Chapter 111 Surgery for Proliferative Diabetic Retinopathy
Introduction
Pars plana vitrectomy was originally developed by Machemer in 1971 as a closed system, allowing for a safe intraocular manipulation and constant viewing of the retina.1 At that time, indications were mainly nonclearing vitreous hemorrhages of greater than 1-year duration and complicated retinal detachments with macular involvement. However, in the past decades, improvements in technique and instrumentation have broadened the use of vitrectomy.2–5 Today, it has an established role in the management of many severe complications of diabetic retinopathy, together with many other surgical procedures.6,7 The principal underlying pathology in this disease is retinal ischemia, which may finally lead to the development of fibrovascular proliferations and membranes with the risk of secondary glaucoma, vitreous hemorrhage or retinal detachment. The principles and techniques described in this chapter may be applied to the medical and surgical treatment of other proliferative vascular retinopathies as well, e.g. retinal vein occlusions, Coats disease, or retinopathy of prematurity.
Following the pathogenetic concept in the evolution of proliferative diabetic retinopathy, the cornerstones are progressive retinal microvascular closures with ischemia (see also Chapter 47, Diabetic retinopathy: NPDR and DME, and Chapter 48, Diabetic retinopathy: PDR). They are the main causes of tissue hypoxia with subsequent development of macular edema and/or retinal and iris neovascularizations. These processes are triggered by diverse local pro-angiogenic factors, as insulin-like growth factor 1 (IGF-1), basic fibroblast growth factor (bFGF), and others.8–12
The conversion from nonproliferative to proliferative diabetic retinopathy was assumed to involve recruitment and proliferation of retinal vascular endothelial cells, eventually promoted by locally activated cytokines, as vascular endothelial growth factor (VEGF). This cytokine provokes endothelial cell growth and permeability,8–12 being associated with higher white blood cell counts and other inflammatory markers. The VEGF protein was found to be expressed in glial cells of the retina and optic nerve, retinal astrocytes, pigment epithelial cells, vascular endothelial cells and ganglion cells.12 VEGF is also suspected to mobilize and augment endothelial progenitor cells (EPC) from bone marrow by acting as a chemoattractant protein.13–16 Circulating EPCs then are assumed to directly go to the sites of ischemia or neovascularizations to initiate new vessel and tissue formation.13,14 The new (fibro-)vascular tissue may then proliferate in the space between retina and vitreous. With further ingrowth it may contract, potentially resulting in vitreous hemorrhage, which may stimulate further fibrosis and vitreous contraction, leading to retinal breaks or tractional detachment.12
Indications and timing of surgery
Cataract
Extracapsular cataract surgery with intraocular lens (IOL) implantation is usually well tolerated in advanced diabetic retinopathy, when there are no anterior segment neovascularizations.17,18 The removal of an opacified lens allows for a better fundus evaluation and visualization, e.g. for panretinal photocoagulation. In the past, higher incidences of iris neovascularizations, secondary glaucoma, and vitreous hemorrhage were reported after intracapsular cataract extraction in proliferative diabetic retinopathy, notably by Aiello et al.19 However, in recent times, as small incision cataract surgery, photocoagulation, and anti-VEGF drugs are widely used, anxiety has shifted from iris neovascularizations to diabetic macular edema. Progression may be lower when grid/focal lasers are applied before cataract surgery, or when panretinal photocoagulation is applied after lens extraction instead of before.20 Today, proliferative diabetic retinopathy should be treated with panretinal photocoagulation before cataract surgery whenever possible, but panretinal photocoagulation may also be applied at time of surgery or shortly thereafter.21 Along with improvements in vitrectomy surgery techniques, an increasing trend for simultaneous vitrectomy and cataract surgery has been observed in the past decades; lensectomy may be considered in revision vitrectomy surgery or in eyes with much reduced prognosis.22,23 The advantage of any cataract extraction is a better intraoperative view and access to the vitreous base, which is of upmost importance in cases of fibrovascular proliferations in diabetic retinopathy or proliferative vitreoretinopathy; there is also evidence from Schiff et al. that reoperation rates seem to decrease when the lens has been removed during vitrectomy.24 Similarly, fear of iris neovascularizations after combined surgery has been reduced by careful application of panretinal photocoagulation and anti-VEGF drugs during surgery.24 In younger patients with clear lenses, the loss of accommodation has to be weighed against possible intra- and postoperative complications, such as earlier cataract formation needing another surgery.17,24
High-risk retinal neovascularization
Fibrovascular proliferations
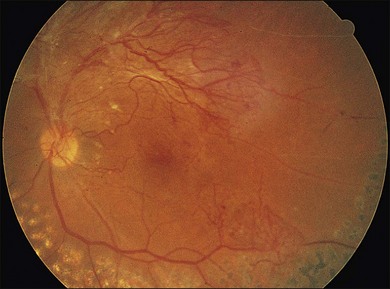
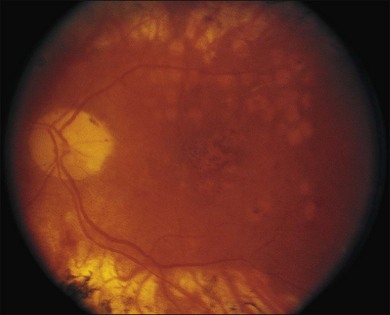
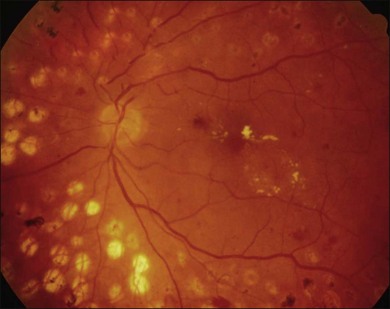
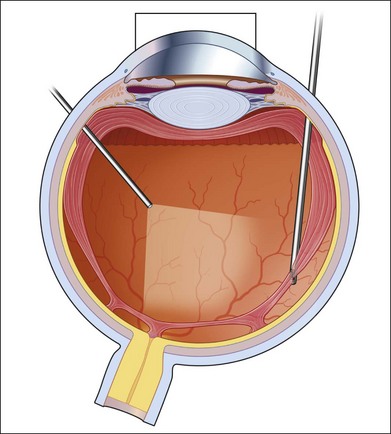
Severe fibrovascular proliferations in proliferative diabetic retinopathy can produce a major threat of profound loss of vision without surgical intervention. A progressive proliferation of fibrovascular preretinal tissue may occur, despite panretinal photocoagulation, as described by Hutton, Smiddy, and Ho (Figs 111.1–111.3).17,18,25
The Diabetic Retinopathy Vitrectomy Study (DRVS)26,27 formed the definition of “advanced, active, neovascular or fibrovascular proliferation” based on a review of studies of the natural history. The term “severe” for new vessels or fibrovascular proliferations was defined in the DRVS according to standard photographs and size definitions.26 Basically, the benefit of surgery tends to increase with increasing severity of neovascularization. Eyes most suitable for early vitrectomy are those with both severe fibrovascular proliferations and at least moderately severe neovascularizations despite extensive panretinal photocoagulation.6,27 More recent papers reported similar favorable surgical results for severe diabetic fibrovascular proliferations.
Stable or improved visual function may be achieved in 78% of cases on average. Good prognostic factors include younger age at baseline (<40 years), preoperative panretinal photocoagulation, better visual acuity (>5/200), no iris neovascularizations, and no iatrogenic breaks at surgery.26,28 Therefore, extensive panretinal photocoagulation is recommended prior to early vitrectomy to improve the patient’s outcome.25,29–31 In patients with relatively asymptomatic pathologies, intensive counseling is essential, as some eyes lose vision despite careful surgery.
Vitreous hemorrhage
Nonclearing vitreous hemorrhage in diabetic retinopathy was the earliest indication for vitrectomy in the 1970s, representing 70% of cases at that time.2 Today, it is still one of the most common indications for vitrectomy, although surgery may be avoided or at least postponed in many cases. Waiting, head elevation or intravitreal injection of hyaluronidase may lead to spontaneous blood clearing, thus allowing for panretinal photocoagulation to induce regression of active retinal neovascularizations.32,33 Diode or krypton laser systems, eventually delivered by indirect ophthalmoscopy might be more effective than argon laser in some cases.25 Early vitrectomy, defined by the DRVS as within 1–4 months from onset, results in earlier recovery of vision and better functional outcome after 2 and 4 years.27 The benefit is greater in patients with type 1 diabetes mellitus, compared to type 2. This difference might be influenced by a greater incidence of maculopathy and posterior vitreous detachment in elderly type 2 diabetic patients.34
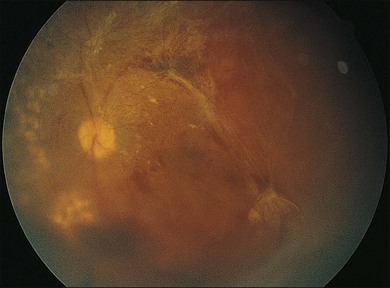
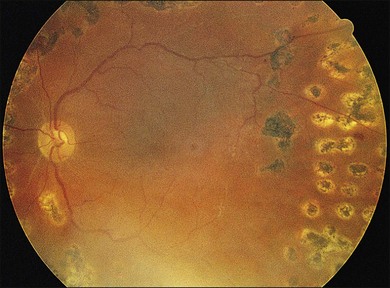
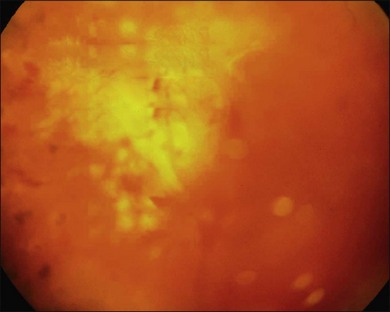
In proliferative diabetic retinopathy with dense premacular (subhyaloidal) vitreous hemorrhage, blood is trapped between the posterior hyaloid interface and the internal limiting membrane. The hemorrhage is usually well demarcated, resulting in massive visual loss. It may be associated with fibrovascular proliferations, preretinal membrane formation or tractional macular detachment, which are common indications for an early vitrectomy (Figs 111.4, 111.5).26 Less-invasive treatment methods include observation, laser membranotomy, or intravitreal injections with recombinant tissue plasminogen activator (r-tPA) or gas. When those methods are not successful, vitrectomy may improve functional recovery or decrease the risk of complications.26,30,35 Again, a longer delay than a few months for surgery is not recommended, as surgical dissection may become more difficult25,36 if the disease progresses. Another relatively urgent indication for vitrectomy in nonclearing vitreous hemorrhage is rubeosis iridis and/or severe progressive proliferation of the fellow eye, especially when no panretinal photocoagulation has been applied before.25,27
Macular traction and macular edema
Vitreomacular traction syndrome, vitreopapillary traction, diabetic macular edema, epiretinal membrane or macular hole formation in patients with proliferative diabetic retinopathy do have specific features in their presentation and management, representing relatively new indications for vitrectomy.37–41 Opacification of posterior vitreous cortex or preretinal membrane formation alone results in substantial visual loss, sometimes associated with metamorphopsia or diplopia.29,42 These changes may occur after premacular hemorrhage or extensive panretinal photocoagulation.26 Vitreomacular traction may be associated with more complex vitreoretinal adhesions than in nondiabetic patients, eventually resulting in tractional retinoschisis.43 Vitreopapillary traction is a relatively new, controversial indication for vitrectomy, with limited evidence for functional improvement.44 In eyes with coexistent macular edema, a causative role of vitreopapillary traction has been suggested. Diabetic epiretinal membranes are more likely to have focal attachments to the macula and more proliferative activity than idiopathic epiretinal membranes.39,40 In cases of diabetic macular edema, tangential traction or former intravitreal surgery, macular holes may develop.37,38,45 To avoid progression of diabetic macular edema, panretinal photocoagulation may be divided into smaller sessions or be applied after intravitreal injections of anti-VEGF drugs.46,47
All these pathologies are indicative for an early, extensive and careful surgical treatment, usually vitrectomy combined with central membrane peeling.48 Generally, the functional outcome is negatively associated with preoperative visual acuity and the degree of maculopathy.37,49
Retinal detachment
Tractional retinal detachment
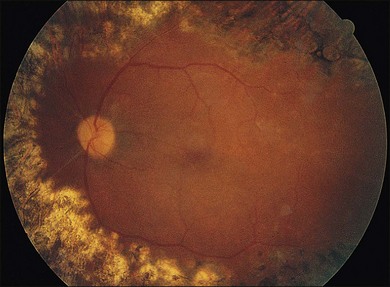
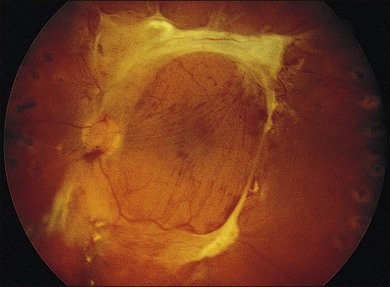
As neovascular membranes in proliferative diabetic retinopathy grow within the cortical vitreous gel, they may produce firm vitreoretinal adhesions and contract over time,41,50 resulting in tractional retinal detachment.51 Diabetic tractional macular detachment therefore has been the most common indication for vitrectomy.25 However, the management of peripheral retinal tractional detachment seems to have changed in recent times. Traditionally, those cases were observed for a while as the risk of complicated vitrectomy seemed to exceed the low progression rates.52 As anatomic and functional results after vitrectomy have substantially improved, an earlier surgical approach in cases with peripheral tractional detachment seems reasonable.53,54 Moreover, functional results after successful vitrectomy in severe macular tractional detachment are still poor (Figs 111.6, 111.7).54 Also, chronic cases of diabetic tractional detachment may be a lesser indication for surgery, as the retina under tractional fibrovascular proliferations usually becomes atrophic.6,18,54 In general, vitrectomy reoperation rates in diabetic tractional detachment are between 24% and 47%.55–57
Factors with a more favorable outcome in the literature are: age <50 years, preoperative panretinal photocoagulation; visual acuity >5/200; no or few iris neovascularizations or retinal proliferations; macular detachments <30 days, and no iatrogenic breaks.6,25,58
Combined tractional–rhegmatogenous retinal detachment
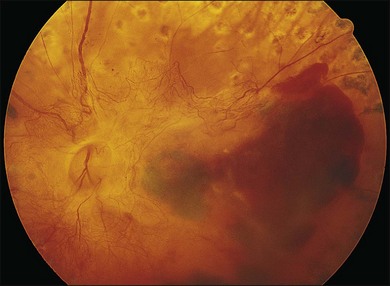
Severe fibrovascular proliferations in proliferative diabetic retinopathy may cause progressive traction and membrane contraction combined with posterior retinal breaks. The shape of the retina appears convex in contrast to tractional detachment, and the dimension of detachment is often greater, extending over the ora serrata.41,59,60 The retinal surface often shows white hydration lines, which are diagnostic of retinal holes. The holes are often small, located posteriorly, paravascular or immediately adjacent to vitreoretinal tractions and retinal elevations.60,61 Sometimes, subretinal hemorrhage may be present.62 Vitrectomy combined with silicone oil tamponade is frequently indicated in particularly severe cases, especially when the second eye shows a poor visual function (Figs 111.8, 111.9).60,63 Reports of silicone oil surgery generally show a high rate of reattachments with just a moderate chance of functional improvements (see below). Silicone oil finally helps to reduce the incidence of further complications, as neovascular glaucoma and phthisis in those desperate cases.60,63
Neovascular glaucoma
Neovascular glaucoma is a very severe complication in proliferative diabetic retinopathy. It is assumed that the ischemic retina is the source of vasoproliferative growth factors that may diffuse into the anterior segment. Consequently, growth of neovascularizations and fibrovascular membranes in the chamber angle obstruct aqueous outflow and intraocular pressure rises; different stages of neovascular glaucoma have been described.64,65 Therefore, the first therapeutic target should be the cause of the neovascular stimulus, indicating extensive panretinal photocoagulation or cryotherapy.66 Intravitreal or intracameral anti-VEGF medications, such as bevacizumab, may be helpful as short-term adjunct to panretinal photocoagulation or when panretinal photocoagulation fails to cause regression of rubeosis.67–69 This treatment alone usually induces regression of neovascularizations, however, fibrovascular proliferations in the chamber angle may contract and the pressure remains high.59
In cases of opaque optical media, as vitreous hemorrhage or cataract, controlled panretinal photocoagulation can only be performed after vitrectomy and/or cataract extraction, which has been shown to reduce rubeosis and improve neovascular glaucoma.70,71 In addition, silicone oil tamponade prevents recurrent vitreous hemorrhage and may induce regression of rubeosis.72
Vitrectomy may be combined with endocyclophotocoagulation of ciliary processes or partial retinectomy to improve perfusion and reduce intraocular pressure.73,74 Patients with higher stage neovascular glaucoma with synechial angle closure almost always need some sort of glaucoma surgery.
Filtering surgery in diabetic neovascular glaucoma has significantly lower success rates than surgery for primary or secondary open-angle glaucoma.75
The intraoperative use of antimetabolites, such as 5-fluorouracil and mitomycin C, is strongly recommended; in addition, intensive perioperative anti-inflammatory and antiproliferative treatments, as well as anti-VEGF injections and panretinal photocoagulation can improve the outcome.68,76
The implantation of glaucoma-drainage tubes (as Molteno, Baerveldt or Ahmed implants) is also very common, although the drainage capacity can be compromised by epibulbar scarring or recurrent intracameral or intravitreal haemorrhages.77,78 Nonpenetrating glaucoma surgery, as well as argon laser trabeculoplasty, is generally not recommended in diabetic neovascular glaucoma, as angle closure due to the rubeotic process can deteriorate postoperatively.75
Additional vitrectomy surgery should be considered at earlier stages of proliferative diabetic retinopathy and not only be reserved for advanced neovascular glaucoma.79 It should be combined with panretinal photocoagulation ab interno; in addition, a pars-plana glaucoma drainage implant may be considered to stabilize the glaucoma.77
Cyclodestruction, as transscleral cryo- or diode-laser cyclocoagulation, is a helpful, widely used method in advanced neovascular glaucoma. However, this treatment is usually reserved for eyes with low visual function at presentation.80,81 Blind, painful eyes may still need retrobulbar alcohol injections or, in the worst case, evisceration or enucleation.82
Preoperative evaluation and informed consent
As the presence of advanced diabetic retinopathy may indicate significant macro- and microvascular disease, all patients should be referred to an internist or endocrinologist before surgery. It is important to evaluate the patient’s medical and glycemic status as well as coexistent problems as hypertension, hyperlipidemia, cardiovascular or renal disease. Those findings will influence the decision for the extent, timing and prognosis of surgery.83,84 Optimal blood glucose management may be protective against perioperative infection.85 Patients should be well informed about adjustments of medications, especially those for blood glucose and blood pressure control. Anticoagulants as well as antiplatelet medications must be stopped or substituted at the surgeon’s suggestion. Another issue is that of patients needing renal dialysis. In those cases, surgery has to be arranged between dialysis sessions. In any case, an optimal medical control will optimize surgical success and reduce intra- and postoperative complications in diabetic patients.
Every patient must undergo a thoroughly ophthalmic evaluation before surgery to determine all anatomic abnormalities as well as actual and possible future visual function. It is important to correlate the history of visual decrease with possible anatomic changes, which can be found out by anamnesis or the referring ophthalmologists. This correlation is a major prognostic factor for surgical success. A complete ophthalmic status, with best corrected distance and near acuity, pupillary function, intraocular pressure and visual field tests is essential. Slit-lamp biomicroscopy of all anatomic abnormalities and, if possible, fundus examination with indirect binocular lenses, provides further important information to plan the surgical approach.86,87
Silicone intraocular lens implants should be avoided, as intraocular silicone oil tamponades would firmly adhere to silicone lenses, thereby affecting intra- and postoperative fundus visualization and visual function.88,89 If applicable, fluorescein angiography and optical coherence tomography (OCT) may add further details, as the presence and extent of retinal or iris neovascularizations, macular or retinal ischemia, macular edema, vitreoretinal tractions, and epiretinal membrane formation.84,86,90
In cases with opacified media preventing fundus visualization, as cataract, intracameral or intravitreal hemorrhage, ophthalmic echography should be performed; it can provide most relevant information, as the presence or absence of vitreoretinal adhesions, vitreoschisis, retinal detachment, or other subretinal opacities and tumors.87,91
Preoperative electrophysiological testing (visually evoked potentials, VEP or electroretinography, ERG) is another tool to evaluate function in those cases. However, in clinical practice it is not routinely used, as results in predicting postoperative outcome are sometimes contradictory.92
Prior to surgery, possible infections of the lid, conjunctiva, cornea or ocular adnexae must be treated. Antibiotic prophylaxis may be reasonable when the risk for endophthalmitis is increased, although there is no evidence-based general recommendation.41,93 The presence of iris neovascularizations or massive fibrovascular proliferations is an indicator for an early surgical intervention, with preoperative intravitreal or intracameral application of anti-VEG medications and panretinal photocoagulation, if possible.94,95 Especially in severe proliferative diabetic retinopathy in type 1 diabetic patients, there is strong evidence to perform adequate panretinal photocoagulation, especially in the anterior periphery to minimize the risk of further anterior neovascularizations or fibrovascular proliferations.6,29,96,97
Surgery
Education and training
Surgery for complications of diabetic retinopathy, notably vitrectomy, requires advanced surgical judgment and skills and the use of highly developed instruments and equipment. Fast technical advances in the development of instruments and surgical techniques require regularly and frequent trainings of surgical skills of all operating personnel as well as modernization and proper maintenance of all surgical equipment and instrumentation.84 Wet laboratories using animal models play an important role in modern ophthalmology surgical residency training. In recent times, new virtual reality simulators can be used as a gated, quantifiable performance goal to expert-level benchmarks (Fig. 111.10).98,99
Anesthesia
Surgery in proliferative diabetic retinopathy can be performed in local or general anesthesia, sedoanalgesia or a combination of those. The adequate form of anesthesia depends on many factors, as the extent and duration of surgery, the patient’s mental or physical condition, or just the patient’s and surgeon’s choice. It also depends on geographic and economic factors, as there are diverging anesthesia standards in different countries. The patient’s vital signs should be continuously monitored by experienced operating staff members, even during local anesthesia. The advantage of local anesthesia is a minimal disturbance of the diabetic metabolism; however, the patient may feel some pain or move during surgery. Local anesthesia can also be applied during sedoanalgesia or general anesthesia to minimize the patient’s postoperative discomfort.100 General anesthesia or sedoanalgesia should only be performed by an anaesthesiologist, who can stabilize the patient or help to medically reduce the intraocular pressure. Nitrous-containing agents should not be used or be terminated before an intraocular gas-bubble is injected.84,100
Preoperative preparation
To provide adequate intraocular visualization, wide pupillary dilatation is always necessary. A combination of different mydriatic, sympathomimetic and cycloplegic drops should be instilled repeatedly before surgery to allow for a maximal pupillary dilatation.84 Additional topical medications, as antibiotic or antiphlogistic drops may be added, as well as systemic sedative or diuretic medications for optimal preparation of the patient. If general anesthesia is scheduled, additional preoperative medication or modification of the patient’s medications should be discussed with the anesthesiologist.
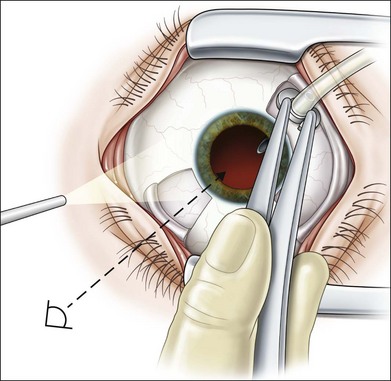
In the operating room, a 5% polyvidone iodine solution must be applied on the eyelids and a 5% solution in the conjunctival sack, respectively, and should dry out for at least 3 minutes to guarantee adequate disinfection.101 The eye is then covered with a sterile plastic sheet, equipped with 1–2 side bags, and a lid speculum is inserted (Fig. 111.11).
Surgical equipment
Microscope and lenses
As prerequisite, a modern binocular surgical (stereo-) microscope is required with co-axial illumination that should allow a magnification of 10–30-fold. It should be equipped with a motorized power zoom, power focusing and X–Y-positioning via foot pedals. The microscope must be fitted with the corresponding laser filters to permit photocoagulation. A light-splitter is necessary for co-visualization of the operating personnel and for the integration of a video system.102
For fundus visualization, different lens systems are available to neutralize the cornea’s refractive power. The initial visualization of the central retina was performed using hand-held, plano-concave lenses or various contact lenses centered by the assistant or a sclera-fixated metal ring. For a better visualization of the fundus periphery, especially in gas-filled phakic eyes, biconcave lenses with 20–35° angle were developed.103,104
Today, 130° wide-angle viewing systems are available, and the inverted image is corrected through a stereoscopic diagonal inverter. Non-contact wide-angle systems (BIOM, EIBOS) are widely in use and can be managed by the surgeon alone.102,105 They offer a greater depth of field and better visualization through media opacities. Also, a lower incidence of postoperative epithelial defects or retinal detachments was reported.106,107 To protect the corneal epithelium and guarantee for optimal fundus visualization, a corneal tear film must be maintained. The adjunctive use of carboxymethylcellulose gel or similar substances at surgery will promote corneal clarity.
Microinstruments and illumination
Various types of surgical instruments have been developed and modified over the years. The instruments vary in the number of functions provided. Currently, a trend is towards single-use instruments or parts of them, providing maximal aseptic conditions. For vitrectomy in proliferative diabetic retinopathy, 20-gauge systems have become the long-time standard and are still recommended. They still offer the greatest number of supplementary instruments with minimal flex.84,108
Small-gauge systems
In the past years, 23-, 25-, and 27-gauge instruments have been developed to provide non-suturing vitrectomy, thereby minimizing inflammation and postoperative discomfort to the patient (Fig. 111.12).109–112 However, their efficiency in complex cases, such as advanced diabetic retinopathy, is still a matter of debate, as a higher rate of postoperative hypotony has been reported.113 In the recent literature, 23-gauge systems showed more stable and reproducible results even in severe proliferative diabetic retinopathy, compared with 25-gauge.109,114,115
Basic equipment
The standard equipment for vitrectomy consists of a vitrectomy cutter, combined with a suction unit, a fiberoptic light pipe, an infusion of balanced salt solution (BSS) and an air pump. A modern vitrectomy unit provides all those base functions, in different combinations with diathermy, endolaser coagulation, gas filling or phacoemulsification modules (Fig. 111.13).
Illumination
Hand-held illuminators range from single-function illumination probes to multi-purpose illuminated scissors, forceps or vitrectomy probes. The use of “chandelier” light illuminators inserted manually or through additional sclerotomies allows for bimanual dissection.116–118
Membrane dissecting instruments
A wide variety of tissue scissors, forceps, spatulas, picks or cannulas are available to peel or remove epiretinal membranes; similarly, the vitrectomy probe can be used with lower suction rates at the decision of the surgeon. Vertical scissors may be used for segmentation of tissues in complex fibrovascular proliferations, whereas horizontal scissors are beneficial to delaminate the vitreous cortex from the retina.119,120
Dyes and tamponades
Various dyes are used to identify vitreous and epiretinal structures. Corticosteroid crystals may be used for easier identification of the vitreous cortex, especially in retinal detachment surgery; e.g., triamcinolone acetonide marks otherwise invisible remnants or patches of vitreous on the retina.101,121–123 In addition, it may help to prevent fibrin exudation in proliferative diabetic retinopathy due to its anti-inflammatory potential. No retinal toxicity was described for intravitreal doses of 2–4 mg of triamcinolone acetonide.102,124 Epiretinal membranes or fibrovascular proliferations must be carefully removed to prevent recurrent proliferative vitreoretinopathy or tractional detachment. Dyes as trypan blue are helpful to stain epiretinal membranes; indocyanine green, infracyanine green, and brilliant blue are more specific for internal limiting membrane identification; epiretinal structures appear in negative contrast with those substances.125–128 There is a divided opinion on whether infracyanine or indocyanine green might have toxic retinal effects, provoking (peripheral) visual field defects. However, this effect could be time- and dose-dependent.128,129
For internal tamponade of the vitreous cavity, various gases and liquids are in use. As a short-term intraoperative instrument, perfluorocarbon liquid is most commonly used. It is helpful to reattach the retina, or to protect the retina against damage from endo-phacoemulsification, intraocular foreign bodies or lens fragments.130–133 Filtered air may serve as a short-term, nontoxic tamponade; for a more prolonged tamponade in cases of retinal detachment or proliferative diabetic retinopathy, different gases as SF6, C2F6, or C3F8 are in use, providing tamponade times from 2 to 8 weeks. Gases are preferred for superior or posterior pathologies, in patients where positioning is possible, or when surgical removal would not be possible.134–136
Silicone oil is the instrument of choice for longer tamponades in most severe cases. Different silicone oil types from 1000 to 10 000 centistokes are available. They can also serve as protective shield to inhibit neovascular growth factors and cytokines to dissolve in ocular tissues. Silicone oils usually should be removed after a short time, usually 3–6 months, to avoid any silicone-related complications.137–141
Additional equipment
To improve the efficacy and outcome of surgery in proliferative diabetic retinopathy, different helpful adjuncts have been developed in the past years. A peri- or intraoperative injection of antiangiogenic drugs might decrease the risk of recurrent intravitreal hemorrhage or neovascular glaucoma with rubeosis iridis. Preoperative injections 7 days prior to surgery have shown to improve the outcome and facilitate surgical manipulations in diabetic tractional detachment.142,143 To facilitate posterior vitreous detachment and to shorten operating time, pharmacologic vitreolysis with plasmin, microplasmin and/or hyaluronidase was developed. Such agents may reduce intraoperative complications, such as retinal tears.144,145 When there is a need for visualization of the ciliary body in cases of severe anterior hyaloidal fibrovascular proliferation or extreme corneal opacification or capsule fibrosis, endoscopy provides a novel, elegant approach. The endoscope is inserted through the pars plana, providing a direct visualization of the entire vitreoretinal anatomy.146,147
Surgical procedure
Cataract surgery
Patients undergoing vitrectomy surgery for proliferative diabetic retinopathy may have concomitant cataract. Surgical management options include cataract surgery followed by vitrectomy surgery later, or combined operations in a single procedure.148 Further variations include cataract extraction, followed by vitrectomy and lens implantation at the end of surgery, or alternatively, cataract extraction with primary lens implantation, followed by vitrectomy. Cataract and vitrectomy surgery may be both performed by one surgeon or two different surgeons, depending on geographic and cultural differences.148
The advantage of any cataract extraction procedure is a much better intraoperative access to the vitreous base, which is of upmost importance in cases of fibrovascular proliferations in diabetic retinopathy or proliferative vitreoretinopathy; there is also evidence that reoperation rates seem to decrease when the lens has been removed during vitrectomy.24
Progression of coexisting diabetic macular edema may be lower when grid/focal laser are applied before cataract surgery, or when panretinal photocoagulation is applied after lens extraction instead of before.20 All cases with proliferative diabetic retinopathy should be treated with panretinal photocoagulation, if possible, and/or anti-VEGF injections before cataract surgery, to avoid a higher incidence of postoperative iris neovascularizations; panretinal photocoagulation may also be applied at time of surgery or shortly thereafter.21,24 In younger patients with less cataract formation, the loss of accommodation after cataract extraction has to be weighed against possible serious intra- and postoperative complications when the lens was not removed. The patient should be informed that an earlier cataract formation is common after vitrectomy.17,24 Likewise, it has been reported that in eyes without a crystalline lens, a more complete panretinal photocoagulation and resection of proliferations was possible.24
Glaucoma surgery
Aqueous shunt procedures
Nonpenetrating procedures, as canaloplasty or viscocanalostomy are usually not indicated, especially when the chamber angle is closed.149 Similarly, filtering surgery as trabeculectomy in neovascular glaucoma has significantly lower success rates than surgery for primary or secondary open-angle glaucoma.75
The reason is an excessive risk for inflammation and hemorrhage in these eyes. To improve the outcome, it is advisable to use intraoperative antimetabolites, as 5-fluorouracil and mitomycin C, as well as perioperative anti-VEGF injections and panretinal photocoagulation.68,76
It has recently been shown that an intravitreal injection of bevacizumab, followed by panretinal photocoagulation and glaucoma surgery 1–2 weeks later may produce a much better pressure control.150 Alternatively, glaucoma-drainage tubes, as Molteno, Baerveldt or Ahmed implants, may be implanted. They can be used either after or at the same time as vitrectomy with endophotocoagulation. The tube may be placed in the anterior chamber or in the sulcus ciliaris or through the pars plana, if the chamber angle is closed.149
Postoperative results are not better than standard trabeculectomy procedures, as the drainage capacity can be compromised by scarring of epibulbar tissue, bleb formation or recurrent intracameral or intravitreal hemorrhages.77,78 Furthermore, other long-term complications, as exposure of tube material or decompensation of the corneal endothelium have been described.150–152 Bevacizumab has been reported to improve the outcome after glaucoma implant surgery as well; however, more trials are needed to clarify the role of anti-VEGF medications in surgery for diabetic glaucoma.
In addition, vitrectomy surgery may be considered even at earlier stages of glaucoma in proliferative diabetic retinopathy, as it can be easily combined with full scatter panretinal photocoagulation ab interno.71,72,79 Furthermore, a pars plana glaucoma drainage implant may be considered to stabilize the intraocular pressure.77
Cyclodestructive therapy
In eyes with extensive retinal ischemia or optic nerve damage in advanced neovascular glaucoma, where visual outcome is expected to be very poor, the efforts and risks of incisional trabeculectomy or glaucoma implant surgery may not be accepted.149 In those eyes, cyclodestruction, as transscleral cryo- or diode-laser cyclocoagulation, is a helpful, method, depending on the grade of angle closure.80,81 In the right indication, it has proven as effectively as trabeculectomy or drainage implant surgery.153,154
Before transscleral cyclophotocoagulation, a retrobulbar injection with lidocaine 2% is usually given and a lid speculum should be used. The handpiece of the diode laser features a footplate designed for this procedure.149 The footplate is placed along the limbus so that the fiberoptic tip sits on the surface directly over the ciliary body to concentrate the laser energy in the target tissue. Power settings of between 1500 and 2500 mW with a pulse delivering time between 1.5 and 2.0 seconds are commonly used. A total of 15–30 applications of the laser are applied to the full circumference, only the horizontal meridians should be spared out.149,155 After surgery, topical prednisolone, diclofenac and atropine, eventually subconjunctival prednisolone-hydrogen succinate are given. The intraocular pressure will be reduced postoperatively for 6–8 weeks on average, providing effective pressure control in about 67% of patients.149,155 The treatment may be repeated after several months. Cyclocryocoagulation of the ciliary body in a similar fashion or cryocoagulation of the peripheral retina by indirect ophthalmoscopy may be an alternative, however “blind” cryocoagulation bears the risk of overtreatment and induction of choroidal neovascularization.70,156
Blind, refractory painful eyes may be still treated with retrobulbar alcohol injections or finally by evisceration or enucleation.82
Pars plana vitrectomy
Preparation of entry sites
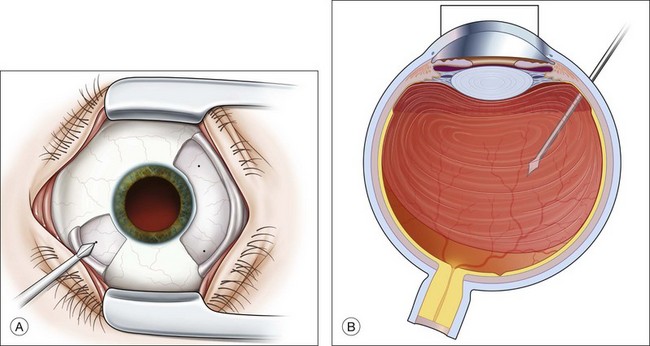
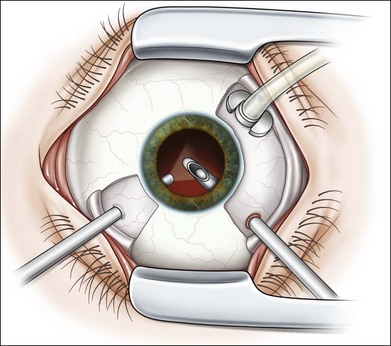
Three-port vitrectomy remains the most used technique, where two sclerotomies are prepared superotemporally and superonasally, and an inferotemporal pars plana incision permits intraocular infusion. Incisions are chosen in 3.5–4.0 mm distances from the limbus. Transconjunctival trocar-guided systems are used with increasing frequency in diabetes-related indications in smaller gauges (23- and 25-gauge) to provide higher comfort for the patients and reduce surgical trauma.109
To avoid intra- or postoperative wound dehiscence, sclerotomy blades should be oriented parallel to the limbus and trocars should be inserted 20–30° oblique to the scleral surface (Fig. 111.14).
However, this technology might be more indicated in easier diabetic cases such as nonclearing vitreous hemorrhage or macular edema.108,115 In eyes with complex pathologies where silicone oil injection is likely, 20-gauge incisions might provide the surgeon with a higher range of instrumentation and an easier silicone oil tamponade.108,113,115 Suturing of sclerotomies is recommended in silicone oil use because silicone oil can evade through unsutured wounds subconjunctivally. Precise infusion cannula placement is critical to avoid suprachoroidal infusion and choroidal detachment (Fig. 111.15).
Vitrectomy
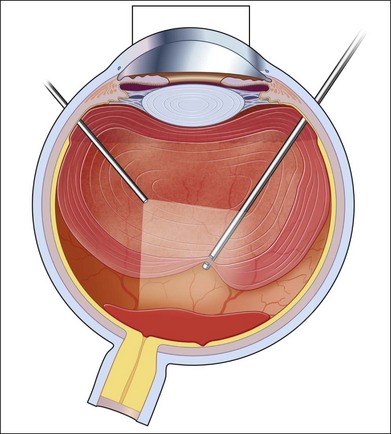
Light probe and vitrector are inserted through the sclerotomies and vitreous removal is started behind the lens (Fig. 111.16). The infusion is turned on only when the cutter is in the eye and vitreous removal can be started simultaneously. Otherwise, the infusion pressure will move the lens implant anteriorly and iris incarceration into the wound can occur. The anterior part of the vitreous is removed under microscopic view, then a wide-angle viewing system (e.g., BIOM) is inserted and central vitreous removal performed.
To facilitate identification of the vitreous cortex, triamcinolone acetonide may mark otherwise invisible remnants or patches of vitreous on the retina.122,123 Due to its anti-inflammatory potential it may also help to prevent fibrin exudation in proliferative diabetic retinopathy. Intravitreal doses of 2–4 mg of triamcinolone acetonide will offer sufficient staining with no retinal toxicity.102,124
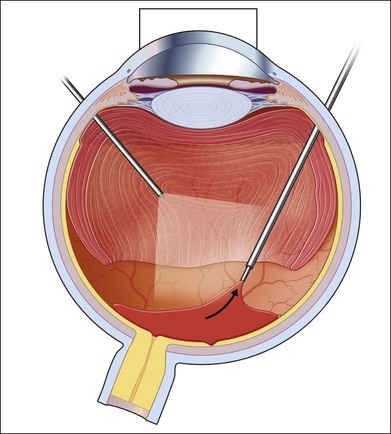
Eyes with complete posterior hyaloid separation
If a nearly complete separation of the posterior hyaloid is present after anterior and central vitreous removal, the posterior hyaloid membrane is incised and the opening enlarged circumferentially to allow adequate visualization of the retinal area (Fig. 111.17).12 While cutting rate is usually highest (2000–4000 cuts/min), aspiration might be increased in the mid-portion of the vitreous, but should be decreased again during posterior hyaloid removal to avoid unnecessary traction. Blood might be pooled at the posterior pole, usually unclotted, and can be aspirated now with a soft-tipped fluid needle (Fig. 111.18). As precise visualization of the retina is now achieved, areas of neovascularizations or small bleeding sources can be identified. Diathermy should be used for bleeding sources and then the vitreous is removed up to the periphery. Indentation is used to remove all anteriorly located blood and vitreous. Otherwise this can be a source for re-bleeding, tissue contraction and rubeosis iridis. Full-scatter endophotocoagulation is now performed up to the peripheral retina for the same reason.
In contrast, it was reported from a pilot study that peeling of the internal limiting membrane in proliferative diabetic retinopathy with fibrovascular proliferations might reduce postoperative epiretinal membrane formation.48
Eyes with incomplete posterior hyaloid separation
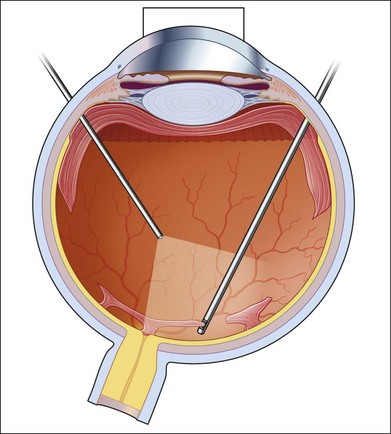
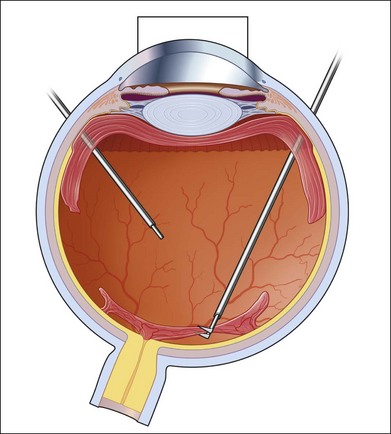
If incomplete separation of the posterior hyaloid is present, surgery can be difficult and should remain in experienced hands. Usually a core vitrectomy is performed at the beginning to gain sufficient view over the areas of adhesion and the connections between them. If there is a wide separation between the formed vitreous and the retina, circumferential release of anterior–posterior traction can be achieved with the vitreous cutter. However, if the posterior vitreous is closely overlying the retina in certain areas, care must be taken not to injure the retina (Fig. 111.19). If the retina is attached, gentle suction with the vitrector might be sufficient to further separate the vitreous from the retina to provide safe dissection. If the retina is detached or holes do exist, separation of tissue might be achieved with the use of some viscoelastic to provide higher safety.157–159 In any case, careful separation, usually with scissors, has to be done. Several surgical techniques for membrane removal have been developed. The first were delamination and segmentation.160 In segmentation, tractions between centres of adhesions are removed (Figs 111.20, 111.21),12,52,161 while in delamination, the connections between the posterior hyaloid and/or fibrovascular tissue and the internal limiting membrane are cut (Fig. 111.22).161–163 The later developed “en bloc” technique includes removal of the vitreous and associated vitreoretinal membranes as a single unit (Fig. 111.23).164 If only one or few focal adhesions do exist, vitrectomy might be started with a core vitrectomy, followed by excision of the posterior hyaloid over 360° in order to separate small islands of adhesions. If extensive, firm adhesions are present, the “en bloc” technique might facilitate surgery: a core vitrectomy is performed and the posterior hyaloid opened in an area close to or over the optic nerve. The fibrovascular tissue is now grasped with an end-gripping forceps and the tissue separated. Gentle traction is exercised to avoid bleeding or hole formation. If it is possible to loosen the connected tissue over the posterior pole in one piece, then the remaining hyaloid of the peripheral vitreous will lift the residual vitreous and membranes into the mid-vitreous cavity, where it can be removed safely (Fig. 111.24). As tempting as it might sound to remove everything in one big piece, two things need to be mentioned: first, complete removal of membranes and vitreous together in cases with adhesions of different strength is rarely possible. Second, bleeding from several sources might create a less controllable situation. If confronted with a firm adhesion during the “en bloc” technique, a change to delamination or segmentation is advisable. The newer cutters of the 23-gauge systems have an opening nearer to the end tip and do allow segmentation of tissue without additional scissors or picks (Fig. 111.25). The use of some perfluorocarbon to prevent bleeding into the foveal area can be helpful if a bridging membrane has been removed, but further dissection needs to be done in the mid-peripheral retina. If the retina is cleaned from all fibrovascular tissue up to the periphery and bleeding sources are cauterized, circular photocoagulation treatment is performed up to the ora serrata under indentation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree