Chapter 35 Surgery for Locally Advanced Thyroid Cancer
Trachea
![]() Please go to expertconsult.com to view related video Tracheal Resection for Locally Advanced Carcinoma of the Thyroid Gland.
Please go to expertconsult.com to view related video Tracheal Resection for Locally Advanced Carcinoma of the Thyroid Gland.
Introduction
Thyroid cancer incidence appears to be rising in the United States, where approximately 45,000 new patients are expected to be diagnosed in 2011.1 Thyroid cancer rates have quadrupled since the 1980s, although the majority of this increase appears to be directly related to early diagnosis from incidental findings of thyroid nodules and microcarcinomas during routine imaging studies or clinical examinations.2 It is likely that the majority of these tumors would have remained silent and thus not recognized for some time. Most incidentalomas noted today are microcarcinomas, and most of these patients do remarkably well—the overall survival in this group of patients exceeds 98%. There is also a clear trend showing a rising incidence of thyroid cancer in women. Interestingly, mortality rates from thyroid cancer have not changed since the 1980s. Approximately 1500 patients succumb to this disease yearly in the United States.1 The majority of these deaths are related to anaplastic thyroid cancer, medullary thyroid cancer, or a high-grade and high-risk differentiated thyroid carcinoma, particularly in elderly individuals. The general concepts on management of thyroid cancer have essentially remained the same since the 1960s. This includes thyroid lobectomy or total thyroidectomy, with or without radioactive iodine ablation and thyroid suppression. Our recent understanding of thyroid cancer and its prognostic factors have generated considerable debate about the necessary extent of thyroidectomy, whether thyroid lobectomy or total thyroidectomy.3–9 However, it is important to recognize that in the low-risk thyroid cancer patients, the prognosis and the outcome are so good that this controversy regarding the extent of thyroidectomy will continue to be an academic debate.
Thyroid cancer is a unique human neoplasm, quite different from other human cancers such as breast, lung, or pancreatic cancers. The majority of patients with thyroid cancer do remarkably well, and the small minority are well recognized with high-risk thyroid cancer right from the outset or from the time of recurrent thyroid cancer.7 Our understanding of thyroid cancer has improved remarkably since the 1980s with detailed analysis of prognostic factors and risk group analysis (see chapter 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer). The prognostic factors in well-differentiated thyroid cancer have been identified as age, grade of the tumor, extrathyroid extension, size of the tumor, and distant metastases. Other factors such as gender, lymph node metastases and histology have a minimal impact on long-term outcome. Risk group stratification is extremely important to provide the best optimal treatment, and at Memorial Sloan-Kettering Cancer Center, patients are divided into low-, intermediate-, and high-risk groups (see Chapters 18, Papillary Thyroid Cancer, 19, Papillary Thyroid Microcarcinoma, 20, Follicular Thyroid Cancer, and 21, Dynamic Risk Group Analysis for Differentiated Thyroid Cancer).6,9 In the low-risk group, survival is higher than 98% and the role of adjuvant therapy is minimal. In the intermediate- and high-risk groups, appropriate and adequate surgery includes excision of all gross tumor, followed by radioactive iodine ablation in selected patients (see Chapters 50, Postoperative Management of Differentiated Thyroid Cancer, and 51, Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer). The role of external radiation therapy remains unclear at this time, except in high-risk patients or where surgical resection was not satisfactory (see Chapter 52, External Beam Radiotherapy for Thyroid Malignancy). The prognostic factors are well known and described by the Mayo Clinic (MACIS), Lahey Clinic (AMES), and Memorial Sloan-Kettering Cancer Center (GAMES). There are many publications regarding prognostic factors in thyroid cancer, and a consensus has been established on risk group stratification and prognostic factors all around the world.7,8
Biology of Thyroid Cancer
Thyroid cancer continues to have the best cancer treatment outcome. Most patients continue to enjoy a normal life after appropriate treatment of well-differentiated thyroid carcinoma. However, thyroid disease does include papillary and follicular thyroid cancers, which have the best outcomes, as well as anaplastic thyroid carcinoma, with the worst outcome. It is also interesting to note that thyroid cancer is a continuum of diseases. It appears that disease progresses from papillary to tall cell, to insular, to poorly differentiated thyroid carcinoma, and finally to the cancer with the most lethal outcome, anaplastic thyroid carcinoma. Molecular biology has clearly demonstrated the molecular changes at each level, from the normal follicular cell of the thyroid to the development of follicular adenoma, follicular carcinoma, aggressive thyroid cancer, and anaplastic thyroid cancer. Understanding these molecular changes is crucial to recognize the clinical behavior of thyroid cancer, the progression of thyroid cancer from well differentiated to anaplastic thyroid carcinoma, and the development of targeted therapy based on molecular targets (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia). This progression model has also helped us recognize that radioactive iodine ablation works best in well-differentiated thyroid carcinoma, although this treatment has little impact on high-grade tumors. Since the 1990s, we have also documented the importance of the positron emission tomography (PET) scan in the follow-up of patients with poorly differentiated thyroid carcinoma, where PET positivity is high.10 This is important in the follow-up of locally aggressive thyroid cancer or high-risk thyroid cancer for recurrent disease in the thyroid bed, the neck, or distant regions.
Our interest in the molecular biology of thyroid cancer has helped us use molecular analysis in fine-needle aspiration biopsy samples to determine the aggressiveness of thyroid cancer even prior to initial surgical intervention. The BRAF mutation aids in differentiating aggressive thyroid cancers. Other marker studies are also available; however, these studies do not appear to have major clinical impact in the everyday practice of management of thyroid cancer to date.11,12 In the future, markers are expected to have a major impact on the understanding of the aggressiveness of thyroid cancer and direct the treatment philosophy based on the molecular differentiation of thyroid cancer. Many institutions are using various targeted treatments in advanced thyroid cancer or in recurrent thyroid cancer where surgical intervention is unlikely to help the patient or gross tumor is left behind after appropriate surgery. However, thyroid cancer continues to be a surgical disease, and the completeness of resection is still the most important prognostic factor for long-term control and best outcome.
Locally Advanced Thyroid Cancer
Most thyroid cancers present in an early stage, with well-localized intrathyroidal cancers. However, approximately 10% to 15% of thyroid tumors present with extrathyroidal extension.13–17 The most common structures involved in this process include the strap muscles (53%), the recurrent laryngeal nerve (47%), the trachea (30%), the esophagus (21%), and the larynx (12%).14 The lateral neck structures, such as soft tissues of the neck and great vessels, are rarely involved in locally aggressive thyroid cancer. This may occur, however, with extension of disease into the lateral compartment, either from the primary thyroid cancer or from nodal disease. The presence of extrathyroidal extension has been recognized as a poor prognostic indicator in various prognostic systems and risk group analyses. Long-term survival in this group declines from 90% to 57%. Extrathyroidal extension can be divided into anterior and posterior.15 Anterior extrathyroidal extension involving the strap muscles can be easily resected and generally does not have a major impact on long-term prognosis. Posterior extrathyroidal extension involving the tracheal wall, recurrent laryngeal nerve, or the esophagus, however, can be an operative challenge for the surgeon and also has a high incidence of local recurrence. This is more common in elderly individuals with high-grade histology or long-standing thyroid cancer. Local control of recurrent thyroid cancer in the central compartment is a difficult problem, because sound oncologic resection does involve consideration of the trachea, recurrent laryngeal nerve, and esophagus. In this chapter we mainly discuss the management of the trachea from involvement with extrathyroidal extension of the thyroid cancer. The other issues related to laryngeal involvement have already been addressed in a separate chapter (see Chapter 33, Surgical Anatomy and Monitoring of the Recurrent Laryngeal Nerve). Locally aggressive thyroid cancer, especially with involvement of the trachea, can be a surgical challenge to a thyroid surgeon to provide resection of all gross tumor (R0), to facilitate long-term follow-up with radioactive iodine ablation and external radiation therapy in selected individuals.18–26 Tracheal invasion can be an indicator of more aggressive tumors with aggressive histology and a greater risk of mortality from thyroid cancer. Uncontrolled local recurrence can be a major surgical challenge both for local control, involvement of the recurrent laryngeal nerve either unilaterally or bilaterally, involvement of the trachea, and intratracheal hemorrhage or airway compromise leading to ultimate demise. The most important aspect of the management of locally aggressive thyroid cancer is a complete preoperative evaluation of the extent of the disease, satisfactory surgical resection of all gross tumor, and the use of adjuvant treatment modalities. These clinical scenarios for locally aggressive thyroid cancer are generally more common in elderly individuals, a reflection of the biology of the disease.27
Initial Evaluation
Preoperative evaluation begins with a good history and physical examination, which is the cornerstone of initial evaluation of the extent of disease. Long-standing tumors, a rapid increase in the size of the tumor, or fixity in the central compartment are indicators of locally aggressive thyroid cancer. Respiratory symptoms such as dyspnea, hemoptysis, and hoarseness of voice cannot be neglected in the initial evaluation. Grillo and his colleagues reported a study of 82 patients who underwent segmental tracheal resection with the presenting symptoms of vocal cord paralysis (35%), dyspnea and wheezing (30%), and hemoptysis (24%). However, when tumors invaded the trachea superficially or were only adherent to the tracheal wall, many were totally asymptomatic. Preoperative evaluation of the vocal cords is extremely important.27,28 Whether this is performed with a mirror examination or a fiberoptic flexible laryngoscopy depends on the choice of the individual surgeon and that surgeon’s expertise. Fiberoptic laryngoscopy is helpful to document the intralaryngeal evaluation including vocal cord paralysis, status of the opposite vocal cord, and the adequacy of the airway. Most of the time, one can visualize the subglottic area and the upper tracheal region. This will help guide the operating surgeon regarding the need for cross-sectional imaging including CT scan or MRI.15,16 The ultrasound commonly performed in the preoperative evaluation of the thyroid is more helpful for intrathyroidal disease than it is for extrathyroidal extension. It may be of help in detecting tracheal invasion in 40% to 90% of the individuals. Cross-sectional imaging is generally more important. Clearly, ultrasound is more operator dependent and the sonographer’s experience is important in the interpretation of extrathyroidal extension. A preoperative fine-needle aspiration biopsy can help to confirm the diagnosis of malignancy, and in select cases the cytologist may be able to advise about the aggressiveness of the thyroid cancers (such as insular or poorly differentiated thyroid tumors). The preoperative evaluation of vocal cord function is also important to make certain critical decisions in the operating room about the contralateral thyroid lobe.28 If one of the patient’s recurrent laryngeal nerves is paralyzed, any injury to the other may lead to airway-related issues from bilateral vocal cord dysfunction. This may be a situation where nerve monitoring may be helpful, even though its use for routine nerve monitoring in standard thyroidectomy is debated. Cross-sectional imaging is best performed with a computed tomography (CT) scan. Generally a CT scan under these circumstances should be performed with contrast for better definition of the extent of the tumor and vascular structures. There is always a debate about the use of contrast during the CT scan, as it will interfere with postoperative radioactive iodine ablation. However, detailed evaluation of the extent of the disease is essential prior to surgery, and a slight delay in treating these patients with radioactive iodine will not have a major impact on their treatment (see Chapter 14, Preoperative Radiographic Mapping of Nodal Disease for Papillary Thyroid Carcinoma).28 An imaging study can help to evaluate the extent of disease and whether there is tumor involvement of the trachea, or irregularity of the tracheal mucosa or tracheal wall with the involvement of the tracheal rings. A thin-cut CT scan may help to evaluate the submucosal area and if there is any compromise of the tracheal lumen.
Preoperative medical evaluation is critical for comorbidities, such as pulmonary issues, to appreciate whether the patient can withstand a long surgical procedure. Pulmonary function tests may be of some help, but they are generally not likely to assist in evaluating the trachea. Preoperative esophagoscopy, tracheoscopy, and bronchoscopy will assist in evaluating the extent of disease with involvement of the trachea and whether there is any submucosal or intraluminal extension of the tumor. It is also important to evaluate the exact location of the tracheal involvement and its proximity to the cricoid cartilage or the carina. This is extremely important for deciding how much of the trachea needs to be removed and whether the subglottic area can be resected with satisfactory margins. It would be embarrassing after a radical surgical resection to have positive margins and parts of the gross tumor that were not resected. There is a finite limit to how much of the trachea can be resected when primary end-to-end anastomosis can be performed. Evaluation of the vertical extent of the disease is critical. Preoperative tracheoscopy or tracheoscopy at the time of surgery will be of great help in these situations. A magnetic resonance imaging (MRI) may be of some help, but cross-sectional imaging with a high-resolution CT scan is more useful. A PET scan may be performed in selected individuals where the tumor appears to be fairly advanced, where there is a suspicion of metastatic disease, or where surgery appears to be technically difficult or unlikely to remove all gross tumor. PET scans may also be of help in evaluating aggressive histologic variants. Patients who present with massive central compartment disease with involvement of the proximal trachea or cricoid cartilage may need to be reevaluated to determine whether they will require a laryngectomy along with resection of the upper trachea (see Chapter 34, Surgery for Locally Advanced Thyroid Cancer: Larynx). Decisions about partial or total laryngectomy along with tracheal resection are extremely critical for satisfactory surgical conduct and outcome.7 Both the patient and the family need to be prepared for these aggressive surgical interventions where morbidity is high. Appropriate preoperative speech evaluation and consultation are important, and the patient needs to understand issues pertaining to a permanent stoma if total laryngectomy is contemplated.
Staging of Tracheal Invasion
Our understanding of the patterns and staging of tracheal invasion mainly comes from the great experience of Grillo and his colleagues. Shin et al. made a significant contribution to our knowledge of the routes of invasion of the trachea and the extent of tracheal invasion.29,30 Based on the large experience of Grillo from Massachusetts General Hospital, a clinical pathologic study of papillary carcinoma with tracheal invasion and surgical resection was conducted. Grillo was a master tracheal surgeon who made enormous contributions in the field of primary tracheal tumors as well as thyroid cancers invading the trachea. Thyroid cancer can invade the surrounding structures by direct extension from the primary tumor or invasion by the paratracheal lymph nodes with metastatic tumor. The paratracheal lymph nodes are more likely to invade the trachea if they are long-standing or of high-grade histology. Based on the studies by Shin and Grillo, tracheal involvement may occur by direct proximity of the thyroid gland in the pretracheal fascia. The posterior capsule of the thyroid gland and the anterior wall of the trachea have only a thin layer of separation, and the tumor can encroach upon the perichondrium of the tracheal wall. Most of the time, unless there is direct invasion of the tracheal wall, the tumor can be easily separated from the anterior wall of the trachea in the pretracheal plane. There is a dense fibrous tissue between the tracheal cartilages and the posterior capsule of the thyroid gland. Blood vessels in this dense, fibrous tissue run perpendicular to the tracheal lumen. When the tumors are separated under these circumstances with dense fibrosis and scarring, one must be very careful not to perforate the trachea during surgical resection.
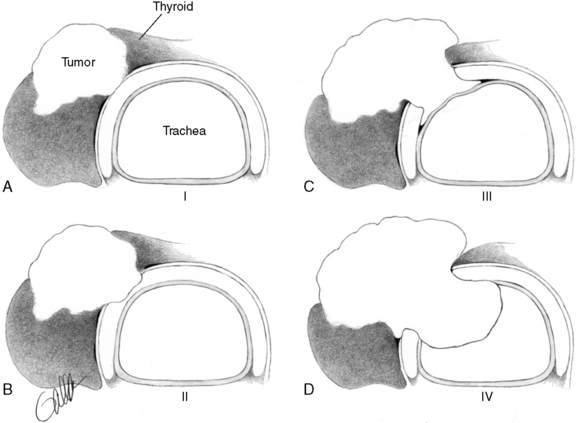
Shin and Grillo18 made a remarkable contribution in developing a staging system for tracheal invasion by thyroid tumor (Figure 35-1). They defined stage 0 disease as confined to the substance of the thyroid gland only; stage 1 disease shows invasion through the thyroid capsule and the thyroid, abutting against the external perichondrium of the trachea. Stage 2 invades between the tracheal rings, causing minor cartilage destruction. Stage 3 disease invades through the cartilage or between the cartilage rings into the lamina propria of the tracheal mucosa, and in stage 4 the disease invades the full thickness of the tracheal wall with expansion of the tracheal mucosa and ulceration of the tracheal lumen and luminal compromise. Stages 3 and 4 disease may be visible as a submucosal mass or ulcerated area during the endoscopy. Shin et al. showed the excellent prognosis in stages 1, 2, and 3, noting that survival in stage 4 disease was only 50%. This staging system helps us evaluate the extent of tumor progression on the tracheal wall or in the trachea, and it also helps us make certain determinations regarding the extent of resection.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree