patient’s age, the diagnosis, the presence of complicating conditions, and the family’s ability to participate in care. In general, keratoplasty should be considered for bilateral congenital opacities estimated to be 20/400 (Snellen notation), 6/120 (metric equivalent), or worse. The indications for keratoplasty for congenital monocular opacities should be more stringent than those for patients with bilateral disease, because the risk–benefit ratio is much higher. In older children with acquired corneal scars the prognosis is similar to that in adults, and surgery should be considered for a visual acuity of approximately 20/80 (6/24) or less.
PREOPERATIVE EVALUATION
Both the child’s eye and the child’s psychological and socioeconomic environment must be evaluated to determine the likelihood of a good surgical result. The parents must be motivated, be psychologically prepared, and have the resources to provide the demanding postoperative care. They must have the economic and social resources to handle the frequent office visits, transportation, loss of work, cost of medications, and hospital and physician fees. They need to have realistic expectations of the results of the procedure and they also need to be able to recognize the warning signs of complications, such as graft rejection, epithelial erosions, suture abscesses, infections, and recurrent herpetic keratitis; to administer medications; and administer therapy for amblyopia.
A thorough ocular examination is necessary to determine the cause of decreased vision, plan surgical treatment, and estimate prognosis. Most of the information usually can be obtained in the office, but general anesthesia may be necessary in very young or uncooperative children, particularly to obtain accurate measurements of intraocular pressure and corneal diameter. Generally, children up to 2 years of age can be forcibly controlled for examination and those older than 4 to 5 years of age can be convinced to cooperate. The greatest difficulty is encountered with children 2 to 4 years of age and those with mental impairment, because they are unable to understand and cannot be overpowered.
Parents should bring small children to the office hungry so that the physician can examine them while they are feeding. The experience should be kept as nontraumatic as possible: the surroundings should be pleasant, dim light should be used for examinations, good behavior should be rewarded, and no pain should be inflicted. If this is not sufficient, sedation with chloral hydrate (50 mg/kg oral syrup) or with midazolam can be helpful in young children.
Although a penlight or operating microscope examination can provide useful information, a portable slit-lamp is necessary for a complete examination. Intraocular pressure is best measured with a pneumotonometer or electronic tonometer, such as the Tono-Pen (Medtronic Solan, Jacksonville, FL), particularly if the cornea is edematous or irregular. Ultrasonic biomicroscopy may be helpful in some cases.
Inflammation of the eyelid margin, abnormal eyelid position, lagophthalmos, and trichiasis should be corrected before corneal surgery. The presence of an adequate tear film and corneal sensation should be determined. The position and size of corneal opacities should be measured with an adjustable slit beam or calipers. Corneal vascularization and thinning, anterior iris synechiae, anterior chamber inflammation, and lens opacities should be noted. If the fundus cannot be adequately visualized, then B-scan ultrasonography is indicated.
PROGNOSIS
The prognosis for obtaining a clear corneal graft in children is poorer than in adults. Overall 3-year survival rates have been estimated to be approximately 65%.8,9,10,11 A better prognosis is achieved with first grafts, avascular corneas, and eyes without ocular surface abnormalities. The prognosis is poorer for eyes where additional, non-corneal procedures (e.g., vitrectomy or cataract extraction) are required at the time of keratoplasty, and for eyes with peripheral anterior synechiae or glaucoma. The probability of achieving a clear graft for eyes with traumatic opacities tends to be higher: 84% at 1 year and 70% at 2 years in one series of 21 eyes.12 Transplants in children with congenital hereditary endothelial dystrophy had a similar success rate.13 Infant eyes with Peters’ anomaly have a poorer prognosis than eyes with other initial diagnoses. In one multicenter study graft survival rates were 79% at 1 year and 62% at 3 years.8 The success rate is higher if the dysgenesis is limited to corneal opacity (75%–80% in one author’s [GWZ] experience). In the largest study to date, 72 eyes of 47 patients, the probability of a first graft staying clear was 49% at 1 year, 44% at 3 years, and 35% at 10 years.9 The probability of second or subsequent grafts surviving for 3 years was less than 10%.
Even if clear grafts are maintained, the prognosis for good postoperative visual acuity is better for eyes with acquired traumatic or nontraumatic opacities than it is for congenital opacities, even when clear grafts are obtained.11,12,14 This observation is explained by the fact that amblyopia, strabismus, astigmatism, and glaucoma commonly limit visual rehabilitation in patients with congenital opacities. Young patients who are aphakic after penetrating keratoplasty have a particularly poor prognosis for retaining good vision.
It must be stressed that visual outcome in young children depends not only on obtaining a clear graft but on treatment of amblyopia. Early and aggressive postoperative refractive correction and amblyopia treatment are essential.
Although final visual acuities obtained with penetrating keratoplasty in children seem disappointing by some standards, several aspects should be considered. First, many eyes undergoing pediatric keratoplasty are severely damaged preoperatively, so that the initial visual acuity is very poor (finger counting or less). In these cases, the eye would have had no useful vision without surgery. A postoperative visual acuity of 20/200 (6/60) may allow children with severe bilateral opacities to ambulate and care for themselves, whereas they would have been unable to do so without surgery.
Second, even an edematous graft that might be classified as a failure may provide better visual acuity than the patient’s original cornea. Improvement in ambulation, communication, and behavior is often noticeable, even when a graft appears to be opaque and there is no measurable improvement in visual acuity.
Finally, the results of published series may underestimate the true success of penetrating keratoplasty in children. The percentage of eyes achieving clear grafts after a single surgical procedure is not as high as the percentage of eyes that eventually achieve long-term graft clarity. Visual acuities, too, may improve with time. This is particularly the case in infants whose visual acuity is measured before maturity. For example, R. Doyle Stulting (personal communication) observed a patient who underwent bilateral penetrating keratoplasty for Peters’ anomaly at the ages of 4 and 14 months who achieved a visual acuity of only 20/200 (6/60) in the right eye at the age of 6 years. Without further surgical intervention, the visual acuity improved in both eyes so that the best corrected vision at the age of 15 was 20/40 (6/12) in one eye and 20/100 (6/50) in the other. The patient now reads, drives, and functions well despite having a poor visual result at the time the anomaly was reported.
SURGICAL TECHNIQUE
Miotics should be avoided on the day of surgery, because they can exacerbate forward displacement of the lens–iris diaphragm. The surgery is performed with the use of general anesthesia, induced by an anesthesiologist who is comfortable managing children. Intravenous mannitol and preoperative ocular massage can be used to reduce vitreous pressure.
The donor should be at least 2 years of age. Younger corneas are less rigid, making tissue handling and suturing more difficult. They produce steeper postoperative corneas, which can reduce hyperopia from aphakia, but the postoperative refractive error is unpredictable and astigmatism appears to be a greater problem.15
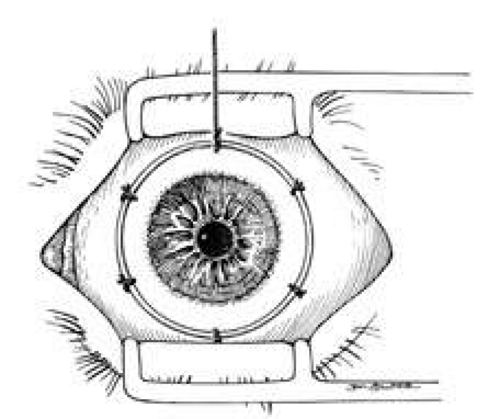
A Flieringa ring or McNeill-Goldmann blepharostat should be sutured to the globe to provide scleral support. One or two of the suture ends can be left long for fixation (Fig. 1). The center of the cornea and eight radial marks can be marked with a surgical pen and a radial keratotomy marker to guide suture placement. Alternatively, some trephines (e.g., Barron Recipient Trephine, Katena) are designed to create evenly spaced marks on the recipient cornea.
The diameter of the tissue to be removed is determined by the size and position of the pathology and the diameter of the recipient cornea. If possible, all thinned, necrotic, or infected tissue should be removed. The donor button should be at least 5.5 mm in diameter, and it is preferable that it be placed at least 2 mm from the limbus. The donor should be 0.5 to 1.0 mm larger than the recipient bed, to facilitate wound closure. Oversized grafts (>0.5 mm) tend to result in steeper postoperative corneas and therefore can be used to reduce hyperopia in aphakia. Similarly, they could lead to myopia in phakic eyes and should be avoided. Therefore, in infants, with a corneal diameter of 10 to 11 mm, typically a 7-mm donor is placed in a 6.5-mm bed. In a microphthalmic eye, particularly those with axial lengths of 13 mm or less, graft–host disparity of 1.5 mm appears to improve results.16
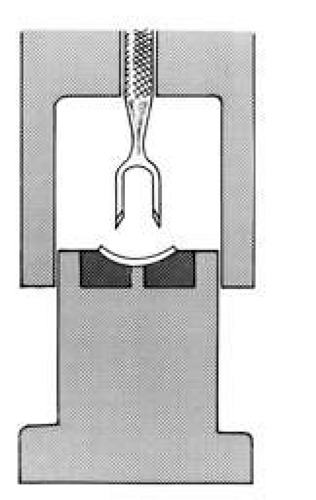
The donor corneoscleral rim is removed from the storage medium and placed in a trephine block, endothelial side up. The cornea is centered in the well, and excess medium is removed. A hollow shaft trephine is carefully centered and aligned vertically over the tissue and then firmly punched through the donor button (Fig. 2). A variety of trephines are available, some of which attempt to ensure centering and verticality of the cut, and some of which place four cardinal marks on the donor epithelium (e.g., Hanna trephine; Barron donor trephine). A drop of storage medium is placed on the endothelial surface, and the cornea is covered. The donor rim should be cultured.
A hollow shaft trephine is also used to cut the recipient cornea. A suction trephine (e.g., Hanna trephine, Barron recipient trephine) facilitates centering and verticality of the cut, and may leave marks for suture placement (Figs. 3 and 4). The trephine is centered on the corneal mark, and a cut of approximately two-thirds depth is made. The anterior chamber is then entered with a knife at the 9 to 10 o’clock (for a right-handed surgeon) position. Right- and left-handed corneal scissors are used to extend the incision (Fig. 5). The scissors are tilted slightly to create a posterior ledge of host cornea, which helps seal the posterior wound. Care is taken to cut the full thickness of the cornea, so Descemet’s membrane is not left in the eye, and to avoid damage to the iris and lens. Introduction of viscoelastic material into the anterior chamber may facilitate excision of the host cornea. It may be necessary to separate anterior synechiae, using blunt or sharp dissection. Vitreocorneal and corneolenticular adhesions are cut with scissors.
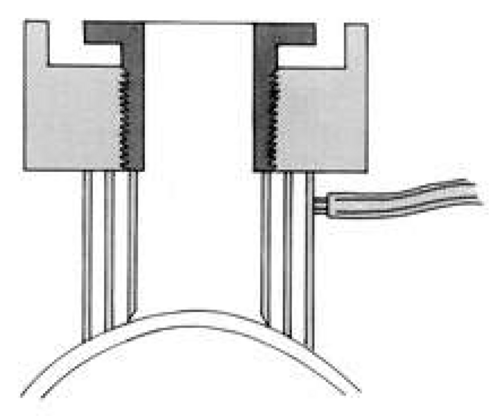 Fig. 3. Suction trephine on recipient cornea, cross section. Suction is formed between two outer rings. Central blade is lowered into cornea along threads.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|