Surgery for Pediatric Glaucoma
James D. Brandt
Abraham W. Suhr
Primary congenital glaucoma is a rare disease, occurring in approximately 1 out of every 15,000 live births.1,2,3 Because of its rarity, it has been estimated that the general ophthalmologist may make the initial diagnosis of this disease only once in a lifelong career. Nonetheless, every ophthalmologist should be familiar with the various surgical approaches to this disease, because they are applicable to the management of the various secondary glaucomas seen in infancy and childhood that, although not common, may be seen a few times a year in a busy general ophthalmic practice.
The purpose of this chapter is to outline a therapeutic and surgical approach to the management of congenital and pediatric glaucoma. It should be emphasized at the outset that this chapter does not substitute for experience in handling these sometimes complicated surgical problems. Because the initial surgery for these problems is often the procedure that has the highest likelihood of success, and because the infant or child faces the prospect of a lifetime of impaired vision should the surgery be unsuccessful, the management of these patients is best left to those experienced with these procedures. The postoperative management of congenital glaucoma in reality lasts a decade or more (with periodic examinations under anesthesia, amblyopia therapy, contact lens fittings for aphakia, etc.); the management of these complicated problems is best left to those physicians and/or practices where a team approach can be brought to bear for the patient’s benefit.
Preoperative Considerations
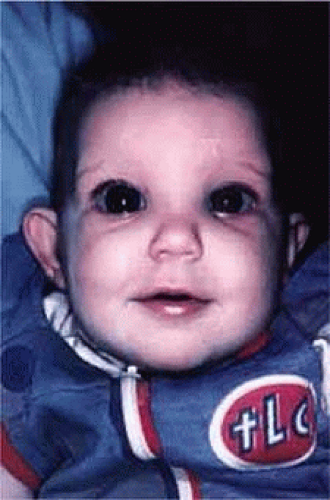
The embryology, epidemiology, and differential diagnosis of primary infantile glaucoma is covered elsewhere in this series. (See Volume 3, Chapter 51, “The Developmental Glaucomas.”) In many cases, particularly where the glaucoma is unilateral, the diagnosis is relatively straightforward (Fig. 1). In bilateral cases in young infants, however, the diagnosis may be less certain and the ophthalmologist will find it useful to perform a brief examination in the office before subjecting the child to a general anesthetic.
In infants less than 3 or 4 months of age, it is usually possible to perform tonometry using either a handheld Goldmann applanation tonometer (Perkins Tonometer), pneumatonometer, or a Tonopen (Reichert Instruments, Depew, NY), an electronic tonometer based on the Mackay-Marg principle. The Schiötz tonometer is relatively inaccurate when used to measure the intraocular pressure (IOP) of an infant eye and can be used only to gauge a general range of IOP. The young infant should be swaddled and held in its mother’s arms while the eyelids are gently pried apart to perform tonometry. The Tonopen device, which has a very small (≈2 mm) measuring surface, is well suited to the small palpebral fissures of an infant4 and can measure intraocular pressures without placing undue pressure on the lids. Also, the use of a lid speculum can often be avoided. The observer’s head does not need to be right next to the patient’s, which can alarm some children. The Tonopen has been found to be accurate when compared to both cannulated cadaver eyes and Goldmann tonometry in adults,5,6 but Eisenberg et al. found in children that the pneumatonometer performed best clinically; in their study, applanation tonometry markedly underestimates IOP in young eyes, and they felt that the Tonopen was not sufficiently accurate for clinical use.7 The authors have found each of these instruments useful in different settings, and the discrepancy between the instruments only emphasizes the fact that a diagnosis cannot be made based on tonometry alone.
The office measurement of an intraocular pressure much greater than 20 mmHg (by whatever technique) in a calm, resting infant is suspicious for glaucoma when other signs and symptoms suggest the disease,8 as is an asymmetry of more than 5 mmHg in suspected unilateral or asymmetric cases. Intraocular pressure measurements while a child is crying are usually not interpretable, because a nonglaucomatous infant can have an intraocular pressure in the forties or fifties when crying vigorously and squeezing the eyes shut.
Ophthalmoscopy is at the heart of the diagnosis of congenital glaucoma, and can be greatly facilitated in the office by using a Koeppe diagnostic infant gonioscopy lens that has no central depression. These lenses permit good visualization of the disc, even through a small pupil.
When the diagnosis of glaucoma is strongly suspected on the basis of the office evaluation, an examination under anesthesia should be recommended to the child’s parents. After the diagnosis is firmly established, the parents are often in a state of shock, unable to believe that their newborn child has a potentially blinding eye disease. The ophthalmologist should devote a significant amount of time to the counseling and education of the child’s parents regarding the diagnostic and surgical plan. It must be emphasized to the parents at the outset that although the surgery is usually highly successful, a decade or more of ophthalmology follow-up will be necessary. Frequent examinations under anesthesia, amblyopia therapy, and other interventions are part of the long follow-up care, particularly in early childhood. Because some of the infantile and pediatric glaucomas represent genetic diseases,9 with subsequent siblings at increased risk for the disease, appropriate counseling by a clinical geneticist familiar with ophthalmic diseases should be offered and recommended.
Newly diagnosed congenital and pediatric glaucomas are relative emergencies, and in most cases, an examination under anesthesia and probable surgery should be scheduled within a few days of the initial diagnosis. When for logistical or medical reasons this proves impossible, the intraocular pressure can attempt to be lowered using aqueous suppressant medications. A 12-hour dosage schedule of topical ß-adrenergic antagonists is often safe,10,11 but because the potential for systemic side effects is increased in a small infant, the lowest dose should be used initially and the infant must be carefully monitored.10 Carbonic anhydrase inhibitors, both topical and oral, can also be used. Acetazolamide can be administered parenterally or orally, 5 to 10 mg/kg of body weight, every 6 hours. For outpatient use, the pharmacist will usually find it easiest to compound an elixir based on the parenteral powder; as much as 500 mg can be made up in 5 mL of strongly flavored elixir, but 250 mg/5 mL is more palatable. The elixir is stable for about 1 week at room temperature; refrigeration improves the taste, but does not lengthen stability. Alpha agonists, such as apraclonidine 1% and brimonidine 0.2% are effective, but are contraindicated in infants, because they can cause apnea and CNS depression.12 All of these medications have the potential for significant side effects, including respiratory12,13,14,15 and electrolyte imbalances, and they should be administered in close cooperation with a pediatrician. Topical prostaglandin analogues administered nightly are systemically safe, but their efficacy in reducing IOP can be difficult to predict.16
Medical therapy for congenital glaucoma is rarely appropriate for long-term use and should be employed primarily when an examination under anesthesia and surgery must be deferred. It is far better to proceed promptly with the definitive evaluation and treatment.
The Examination under Anesthesia
General anesthetics are known to have a potent effect on the intraocular pressure in both normal and glaucomatous individuals. The anesthesiologist responsible for the general anesthetic in these cases should be familiar with the issues involved and be willing to work with the surgeon to obtain the most useful information possible during the crucial first few minutes of the anesthetic.
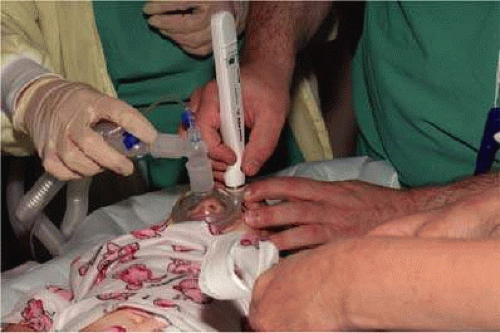
Virtually all of the inhalational anesthetics are known to depress the intraocular pressure within minutes of their administration.17 Ketamine HCl slowly elevates the intraocular pressure as deeper anesthesia is attained.18,19 The benzodiazepines do not appear to have a significant effect on intraocular pressure when used in preoperative doses. Midazolam HCl (Versed) is often used as a preoperative sedative in children and, in many cases, the child is sufficiently sedated with this medication to perform a quick intraocular pressure measurement prior to the administration of an inhalational general anesthetic. Such a measurement, with the child resting comfortably but not under the influence of the potent inhalational anesthetics, is probably the most accurate. If such a situation cannot be achieved, as in the case of a highly agitated infant or toddler, the anesthesiologist should be aware that it is preferable for the surgeon to measure the intraocular pressure as early as possible during the induction of general anesthesia (Fig. 2). At this point in the anesthetic induction, however, airway management is of utmost importance, and the surgeon must defer to the judgment of the anesthesiologist as to when measurements can safely be taken. In most cases, laryngeal mask or endotracheal intubation is appropriate, especially if an extensive EUA with gonioscopy, photography, and ultrasound is planned.
Table 1 lists the information that should be obtained during the course of an initial or follow-up examination under anesthesia. After the initial IOP measurements are made, the surgeon should indicate to the anesthesiologist what remains to be performed during the course of the examination, if surgery is not planned. Portions of the examination that require a clear media, such as ophthalmoscopy, retinoscopy, and fundus photography, should be performed first, followed by axial length measurement, gonioscopy, and corneal diameter measurements.
Table 1. Essential Information for the Examination under Anesthesia | |
|---|---|
|
Preoperative dilation should be avoided in cases where the iris and anterior chamber angle appearance will be important factors in making the initial diagnosis or in cases where corneal opacities prevent any view of the fundus. However, ophthalmoscopy, retinoscopy, and fundus photography are important parts of the initial and subsequent evaluations of a child with glaucoma, and preoperative dilation with a short-acting mydriatic should be entertained before entering the operating room to avoid needlessly prolonging anesthesia while waiting for dilation to occur. If necessary, intracameral acetylcholine can be instilled during surgery to constrict the pupil to allow safe intraocular maneuvers.
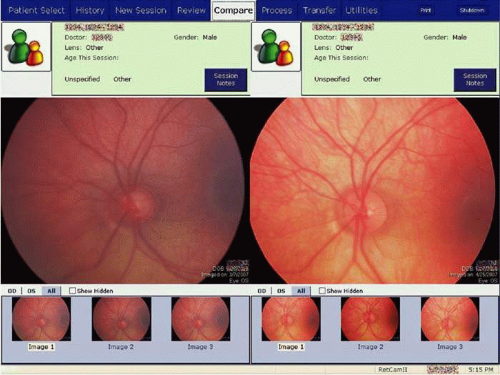
Funduscopy is essential, both with a direct and indirect ophthalmoscope. Both the posterior pole and the periphery should be carefully evaluated. Fundus photography of the optic nerve allows the surgeon to document the extent of the progression of the disease. We have found the RetCam II (Clarity Medical Systems, Inc., Pleasanton, CA) to be invaluable for both capturing the baseline optic nerve appearance and for comparing optic nerve appearance during serial examinations under anesthesia (Fig. 3). It is well known that the optic nerve cupping seen in congenital and juvenile glaucoma can to some extent reverse itself,20,21,22 and this can be used in some cases to gauge the success of therapy (Fig. 4). Similarly, axial length measurements by ultrasonic biometry can help establish the degree of disparity between the two eyes, particularly in unilateral cases.23,24,25 In many cases, the disparity decreases after successful therapy25 and, thus, a constant or increasing disparity between the two eyes may signal a worsening clinical situation.
 Figure 3. Use of the RetCam II to document the appearance of the optic nerve during an examination under anesthesia. |
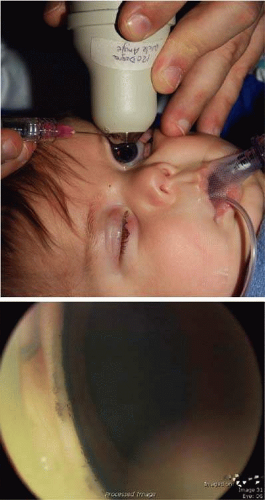
Gonioscopy is a mandatory part of the examination under anesthesia. It allows the surgeon to identify the underlying congenital or juvenile glaucoma diagnosis and permits appropriate surgical planning. The Koeppe gonioscopic lens introduced in 1919.26 has significant advantages over the Zeiss 4-mirror or Goldmann 3-mirror gonioprism. Chief among these is the ability to simultaneously compare the gonioscopic appearance of two eyes in the same patient (Fig. 5). The surgeon should be familiar with the gonioscopic findings in primary congenital glaucoma as well as the various secondary glaucomas seen in childhood. Gonioscopic photography can help document the appearance of the anterior chamber angle before and after treatment. We have found the RetCam II wide angle lens directed at the angle with a large glob of gonioscopic gel to overcome total internal reflection can document angle structures well (Fig. 6).
 Figure 5. A significant advantage of Koeppe gonioscopy is the ability to compare the appearance of the anterior chamber angle in both eyes at one time. |
The cornea should be measured both horizontally and vertically, and the presence or absence of Haab’s striae or other corneal abnormalities should be noted. Enlargement of the corneal diameter over time is worrisome for progressive buphthalmos, and some clinicians feel these measurements to be more useful than axial length determinations.27 In many cases, however, these measurements are difficult to make accurately or consistently because of uncertain landmarks at the corneo-scleral limbus. Considered a standard component of the adult glaucoma evaluation, the measurement of central corneal thickness can prove useful in select pediatric glaucomas associated with other ocular or systemic abnormalities.28,29,30,31,32,33
Cycloplegic retinoscopy during follow-up examinations (both under anesthesia and in the office) are used to monitor for progressive axial myopia34 as well as to provide the spectacle correction to be used in conjunction with patching and/or other amblyopia therapy.
Choosing the Surgical Approach
The surgical options in the management of congenital and pediatric glaucomas, along with their relative indications, advantages and disadvantages are outlined in Table 2.
Table 2. Surgical Options in Surgery for Pediatric Glaucoma | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In cases of primary infantile glaucoma, several studies have demonstrated that goniotomy ab interno and trabeculotomy ab externo are essentially comparable, with surgical success rates approaching 80% to 90%1,35,36. McPherson found that with multiple operations both procedures controlled intraocular pressure in most patients, but more operations were required after goniotomy37. Goniotomy ab interno is a simple and elegant procedure, but requires excellent visualization of the anterior chamber angle. Trabeculotomy ab externo is no less elegant and can be performed in virtually all cases. Both procedures have been made much safer by the introduction of viscoelastic materials. The surgeon who anticipates performing a significant amount of congenital glaucoma surgery should be familiar with both techniques.
In cases where the clinical situation permits the use of either approach, the surgeon’s personal preference and logistical considerations should dictate the choice. A goniotomy is greatly facilitated by a surgical assistant who is familiar with the procedure and understands what the surgeon is trying to visualize and achieve. A trabeculotomy, on the other hand, can be performed with a scrub nurse as the only surgical assistant.
The choice of surgical approach in the case of secondary pediatric glaucomas is more complicated. As pediatric ophthalmologists have become more aggressive about treating congenital cataract, perhaps the most common secondary glaucoma seen in children today is that associated with surgical aphakia.38,39,40 In this form of glaucoma, both open-angle and secondary-angle closure variants are recognized; it is thought that both may be the result of chronic, low-grade inflammation caused by residual lens material. In these cases, the surgeon is confronted with the dual challenge of lowering the intraocular pressure and maintaining aphakic correction to prevent the development of amblyopia. Careful preoperative gonioscopy is mandatory to determine whether (and where) a goniotomy should be attempted or whether the peripheral anterior synechiae present will lend themselves to goniosynechialysis (performed in a manner similar to goniotomy). Trabeculotomy ab externo should be avoided in cases where extensive, broad peripheral anterior synechiae are present. Lysis of these adhesions by the trabeculotome can be associated with significant intraocular bleeding and result in a slowly clearing vitreous hemorrhage and/or hyphema which can be profoundly amblyogenic. In such cases, the implantation of an aqueous drainage device should be considered as a primary procedure. As will be discussed subsequently, even though filtration surgery with antimetabolite use has a reasonable chance of pressure-lowering success, it should probably be avoided, because of the problems associated with extended wear contact lens use in the presence of a filtration bleb.
In cases of inflammatory glaucoma in children and young adults where significant peripheral anterior synechiae are absent, trabeculodialysis has proven to be useful in some hands41,42 with success rates of 50% to 60%. This approach is attractive in that it avoids the creation of a filtering bleb, which is prone to endophthalmitis in patients on long-term topical steroids.
Another secondary glaucoma seen in children is that associated with Sturge-Weber syndrome. Glaucoma presenting with this and related syndromes (e.g., Klippel-Trenaunay-Weber Syndrome) at or shortly after birth is usually associated with an angle appearance similar to primary infantile glaucoma. Later presentation of glaucoma in this group of syndromes is probably associated with a combination of abnormal angle structure and elevated episcleral venous pressure. A variety of surgical approaches have been used in the management of this glaucoma.43,44 In infantile cases where an abnormal angle is noted, goniotomy should probably be used as the initial approach because this procedure allows the surgeon to best control the intraoperative intraocular pressure in these eyes, which are prone to develop intraoperative choroidal expansions.
Congenital aniridia is another syndrome associated with glaucoma for which many procedures have been attempted with varying success.45,46 In this syndrome, true agenesis of the iris is rare; in most cases, a tiny, hypoplastic iris can be identified on gonioscopy.47 Although angle surgery can be successful in cases presenting in infancy,45 it is common for this iris stump to rotate anteriorly, closing off what little open angle remains, leading to late failures. The Molteno implant and other aqueous drainage devices have proven extremely helpful in these difficult cases and should be considered as a primary procedure when gonioscopy demonstrates angle closure.46 A recent finding among patients with aniridia is their profoundly thickened corneas, making applanation intraocular pressure measurements readings difficult to interpret.28,29 Serial gonioscopy and optic nerve examination are central to the management of glaucoma in patients with aniridia, as tonometry is unreliable.
Other more unusual syndromes associated with glaucoma, including Peters anomaly, sclerocornea, and microcornea, are handled on a case-by-case basis. The results of surgery for these problems are dismal, particularly in unilateral cases, but in bilateral cases, it is difficult for the surgeon to stand back and do nothing. These eyes usually must be tackled using a team approach, often combining a penetrating keratoplasty with a cataract extraction, vitrectomy, and aqueous drainage device. Such an aggressive approach may provide the patient with some ambulatory vision.
Goniotomy ab interno
When Otto Barkan introduced goniotomy ab interno in the late 1930s,48,49,50 the management of congenital glaucoma was revolutionized. Congenital glaucoma was an incurable disease and among the leading causes for admission to schools for the blind. The basic principles of the procedure have changed little since it was first described, but goniotomy has been made safer and more successful with the introduction of better instrumentation, viscoelastics, and the operating microscope.
Excellent visualization of the anterior chamber angle is needed to perform the procedure safely and accurately. Mild corneal edema often clears as IOP drops following general anesthesia. If not, edema can sometimes be overcome by denuding the cornea of its epithelium, either by blunt scraping or by topical alcohol and glycerin. Unfortunately, corneal clarity sometimes worsens during the course of surgery. If the surgeon anticipates that visualization may become a problem during the course of surgery, or if large Haab’s striae or other corneal opacities prevent adequate visualization of the anterior angle structures, a trabeculotomy ab externo should be performed instead.
When a goniotomy is planned, the anesthesiologist should be informed that the surgeon and assistant will be manipulating the patient’s head back and forth during the course of the procedure and that the endotracheal tube should be secured with this in mind.
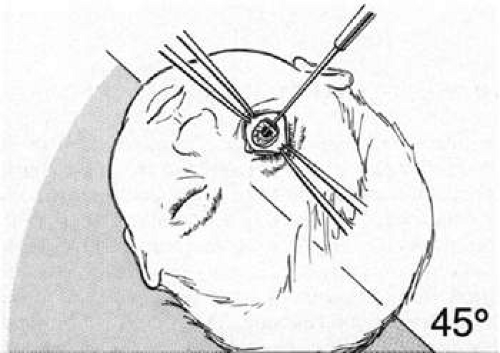
It is technically easiest to perform a goniotomy over the nasal 120 degrees of the anterior chamber angle, using a temporal approach. This is the usual site of an initial goniotomy. The patient’s head is turned away from the surgeon approximately 45 degrees from vertical and is secured firmly in place (Fig. 7). If the surgeon uses an operating microscope rather than loupes, the microscope head should be tilted in a manner appropriate to permit adequate visualization of the nasal anterior chamber angle after the gonioscopic prism is placed on the eye.
The assistant, who is seated across from the surgeon or at the head of the table, grasps the eye at the 6 o’clock and 12 o’clock positions with toothed forceps to rotate the eye nasally or temporally at will. Bridle sutures are usually not necessary. The surgeon then creates a limbal stab incision through the cornea just anterior to the corneoscleral limbus and injects a viscoelastic substance into the anterior chamber in order to deepen it.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree