Chapter 110 Surgery for Ocular Trauma
Principles and Techniques of Treatment
Extent of ocular injuries
It is estimated that 1.6 million blinding ocular injuries occur worldwide each year, with an additional 2.3 million sustaining bilateral low vision from trauma, and almost 19 million with unilateral blindness or low vision.1 Ocular trauma is second only to cataract as the most common cause of visual impairment in the USA. Of 2.4 million eye injuries each year, 40 000 are left with significant impairment.2 Overall, approximately 1 million Americans are visually impaired due to ocular trauma, and the annual cost of ocular trauma in the USA is estimated at between US$175 million and US$200 million for hospital care alone.3
According to the United States Eye Injury Registry, ocular injuries are more common among younger people, males, and those with less education and wealth. Statistics from the National Institute for Occupational Safety and Health on work-related diseases and injuries, indicate that over 800 000 work-related ocular injuries are reported annually in the USA. The impact of these injuries is compounded by the personal and national financial burden incurred. Total direct costs of workers’ compensation claims can be estimated at US$924 million, and estimated additional indirect costs (such as time lost by non-injured workers and production slowdowns) exceed US$3 billion.2,4
Ocular trauma classification
In 1996, the Ocular Trauma Classification Group devised a standardized terminology for ocular trauma. The classification has improved communication between physicians and accuracy in clinical practice and in research (Table 110.1),5 and it has been incorporated into many subsequent retrospective studies.
Table 110.1 New standardized classification of ocular trauma terminology
| Term | Definition |
|---|---|
| Eyewall | Sclera and cornea |
| Closed-globe injury | The eyewall does not have a full-thickness wound |
| Open-globe injury | The eyewall does have a full-thickness wound |
| Rupture | Full-thickness eyewall wound caused by a blunt object; the impact results in momentary increase of the IOP and an inside-out injury mechanism |
| Laceration | Full-thickness wound of the eyewall, usually caused by a sharp object; the wound occurs at the impact site by an outside-in mechanism |
| Penetrating injury | Single laceration of the eyewall, usually caused by a sharp object |
| Intraocular foreign body (IOFB) | Retained foreign object(s) causing body injury and entrance laceration(s) |
| Perforating injury | Two full-thickness lacerations (entrance + exit) of the eyewall, usually caused by a sharp object or missile |
Closed-globe injuries
Hyphema
Traumatic hyphema, or anterior chamber hemorrhage, can often manifest after blunt trauma. Most hyphemas clear spontaneously and do not cause residual visual impairment. However, permanent visual impairment can arise from complications of hyphema. These complications include: (1) corneal blood staining;6 (2) ghost cell glaucoma as a result of blocked outflow from clogging of the trabecular meshwork by erythrocytes;7 or (3) central retinal artery occlusion from elevated intraocular pressure.8 All these complications are more common when secondary bleeding occurs, usually 48–72 hours after the injury.
The laboratory workup for traumatic hyphema is tailored to the specific patient. While laboratory evaluation is not indicated in most patients with a hyphema, blood coagulation tests (partial thromboplastin time, prothrombin time, bleeding time, platelet counts, and liver function tests) should be considered in patients with a known bleeding diathesis or pertinent review of systems. African American patients should be questioned about a family or personal history of sickle cell disease or trait. If unknown, a sickle preparation or hemoglobin electrophoresis should be obtained. Patients with sickle cell hemoglobinopathies and traumatic hyphema present special problems because of their decreased ability to tolerate modest rises in intraocular pressure. Goldberg9 reported four patients with sickle cell hyphemas who developed optic atrophy with elevated pressure for short periods. This presumably is due to decreased perfusion pressure, sludging and sickling of erythrocytes and subsequent infarction. In addition, because of its lower likelihood to cause systemic acidosis that promotes sickling, methazolamide (Neptazane) rather than acetazolamide (Diamox) should be used to decrease intraocular pressure in these patients. Mannitol should be used only once to avoid hemoconcentration and the subsequent erythrocyte sludging and vascular obstruction.
Primary management of traumatic hyphema is directed at prevention of rebleeding, which is a significant risk factor for the development of glaucoma.10 Rebleeding has been reported to complicate up to 35% of cases11; the wide range in reported incidence likely relates to variability in patient ethnicity, treatment, and severity of injury. Previous clinical studies have demonstrated the benefits of shielding the injured eye to prevent accidental repeat trauma, using atropine for cycloplegia, and daily checking of visual acuity, corneal status, and intraocular pressure.12,13 On the other hand, no benefit in the prevention of rebleeding has been proven for hospitalization with enforced bed rest or for bilateral patching to reduce eye movement.14
Several pharmacologic measures have been used to prevent rebleeding. Corticosteroids, both topical (prednisolone acetate 1% q.i.d.) and systemic (prednisone 0.5–1.0 mg/kg per day), reduce iritis and ciliary spasm, increase patient comfort, and theoretically stabilize the clot formation, thereby decreasing the rate of rebleed.15 Clinical trials have demonstrated the efficacy of oral epsilon-aminocaproic acid, both systemic (Amicar, Lederle Laboratories, Pearl River NY, given 50 mg/kg every 4 hours for 5 days) and topical (Caprogel, ISTA Pharmaceuticals Irvine, CA, given every 6 hours for 5 days) and tranexamic acid (Cyklokapron, Pfizer, New York, NY) in reducing the incidence of secondary bleeding.16,17 These antifibrinolytic agents reduce clot degradation from traumatized blood vessels by inhibiting the conversion of plasminogen to plasmin, the protein responsible for clot breakdown.
A recent review and meta-analysis examined the use of antifibrinolytic agents, corticosteroids, cycloplegics, miotics, aspirin, conjugated estrogens, unilateral or bilateral patching, elevation of the head, and bed rest. No intervention had a significant effect on visual acuity, whether measured at ≤2 weeks after the trauma or at later time points. All antifibrinolytic agents significantly reduced the incidence of recurrent hemorrhage. The evidence to support an associated reduction in the risk of complications from secondary hemorrhage (i.e., corneal blood staining, peripheral anterior synechiae, elevated intraocular pressure, and development of optic atrophy) by antifibrinolytics was limited by the small number of these events. Use of aminocaproic acid was associated with increased nausea, vomiting, and other adverse events compared with placebo. There was no difference in the number of adverse events with the use of systemic versus topical aminocaproic acid or with standard versus lower drug dose.18
In most cases the hyphema improves with medical management. However, empiric criteria for surgical evaluation have been developed13,19,20: (1) intractably elevated intraocular pressure (IOP) despite medical therapy (>60 mmHg for 2 days in sickle-negative patients; or >24 mmHg for more than 1 day in sickle patients); (2) total hyphema for more than 5 days with IOP >25 mmHg; (3) corneal bloodstaining; (4) persistence of hyphema occupying at least one-half of the anterior chamber volume. Several surgical techniques have been suggested to manage this problem, including paracentesis, anterior chamber washout with a one-needle irrigation or irrigation–aspiration technique,21 washout with a two-needle technique, clot evacuation with a forceps or cryoprobe through a large limbal incision,22 or clot evacuation associated with a trabeculectomy filtering operation.23
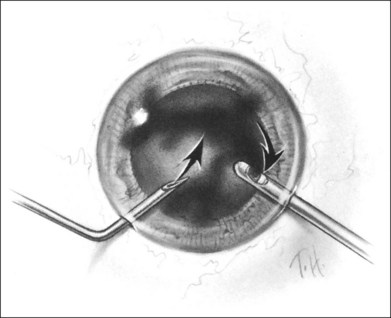
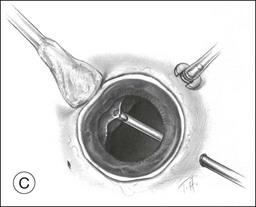
A two-instrument bimanual technique using vitrectomy instrumentation inserted through limbal incisions permits controlled removal of anterior chamber hemorrhage (Fig. 110.1).24,25 The surgeon inserts through one incision a blunt infusion cannula (20-gauge (G), or smaller) or a bent needle (typically 23G) connected to balanced salt solution and a vitrectomy instrument through the other. Using both the aspiration and cutting functions of the instrument, as much of the clot and free blood is removed as possible. Care is taken to avoid damaging the crystalline lens and corneal endothelium. It is not uncommon to leave some residual blood at the end of the procedure. The incisions are closed with “X-type” sutures using 9–0 or 10–0 monofilament nylon with knots buried in the incision. However, this technique may also now be performed with the newer small-gauge vitrectomy instruments. By using a 23G or 25G approach, self-sealing incisions may be possible.
Lens subluxation and dislocation
Blunt trauma can lead to either subluxation or dislocation of the crystalline lens. In lens subluxation, zonular filaments are broken, and the lens is no longer held securely in place but remains in the pupillary aperture. Lens dislocation occurs following complete disruption of the zonular filaments and displacement of the lens from the pupil. Trauma is the leading cause of lens dislocation.26
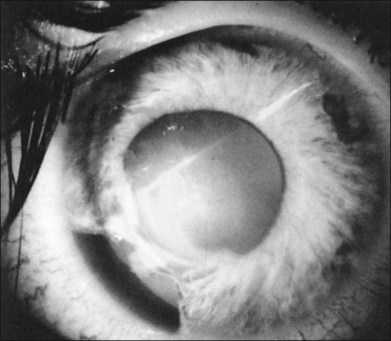
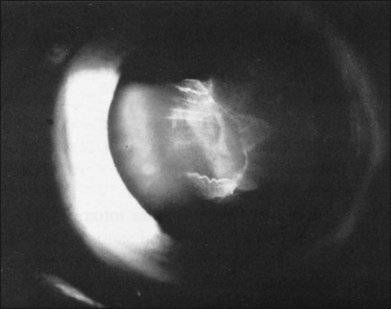
In the evaluation of a patient with a cataractous lens after blunt trauma, it is important to remember that the lens may be subluxated. During the slit-lamp examination, evidence of iridodonesis or phacodonesis must be sought. This usually is more apparent in the undilated state, when the remaining intact zonular filaments are under less tension. Phacodonesis can be detected by having the patient look quickly from side to side or up and down, or by jarring the slit-lamp table. If an associated iridodialysis is present, the absence of zonular fibers can be identified by looking through the area of absent iris (Fig. 110.2). In addition, vitreous sometimes can be identified extending around the lens, either through the pupil or through the iridodialysis. Presence of vitreous in the anterior chamber is proof of zonular rupture and probable lens subluxation.
For cases with minimal lens instability, the traditional phacoemulsification techniques through a corneal limbal or scleral tunnel incision may be used, with a thorough hydrodissection and care to minimize the stress on the zonule and vitreous during all steps of the procedure. A capsular support device may increase the safety and ease of phacoemulsification by stabilizing the capsular bag. Alternatively, a larger capsulorrhexis may be used, and the lens may be prolapsed into the anterior chamber for phacoemulsification. Of great concern is vitreous prolapse through the cataract wound, and if vitreous is pulled on, traction transmitted to the vitreous base can cause retinal tears, giant retinal tear, and retinal detachment.27 Consequently, the surgeon must recognize that vitreous prolapse through the wound is probable and be prepared for its management.
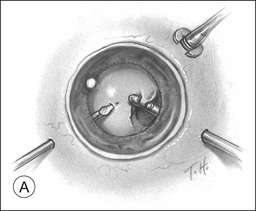
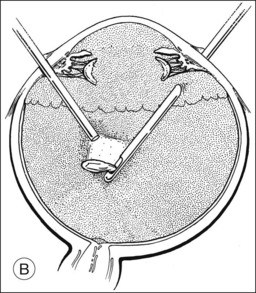
A pars plana approach using vitreoretinal instrumentation avoids many of the risks associated with limbal extraction (Fig. 110.3). Using a two- or three-port system, a myringotomy blade or a bent 21G or 23G butterfly intravenous needle connected to irrigation is inserted through the pars plana into the lens for fixation, and either a vitrectomy or a phacofragmentation instrument is inserted through the opposite pars plana to digest the lens. If the lens is soft, as in children and young adults (the most frequent victims of trauma), the entire lensectomy can be performed with the vitreous cutter. Vitreous around the lens can be managed with high-speed cutting to prevent traction on the vitreous base. Should the lens nucleus be harder and require phacofragmentation, care should be taken to perform either a limited vitrectomy around the lens before introduction of the phacofragmatome or to use the phacofragmatome only when inside the lens capsule. This avoids unintentional aspiration pressure on the vitreous that could lead to retinal damage.
Pars plana vitrectomy and pars plana lensectomy (PPV/PPL) with implantation of IOL can be used for cases of subluxated traumatic cataract with extensive zonular damage. Kazemi et al. reported on nine such eyes, in which an anterior chamber IOL was placed. Complications included retinal detachment in one patient, transient limbal wound leak in one patient, and cystoid macular edema in three patients. All of the complications were managed successfully. Best-corrected visual acuity of better than 20/40 was achieved in six of the nine eyes, and the etiology of poor visual acuity in the remaining three eyes was attributed to the original injury.28
Kodjikian et al. reported on nine consecutive cases of traumatic cataract with zonular dialysis of at least 180° and visual acuity of 20/100 or worse. Following PPV/PPL and implantation of an iris-fixated Artisan IOL, all patients achieved best-corrected visual acuity of 20/30 or better. Postoperative complications included one instance each of iris incarceration into the corneal wound and anterior uveitis, which was medically managed.29 Other authors have reported similar results using PPV/PPL and sutured sulcus IOLs.30 A consistent finding in these studies is that the primary cause of poor visual acuity postoperatively is posterior segment pathology related to the original injury.31
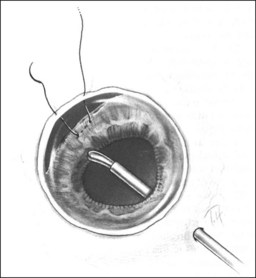
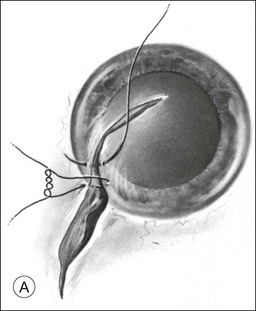
Should an iridodialysis be present, it can be repaired at the end of the case using a 10–0 Prolene suture introduced through the cornea in a modification of the technique described by McCannel32 to fixate IOLs to the pars plana (Fig. 110.4). At the end of the procedure, the peripheral retina should be carefully inspected with scleral depression to identify retinal breaks, dialyses, or areas of retinal detachment. Inspection can be performed either with binocular indirect ophthalmoscopy or through the microscope equipped with wide-field viewing. Should retinal breaks or detachment be present, they should be treated immediately with retinopexy and, if necessary, scleral buckling and endotamponade.
Vitreous hemorrhage
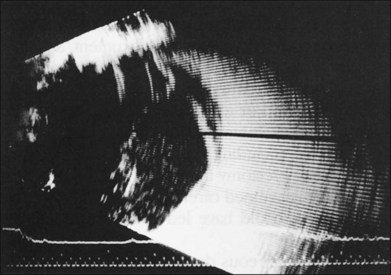
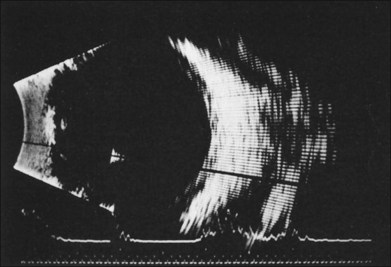
If vitreous hemorrhage obscures examination of the retina, ultrasonography plays a critical role in decision-making. With combined contact A-scan and B-scan techniques, many details of the posterior segment can be determined, including presence of retinal detachment, posterior vitreous detachment, occult scleral rupture, hemorrhagic or serous choroidal detachment, and giant retinal tear (Fig. 110.5). For example, if ultrasound demonstrates vitreous strands emanating from an equatorial location, an occult scleral rupture with vitreous incarcerated in the wound can be inferred, requiring conjunctival peritomy and exploration.
The visual prognosis for eyes with vitreous hemorrhage associated with nonpenetrating trauma depends on associated macular damage (from choroidal rupture, traumatic macular hole, Berlin’s edema, or macular contusion), retinal dysfunction from associated retinal detachment, and occlusional amblyopia in young children. In a study of 33 eyes with severe vitreous hemorrhage associated with closed-globe injury, best-corrected visual acuity following resolution and/or treatment of hemorrhage was <20/200 in 54%. The most common cause of poor visual outcome was macular scar. Poor prognostic factors included presenting visual acuity of light perception or worse, hyphema, traumatic cataract, and age 55 years or younger.33
“Commotio retinae”, avulsion of the vitreous base, and retinal tears
Blunt trauma can damage the retina in many ways, ranging from retinal edema to retinal detachment. “Commotio retinae” is commonly seen after a contusive injury to the globe and appears ophthalmoscopically as retinal whitening. Edema involving the macula (termed Berlin’s edema) can impart an appearance similar to a cherry-red spot. The reasons for this retinal opacification are disputed, with some attributing it to extracellular edema34; however, experimental and histopathologic studies have shown that disruption of the photoreceptor cell outer segment and damage to the retinal pigment epithelium accounts for the retinal whitening.35,36 Vision can be markedly decreased with commotio retinae, but vision most often improves as the swelling resolves over a 3–4-week period. However, the macula can develop an atrophic appearance with granular hyperpigmentation associated with decreased vision. In addition, the cystoid areas can coalesce to form a large cyst, which can lead to macular hole.
Blunt trauma can also lead to large areas of irregular, jagged retinal holes. These breaks have been observed almost immediately after the contusive injury and are believed to be the result of mechanical disruption and fragmentation of the retina. Similar-appearing breaks have been produced in an experimental model of concussive injury to the globe.37 This syndrome has been given a variety of names, including “acute retinal necrosis”38 and “chorioretinitis sclopetaria.”39 We prefer the latter term to avoid confusion with the acute retinal necrosis inflammatory syndrome of herpetic viral origin.
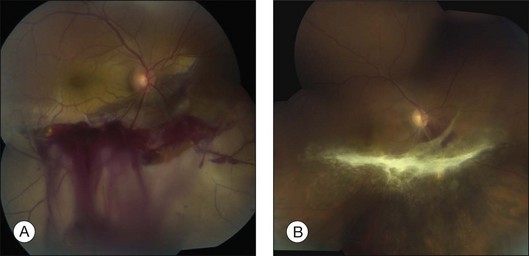
In eyes with chorioretinitis sclopetaria, it is tempting to intervene in some way because of the frightening size and often posterior location of the retinal breaks (Fig. 110.6). However, the retina rarely detaches in this situation, presumably because of the inflammation at the edges of the necrotic retina that leads to a firm chorioretinal adhesion. If a retinal detachment occurs, it is usually from another site. Therefore, it is not necessary to perform prophylactic cryopexy or photocoagulation. Patients with this syndrome often have poor visual acuity because of the effects of the severe blunt force on the macula.
Blunt trauma also can cause retinal breaks by transmission of the force to the vitreous, leading to acute severe vitreoretinal traction. Rapid displacement of the vitreous can tear the retina in various ways, including retinal dialysis with or without avulsion of the vitreous base, operculated retinal tear, macular hole, and horseshoe-shaped retinal tears at the posterior margin of the vitreous base, at the edge of a meridional fold, or at the equator.40,41 A study by Cox37 concluded that the different forms of retinal abnormalities are a result of the point of impact of blunt trauma.
The most common retinal break from trauma is a retinal dialysis. This is seen most commonly in the superonasal and inferotemporal quadrants because of blunt trauma frequently striking the globe inferotemporally.42,43 The blunt force causes equatorial stretching of the globe, leading to traction on the vitreous base at the site of impact and diametrically opposite. Cox et al.40 reviewed 160 patients with retinal detachment caused by contusion. Retinal dialyses larger than one oral bay were found to be the most common retinal break.
Retinal detachment and macular hole
In treating a retinal detachment in a patient with a history of ocular contusion, the surgeon must remember the types of retinal breaks that are most common in this setting, specifically retinal dialysis. Most traumatic retinal detachments can be treated with conventional scleral buckling techniques. Because of the potential for retinal damage 180° from the impact site, an encircling element is recommended. The selection of chorioretinal adhesive procedure and determination of the need to drain subretinal fluid should be made by the surgeon using the same principles as for nontraumatic detachments. Johnston44 reported 77 eyes with retinal breaks after contusive injury; 65 developed rhegmatogenous detachment. Surgical treatment restored or maintained retinal apposition in 96% of the eyes.
In rare cases, blunt trauma may cause a rhegmatogenous retinal detachment from a giant retinal tear or a macular hole. Although these detachments have been repaired successfully in the past with a variety of scleral buckling techniques, modern vitreoretinal surgery has significantly increased the rate of anatomic reattachment. A giant retinal tear is defined as a retinal tear of greater than 3 clock-hours. Vitreous surgery allows mobilization of the flap by excising adherent vitreous. With the aid of surgical adjuvants, such as perfluorocarbon liquids, and current surgical techniques (as described in Chapter 105, Optimal procedures for retinal detachment repair), the rate of successful retinal reattachment for such cases has greatly improved.
Traumatic macular hole formation is uncommon. Like idiopathic macular holes, traumatic holes can be rehabilitated with vitrectomy. However, a significant number of traumatic macular holes may close spontaneously. In a consecutive series of 18 such cases, spontaneous closure and accompanying visual improvement were seen in eight (44%) and occurred between 1 week and 4 months following injury.45 Although the visual outcome may be limited by associated macular damage (such as a macular choroidal rupture), there are encouraging reports of successful anatomic and functional outcomes with vitrectomy. Since patients with traumatic macular hole are typically younger and phakic, we elect to use long-acting gas endotamponade with 14% perfluoropropane to optimize the chance of hole closure. It has been suggested that surgical adjuvants such as autologous plasmin enzyme46 or removal of internal limiting membrane47,48 may improve the rate of anatomic success. Improvement in visual acuity of at least 2 Snellen lines has been reported in 69–94% of cases.47–49
Open-globe injuries
Preoperative evaluation
It is important to obtain a history from any patient being evaluated for a possible eye injury. Although many trauma patients will be poor historians because of associated shock, intoxication, or neurologic problems, the history can give important clues about the ocular damage. For instance, if a patient felt something flying into his or her eye while hammering a nail but did not note any visual disturbance, the physician must carefully search for evidence of an intraocular foreign body (IOFB), even if there is no evidence of obvious ocular laceration. Furthermore, since many of the medical malpractice cases brought against vitreoretinal specialists involve trauma, careful documentation of the details surrounding the injury may prove valuable in case of litigation later on.50
Careful evaluation is important not only for appropriate management, but can also be helpful in prognostication of visual outcome, when used according to the ocular trauma score (OTS). The OTS was developed by Kuhn et al. following review of over 2500 registered cases of ocular trauma. The OTS provides a single probability estimate that an eye trauma patient will obtain visual acuity within a specific visual range by 6 months after injury.51 In the OTS, a numerical score is derived by adding or subtracting points on the basis of initial visual acuity, presence of rupture, endophthalmitis, globe perforation, retinal detachment, or afferent pupillary defect. Higher OTS scores are associated with a better visual prognosis.51 The OTS can thus be used as an aid in patient counseling and can assist in identifying severe ocular injuries with a poor visual prognosis.
The initial step in examination is determination of visual acuity (VA) with either a near vision card or a Snellen acuity chart. When a retrospective review of penetrating injury patients was subjected to multivariate analysis, VA was the most important determinant of final visual outcome. Patients with initial VA of 5/200 or better had a 28 times greater chance of salvaging acuity at this level than those with vision worse than 5/200.52 Two subsequent prospective studies of penetrating injuries have confirmed this finding. In the first of these series, 94% of patients with initial VA of 20/200 or better achieved a final VA of 20/200 or better.53 In the second series, multivariate analysis revealed that presenting visual acuity of 5/200 or worse was the most important factor contributing to poor visual outcome; statistically insignificant factors were time since injury, cataract, and presence of IOFB.54 In one review of 167 open-globe injuries, only 3% of eyes presenting with vision better than light perception ultimately underwent enucleation, as compared with 39% of eyes presenting with light perception and 89% of eyes presenting with no light perception.55
Others have demonstrated that the presence or absence of an afferent pupillary defect (APD) is a strong predictor of outcome.56,57 A multivariate analysis of 240 traumatized eyes found 69% of eyes without an APD obtained a final VA of 20/200 or better, as compared with 34% of eyes with an APD (P < 0.00001).56
On rare occasions, the surgeon may contemplate primary enucleation when encountering a totally disorganized globe with prolapse of intraocular contents, including retina. In these cases it is important to determine carefully whether the patient can perceive light. This is best done with the uninjured eye covered, with the indirect ophthalmoscope light at maximal intensity, and with the light held at sufficient distance so that the patient cannot perceive the heat of the ophthalmoscope light. Unfortunately, since many patients with injuries of this severity often are either intoxicated or have other injuries affecting their mental status, determination of light perception is questionable. Additionally, there have been rare reports of eyes with preoperative measurement of no light perception achieving visual acuity of light perception or better following reconstructive ocular surgery.51,58 For these reasons, primary enucleation is rarely performed.
Occasionally, a scleral rupture can be occult (hidden under conjunctiva, Tenon’s capsule, or rectus muscles). Diffuse chemosis or subconjunctival hemorrhage suggests the presence of occult rupture. An open globe often has low IOP, but normal or elevated IOP does not rule out the possibility of a rupture. On occasion, slit-lamp biomicroscopy of the anterior vitreous can be useful, demonstrating vitreous strands directed toward a hidden scleral rupture site (Fig. 110.7). Similarly, ultrasonography can detect this finding. Computed tomography (CT) has also been reported to show occult rupture, with flattening of the posterior contour of the sclera (the “flat tire” sign).59
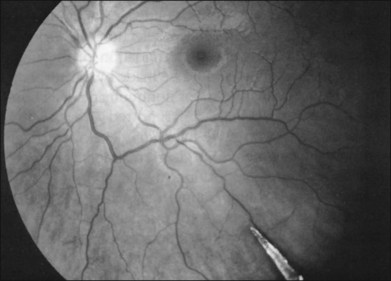
Confirming the presence or absence of a retained IOFB is critical to determining management of a patient with trauma. In many cases, the IOFB can be identified by slit-lamp biomicroscopy or indirect ophthalmoscopy (Fig. 110.8). However, if the ocular media are clouded by corneal damage, hyphema, cataract, or vitreous hemorrhage, ancillary techniques are necessary. For many years, the standard technique of localizing foreign bodies involved special applications of conventional radiographic imaging; however, these methods often were inaccurate and cumbersome, could cause iatrogenic damage to the globe, and were useless in cases involving radiolucent foreign bodies. Consequently, radiographic localization of IOFBs has been replaced by ultrasonography and CT. Ultrasonography can accurately localize foreign bodies, particularly with a systematic approach providing transverse and longitudinal views in all meridians (Fig. 110.9). When the globe is open, however, the resolution of ultrasound may be limited, as the examination often must be performed gently through closed lids. The usefulness of echography is also limited by shadowing caused by highly reflective surfaces such as air, reverberation artifacts created by some IOFBs, and by the user’s skill and familiarity with ocular pathology.60
CT has emerged as the imaging technique of choice in the evaluation of IOFBs. It is a noninvasive technique requiring minimal patient cooperation and can be used to image radiolucent and radiopaque foreign bodies (Fig. 110.10). By assembling many consecutive orbital slices that demonstrate the ocular anatomy and by using computer-generated reconstructions in three dimensions, the presence and location of an IOFB can be determined. In an experimental study to determine the size of CT cuts required to detect small IOFBs, Dass et al. concluded that with modern spiral CT scanning both 3 mm and 1 mm cuts were effective (100% sensitivity) in detecting a 0.5 mm metallic, glass, or stone IOFB.61 The absorption characteristics of the foreign body are quantitated in Hounsfield units and can be compared with the absorptions of various materials already determined experimentally. Zinreich et al.62 have shown that wood is the least dense of the nonmetallic foreign bodies, followed by plastic and then glass. Unfortunately, all metallic foreign bodies have the same absorptions and are impossible to differentiate. CT scanning does have limitations, in that metallic IOFBs often create significant scattering artifact that may obscure their precise location. This can be particularly bothersome when attempting to determine whether a foreign body is intraretinal or intrascleral. In addition, identifying some of the lower-density foreign bodies, such as wood, may be difficult with CT scanning.63
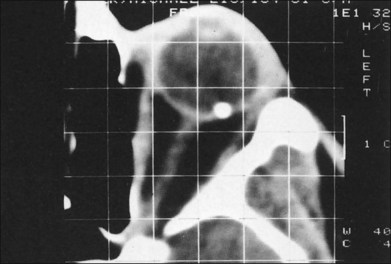
Fig. 110.10 This computed tomography (CT) scan demonstrates an intraretinal shotgun pellet in the patient’s left eye.
Magnetic resonance imaging (MRI) has proved useful for imaging intraocular tumors and other lesions. However, the magnetic fields and heat generated during MRI scanning preclude examination of patients with potential intraocular or intraorbital metallic foreign bodies. Investigators have shown that ferromagnetic foreign bodies implanted in animal eyes can move 7–8 mm in the suprachoroidal space and up to 10 mm in the vitreous space when subjected to the torsional forces of MRI.64 Furthermore, retained intraocular ferromagnetic foreign bodies cause surrounding image distortion, making the globe itself difficult to visualize. Image distortion can similarly occur from the paramagnetic effects in granite, solder, and mascara. Another disadvantage of MRI is its inability to image bone. MRI’s principal advantage over CT is that it can accurately and safely detect wood and plastic IOFBs.
Repair of laceration
A watertight closure can be difficult to achieve with stellate lacerations. In some cases the wound leaks even with multiple sutures criss-crossing the laceration. A purse-string closure may help in these cases. The suture is placed in a circle through the various portions of the cornea involved in the laceration and then tied to itself with the knot buried in the laceration. If this technique does not work, corneal tissue may have been lost. In these situations, use of cyanoacrylate tissue adhesive can seal the leakage site.65 When there is too much tissue loss for glue to work, a corneal patch graft can be used.
When iris is incarcerated in the corneal wound, it can be reposited in most cases. We advocate excision of uveal tissue only if it looks extremely necrotic, usually having been externalized for more than 24 hours. Reposition of incarcerated iris usually can be accomplished with introduction of an iris or cyclodialysis spatula or a 27G cannula connected to viscoelastic through a limbal stab incision made about 90° away from the laceration. This maneuver is best done after the corneal integrity has been partially restored by the first few sutures and the anterior chamber has been reformed with balanced salt solution, air, or hyaluronic acid.66 When there is positive pressure or in the presence of a hyphema, the surgeon may be helped by having the assistant reposit incarcerated iris while the surgeon places the suture.
In some cases, the scleral laceration extends posterior to the ora serrata and may have an associated retinal laceration. Because of associated vitreous hemorrhage, it is rare to be able to identify a retinal laceration at the time of primary repair. Cryopexy should not be performed prophylactically unless a specific retinal tear can be visualized, since cryotherapy may cause excessive inflammation that may promote the development of later traction retinal detachment.67
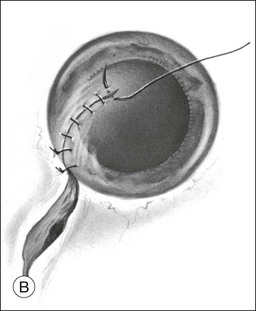
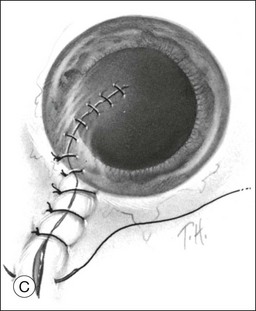
In cases of lacerations involving both the cornea and sclera, the extent of the laceration must be determined first. In an effort to restore structural integrity, the limbus is the best place to start, as it provides a recognizable anatomic landmark. Limbal wounds are closed using 9–0 nylon sutures (Fig. 110.11). After the limbal portion of the laceration is closed, it is easier to repair the corneal laceration first, then the scleral laceration. The same techniques and principles described for lacerations limited to the cornea or sclera should be employed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree