Visually significant lens opacity
 Rarely, a cataract causing intraocular inflammation and/or glaucoma
Rarely, a cataract causing intraocular inflammation and/or glaucoma
Surgical Technique
 Anesthesia: topical or local
Anesthesia: topical or local
 Extracapsular method: An incision is made near the limbus, an anterior capsulotomy is performed, the lens nucleus is prolapsed out of the eye, the residual cortical material is removed, an IOL is placed, and the wound is usually sutured.
Extracapsular method: An incision is made near the limbus, an anterior capsulotomy is performed, the lens nucleus is prolapsed out of the eye, the residual cortical material is removed, an IOL is placed, and the wound is usually sutured.
 Phacoemulsification method: A small limbal or clear corneal incision is made, an anterior capsulotomy is performed, the lens nucleus is fragmented and removed with an ultrasound probe, the residual cortical material is removed, and an IOL is placed. Often, no sutures are required.
Phacoemulsification method: A small limbal or clear corneal incision is made, an anterior capsulotomy is performed, the lens nucleus is fragmented and removed with an ultrasound probe, the residual cortical material is removed, and an IOL is placed. Often, no sutures are required.
 Postoperative management: topical corticosteroid and antibiotics, often a NSAID
Postoperative management: topical corticosteroid and antibiotics, often a NSAID
Complications
 Intraoperative
Intraoperative
 Expulsive hemorrhage
Expulsive hemorrhage
 Vitreous prolapse
Vitreous prolapse
 Lens material falls into posterior segment
Lens material falls into posterior segment
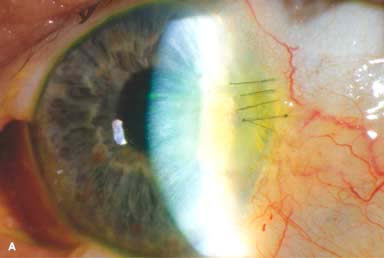
 Phacoemulsification wound burn (Fig. 10-1A)
Phacoemulsification wound burn (Fig. 10-1A)
 Postoperative
Postoperative
 Endophthalmitis
Endophthalmitis
 Wound leak, wound infection
Wound leak, wound infection
 Cystoid macular edema
Cystoid macular edema
 Glaucoma
Glaucoma
 Retinal detachment
Retinal detachment
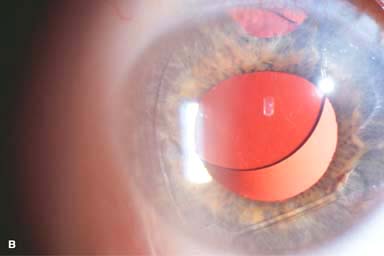
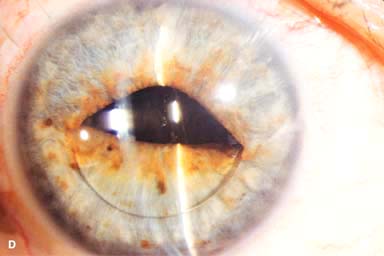
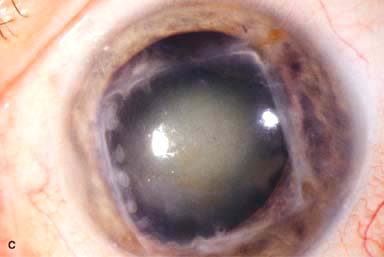
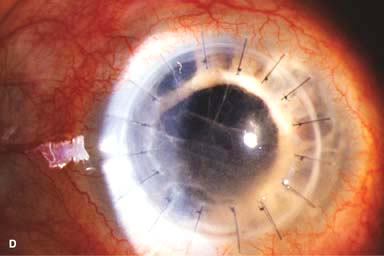
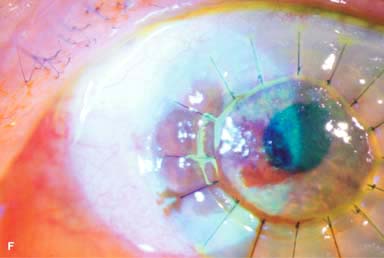
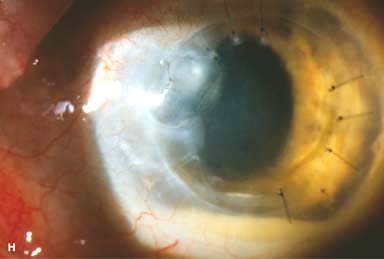
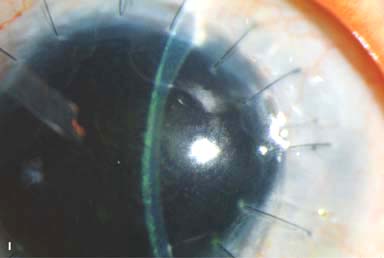
 IOL subluxation or dislocation; pupillary capture of IOL (Fig. 10-1B –D)
IOL subluxation or dislocation; pupillary capture of IOL (Fig. 10-1B –D)
 Traumatic wound rupture
Traumatic wound rupture
 Temporary or permanent corneal edema
Temporary or permanent corneal edema
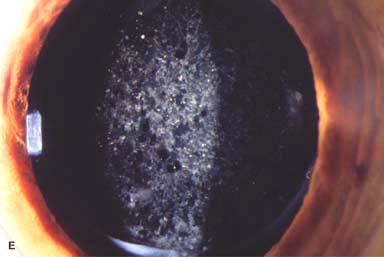
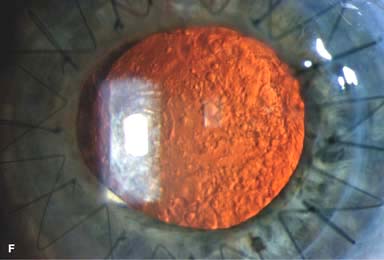
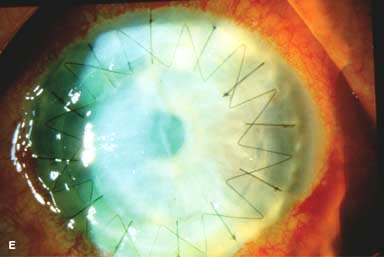
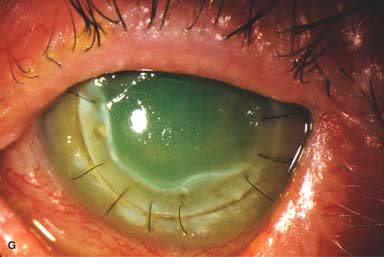
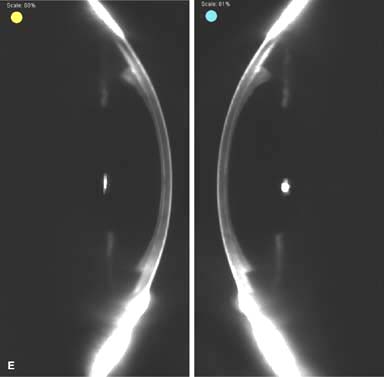
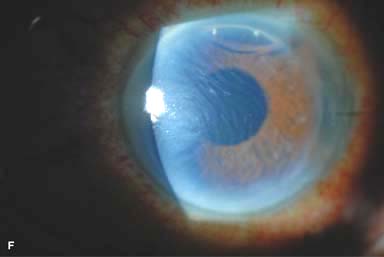
 Posterior capsular opacity (PCO), very common (Fig. 10-1E and F)
Posterior capsular opacity (PCO), very common (Fig. 10-1E and F)
Success Rate
 Excellent. PCO may require a laser capsulotomy.
Excellent. PCO may require a laser capsulotomy.
Figure 10-1. Phacoemulsification burn. A. This eye withstood a phacoemulsification burn during temporal clear corneal cataract surgery. A phacoemulsification burn during cataract surgery is caused, at least in part, by inadequate cooling of the phacoemulsification handpiece tip during use, causing a thermal burn at the wound. It can occur very quickly, within seconds. This wound required five sutures to close. There are significant corneal striae as a result of the tightness of the sutures required to keep the wound watertight. Intraocular lens subluxation. B. This anterior chamber intraocular lens is superiorly subluxed. The superior haptic has migrated through the superior peripheral iridectomy, allowing the inferior haptic to come out of the angle and intermittently contact the inferior corneal endothelium, in turn causing corneal edema. Intraocular lens subluxation. C. This posterior chamber intraocular lens has subluxed inferiorly. An oblique rent in the posterior capsule can be seen, compromising capsular support. An inferior subluxation is often termed a “sunset syndrome.” Pupillary capture. D. This posterior chamber intraocular lens is partially captured by the iris in this eye 2 weeks after cataract surgery. It was repositioned into the posterior chamber in the operating room. Posterior capsular opacity. E. A moderate posterior capsular opacity is evident in this eye several years after cataract surgery. Fibrosis of the initially clear posterior capsule often occurs after cataract surgery and can affect visual clarity. Note the arc of opacified anterior capsular rim inferiorly and the fibrotic posterior capsule centrally. When this affects visual function, treatment is with a laser capsulotomy. F. Using retroillumination off the retina, significant posterior capsule opacity is readily appreciated in this eye after combined penetrating keratoplasty and cataract surgery.

FULL-THICKNESS CORNEAL TRANSPLANTATION (PENETRATING KERATOPLASTY)
Penetrating keratoplasty involves removal of diseased host tissue and replacing it with full-thickness normal donor cornea. It is one of the most successful transplant operations today, depending on the primary corneal disease.
Indications
 Optical: to improve vision
Optical: to improve vision
 Tectonic: to restore the structural integrity of the eye
Tectonic: to restore the structural integrity of the eye
 Therapeutic: usually performed to remove infected and/or perforated corneal tissue that is not responsive to medical treatment
Therapeutic: usually performed to remove infected and/or perforated corneal tissue that is not responsive to medical treatment
 Pain: to improve pain from chronic bullous keratopathy
Pain: to improve pain from chronic bullous keratopathy
 Cosmetic: to restore a more normal appearance to the eye
Cosmetic: to restore a more normal appearance to the eye
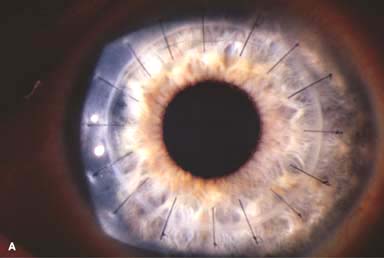
 Most common indications for penetrating keratoplasty are regrafts, keratoconus, aphakic or pseudophakic bullous keratopathy, Fuchs dystrophy and other corneal dystrophies, and infectious and traumatic corneal scarring.
Most common indications for penetrating keratoplasty are regrafts, keratoconus, aphakic or pseudophakic bullous keratopathy, Fuchs dystrophy and other corneal dystrophies, and infectious and traumatic corneal scarring.
Donor Tissue Contraindications
 Death of unknown cause
Death of unknown cause
 Death from central nervous system diseases of unknown etiology
Death from central nervous system diseases of unknown etiology
 Central nervous system infections (e.g., Creutzfeldt-Jakob disease, subacute sclerosing panencephalitis, progressive multifocal leukoencephalopathy)
Central nervous system infections (e.g., Creutzfeldt-Jakob disease, subacute sclerosing panencephalitis, progressive multifocal leukoencephalopathy)
 Systemic infections (e.g., AIDS, viral hepatitis, rabies, septicemia, cytomegalovirus infection)
Systemic infections (e.g., AIDS, viral hepatitis, rabies, septicemia, cytomegalovirus infection)
 Reye’s syndrome
Reye’s syndrome
 Lymphoma and leukemia
Lymphoma and leukemia
Unfavorable Prognostic Factors
 Severe stromal vascularization
Severe stromal vascularization
 Previous graft rejection
Previous graft rejection
 Decreased corneal sensation (e.g., herpes keratitis)
Decreased corneal sensation (e.g., herpes keratitis)
 Pediatric patients
Pediatric patients
 Active uveitis
Active uveitis
 Significant anterior synechiae
Significant anterior synechiae
 Uncontrolled glaucoma
Uncontrolled glaucoma
 Active corneal or intraocular infection
Active corneal or intraocular infection
 Severe dry eyes
Severe dry eyes
 Ocular surface inflammation, cicatrization, and keratinization
Ocular surface inflammation, cicatrization, and keratinization
 Significant limbal stem cell deficiency
Significant limbal stem cell deficiency
 Extreme thinning or irregularity at the intended graft–host junction
Extreme thinning or irregularity at the intended graft–host junction
 Eyelid disorders: ectropion, entropion, exposure
Eyelid disorders: ectropion, entropion, exposure
Surgical Technique
 Anesthesia: local or general anesthesia
Anesthesia: local or general anesthesia
 Preparation of operative eye: often, placement of a Flieringa ring helps to stabilize the globe.
Preparation of operative eye: often, placement of a Flieringa ring helps to stabilize the globe.
 Determination of graft size: Routine graft sizes vary from 7.25 to 8.5 mm.
Determination of graft size: Routine graft sizes vary from 7.25 to 8.5 mm.
 Trephination of donor cornea: typically performed using a posterior endothelial punch method, but can also be performed with a femtosecond laser. The trephine is usually 0.25 to 0.5 mm larger than that for host cornea trephination.
Trephination of donor cornea: typically performed using a posterior endothelial punch method, but can also be performed with a femtosecond laser. The trephine is usually 0.25 to 0.5 mm larger than that for host cornea trephination.
 Excision of host cornea: Trephination of host cornea can be done by using a manual trephine (e.g., Weck trephine), a vacuum trephine (e.g., Barron-Hessburg or Hanna trephine), or a femtosecond laser. A partial-thickness trephination is first carried out, the anterior chamber is then entered using a blade, and finally excision is completed using corneal scissors.
Excision of host cornea: Trephination of host cornea can be done by using a manual trephine (e.g., Weck trephine), a vacuum trephine (e.g., Barron-Hessburg or Hanna trephine), or a femtosecond laser. A partial-thickness trephination is first carried out, the anterior chamber is then entered using a blade, and finally excision is completed using corneal scissors.
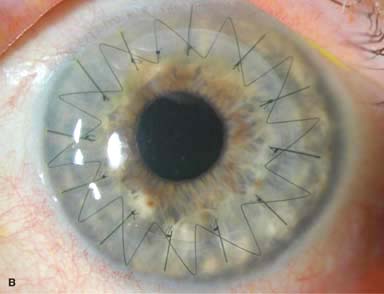
 Wound closure: Various suture methods, including interrupted, running, double running, or a combination may be used. 10-0 nylon is usually used (Fig. 10-2A–C).
Wound closure: Various suture methods, including interrupted, running, double running, or a combination may be used. 10-0 nylon is usually used (Fig. 10-2A–C).
 Postoperative management: topical corticosteroid and antibiotics.
Postoperative management: topical corticosteroid and antibiotics.
Combined Procedures
 Where indicated, penetrating keratoplasty can be combined with any of the following procedures:
Where indicated, penetrating keratoplasty can be combined with any of the following procedures:
 Cataract extraction with intraocular lens implantation (triple procedure)
Cataract extraction with intraocular lens implantation (triple procedure)
 Anterior vitrectomy with intraocular lens implantation
Anterior vitrectomy with intraocular lens implantation
 Intraocular lens exchange, either anterior chamber lens or scleral or iris fixated posterior chamber lens
Intraocular lens exchange, either anterior chamber lens or scleral or iris fixated posterior chamber lens
 Glaucoma drainage devices or trabeculectomy
Glaucoma drainage devices or trabeculectomy
 Pars plana vitrectomy, often with a temporary keratoprosthesis
Pars plana vitrectomy, often with a temporary keratoprosthesis
Complications
 Intraoperative
Intraoperative
 Eccentric, tilted, or irregular trephination of host or donor cornea
Eccentric, tilted, or irregular trephination of host or donor cornea
 Damage to iris or lens
Damage to iris or lens
 Expulsive hemorrhage
Expulsive hemorrhage
 Vitreous prolapse
Vitreous prolapse
 Postoperative
Postoperative
 Sight-threatening
Sight-threatening
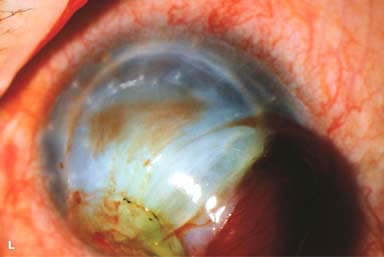
► Endophthalmitis (Fig. 10-2D)
► Glaucoma
► Retinal detachment
► Cataract
► Cystoid macular edema
 Graft-threatening
Graft-threatening
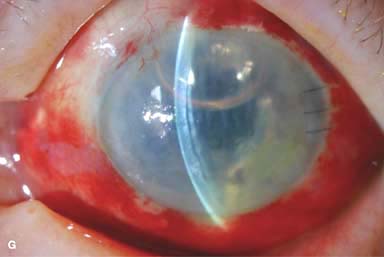
► Primary failure: endothelial damage during surgery, poor-quality donor tissue (Fig. 10-2E)
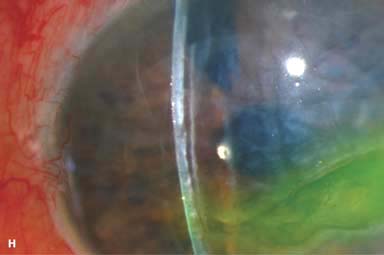
► Wound leak (Fig. 10-2F)
► Persistent epithelial defect (Fig. 10-2G)
► Flat anterior chamber (typically from a wound leak or angle-closure glaucoma)
► Immunologic rejection (see Chapter 8, Fig. 8-11)
► Infectious keratitis, suture abscess (Fig. 10-2H)
► Recurrence of disease: dystrophies, infection
► Epithelial downgrowth and retrocorneal fibrous membrane (Fig. 10-2I)
► Vitreous touch
 Other
Other
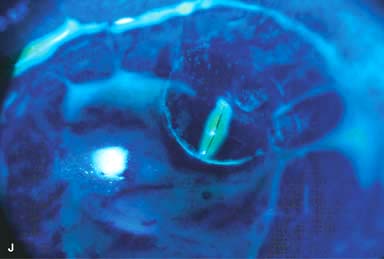
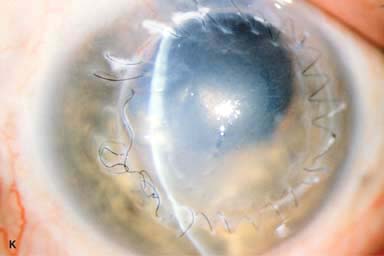
► Broken suture (Fig. 10-2J and k)
► Iris prolapse
► Wound dehiscence
► Traumatic wound rupture (Fig. 10-2L)
► Irregular astigmatism
Success Rate
 Poor to excellent, depending on the indication for surgery.
Poor to excellent, depending on the indication for surgery.
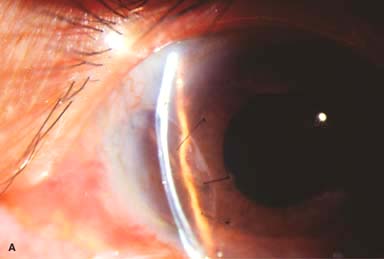
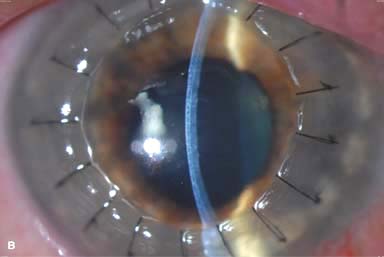
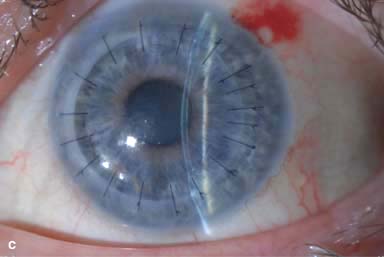
Figure 10-2. Corneal transplant. A. A clear corneal transplant is seen 6 weeks after surgery for herpes simplex scarring. Sixteen interrupted 10-0 nylon sutures are in place. B. A clear corneal transplant is seen 4 months after a corneal transplant for worsening Fuchs’ dystrophy with scarring after cataract surgery. Twelve interrupted and 12 bite running 10-0 nylon sutures are present. Corneal transplant. C. An old square corneal transplant performed by Dr. Castroviejo in New York City about 40 years earlier. The graft remains remarkably clear, although a cataract is developing. Corneal transplant complication—early endophthalmitis. D. Ten days after a corneal transplant, severe intraocular inflammation with fibrin in the anterior chamber and a small hypopyon at the graft–host margin inferiorly can be seen. Endophthalmitis was suspected and a vitreous tap revealed Staphylococcus epidermidis, which responded well to antibiotic treatment. Corneal transplant complication—primary graft failure. E. Diffuse severe corneal edema with whitening of the corneal stroma is present 1 day after a corneal transplant in an eye with primary graft failure. Primary graft failure generally occurs as a result of poor-quality tissue or damage to the corneal endothelium during surgery. Corneal transplant complication—wound leak. F. A wound leak is seen 3 days after a corneal transplant. Dark, concentrated fluorescein dye was placed at the graft–host margin at the 9 o’clock position. Aqueous fluid leaking through the graft–host wound can be seen to dilute the dye and turn it bright yellow-green. If the leak is minimal and the anterior chamber is formed, it can be treated medically and followed closely. If the leak is significant or the chamber is flat, the leak should be repaired surgically. Corneal transplant complication—wound melt. G. A severe corneal wound melt is present in the inferior half of this cornea 5 months after a corneal transplant. Multiple loose sutures can be seen. A small, permanent lateral tarsorrhaphy had already been performed. Corneal transplant complication—suture abscess. H. A broken corneal transplant suture and underlying abscess are present at the 10 o’clock position. A large hypopyon with a second small hypopyon at the graft–host junction can be seen inferiorly. Corneal transplant complication—epithelial downgrowth. I. A curvilinear retrocorneal membrane can be seen from 9 o’clock toward 12 o’clock and back down toward 3 o’clock. It is again visible around 5 o’clock. It advanced centrally over several weeks. It was diagnosed as epithelial downgrowth. Corneal transplant complication—broken interrupted suture. J. A broken corneal transplant suture is present at the 2 o’clock position. Fluorescein dye and the cobalt blue light demonstrate the “windshield wiper” effect of the broken suture. Corneal transplant complication—loose, exposed running suture. K. An extremely loose and exposed running suture can be seen a year after a corneal transplant. There is mucus adherent to the suture and secondary peripheral corneal neovascularization superiorly and corneal edema centrally. Expulsive hemorrhage after a corneal transplant wound dehiscence. L. Several years after a penetrating keratoplasty, blunt trauma caused the wound to dehisce and resulted in an expulsive hemorrhage. Note the large clot of blood exiting the wound. Iris pigment and vitreous jelly can be seen on the inferior cornea.
ENDOTHELIAL KERATOPLASTY
Endothelial keratoplasty involves removal of diseased Descemet’s membrane and endothelium (most commonly) and replacement with healthy Descemet’s and endothelium (Descemet’s membrane endothelial keratoplasty [DMEK]), usually with some posterior stroma (Descemet’s stripping endothelial keratoplasty [DSEK]).
Advantages
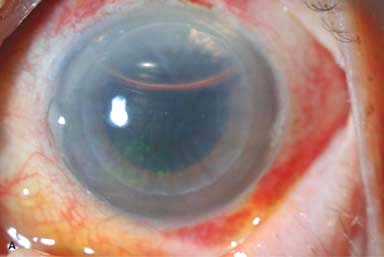
 More rapid visual recovery (Fig. 10-3A–E)
More rapid visual recovery (Fig. 10-3A–E)
 Stronger wound
Stronger wound
 No corneal sutures
No corneal sutures
 Less change in corneal curvature
Less change in corneal curvature
 Not an open-sky procedure
Not an open-sky procedure
Disadvantages
 Technically more challenging; more difficult to perform other intraocular surgeries
Technically more challenging; more difficult to perform other intraocular surgeries
 Potentially more traumatic to the donor endothelium
Potentially more traumatic to the donor endothelium
Most Common Indications
 Fuchs’ dystrophy
Fuchs’ dystrophy
 Pseudophakic bullous keratopathy
Pseudophakic bullous keratopathy
Surgical Technique
 Anesthesia: topical, local, or general anesthesia
Anesthesia: topical, local, or general anesthesia
 Determination of graft size: Routine graft sizes vary from 7.5 to 8.5 mm.
Determination of graft size: Routine graft sizes vary from 7.5 to 8.5 mm.
 Preparation of donor cornea: DSEK graft is typically fashioned with a microkeratome but can be fashioned with a femtosecond laser using an artificial anterior chamber or manually using a whole globe. DMEK tissue is generated manually.
Preparation of donor cornea: DSEK graft is typically fashioned with a microkeratome but can be fashioned with a femtosecond laser using an artificial anterior chamber or manually using a whole globe. DMEK tissue is generated manually.
 Removal of host Descemet’s and endothelium: Through a small scleral, limbal, or clear corneal incision, Descemet’s membrane is scored, stripped, and removed from the eye.
Removal of host Descemet’s and endothelium: Through a small scleral, limbal, or clear corneal incision, Descemet’s membrane is scored, stripped, and removed from the eye.
 Insertion of the donor cornea: Numerous insertion methods, including folding and using a variety of inserters, are used to place the donor cornea in the eye. An air bubble is used to push and hold the donor cornea up against the posterior stroma. The wound may be sutured closed; 10-0 nylon is usually used.
Insertion of the donor cornea: Numerous insertion methods, including folding and using a variety of inserters, are used to place the donor cornea in the eye. An air bubble is used to push and hold the donor cornea up against the posterior stroma. The wound may be sutured closed; 10-0 nylon is usually used.
 Postoperative management: topical corticosteroid and antibiotics
Postoperative management: topical corticosteroid and antibiotics
Combined Procedures
 As in penetrating keratoplasty, endothelial keratoplasty can be combined with other intraocular surgeries.
As in penetrating keratoplasty, endothelial keratoplasty can be combined with other intraocular surgeries.
Complications
 Donor graft folds or decentration (Fig. 10-3F)
Donor graft folds or decentration (Fig. 10-3F)
 Donor graft dislocation requiring rebubbling or repeat surgery (Fig. 10-3G and H).
Donor graft dislocation requiring rebubbling or repeat surgery (Fig. 10-3G and H).
 Others similar to penetrating keratoplasty, although there is less chance of expulsive hemorrhage, wound leaks, wound dehiscence, persistent epithelial defects, suture-related problems and irregular astigmatism
Others similar to penetrating keratoplasty, although there is less chance of expulsive hemorrhage, wound leaks, wound dehiscence, persistent epithelial defects, suture-related problems and irregular astigmatism
Success Rate
 Good to excellent, depending on the indication for surgery
Good to excellent, depending on the indication for surgery
Figure 10-3. Endothelial keratoplasty. A–D show the same eye. A. One day after surgery, the DSEK button is well centered. A 30% air bubble can be seen superiorly. B. Slit-beam view shows that the DSEK graft is 100% attached, with mild edema on postoperative day 1. Endothelial keratoplasty. C. Five weeks after DSEK surgery, the graft is perfectly centered and clearing nicely. D. Slit-beam view 5 weeks after surgery shows that the DSEK graft has minimal edema. The slit beam highlights the interface between the host anterior cornea and the thin posterior donor DSEK graft. Endothelial keratoplasty. E. Dual Scheimpflug photography using the Galilei system® shows the DSEK graft to be attached to the recipient cornea on postoperative day 1. There is mild DSEK edema centrally, but moderate edema at the edge of the graft. This peripheral graft edema typically resolves over several months. F. Moderate folds in this DSEK are seen a few days after surgery. Note the small air bubble still present superiorly. While the folds improved somewhat with time, vision did not improve significantly until the DSEK was repeated. Endothelial keratoplasty. G. On postoperative day 1, the host cornea and DSEK button are severely edematous because of a large fluid cleft in the interface. The edema did not resolve over the next 2 weeks. The DSEK graft was rebubbled in the office 2 weeks postoperatively and was 100% attached and essentially clear the following day! H. One day after surgery, this DSEK graft is detached and inferiorly displaced. There is also a large inferior epithelial defect. It was thought that the eye’s low pressure from previous glaucoma surgery predisposed it to poor graft adherence.

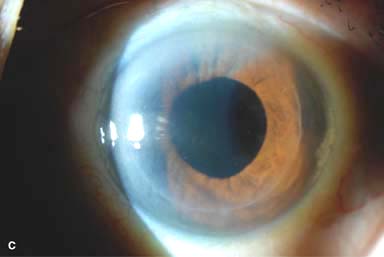
ANTERIOR LAMELLAR KERATOPLASTY
Anterior lamellar keratoplasty involves removal of the anterior cornea and replacing it with partial-thickness normal donor cornea.
Advantages
 Donor endothelial cell density is not important.
Donor endothelial cell density is not important.
 The anterior chamber is not entered, so complications such as expulsive hemorrhage and endophthalmitis are avoided.
The anterior chamber is not entered, so complications such as expulsive hemorrhage and endophthalmitis are avoided.
 There is no risk of endothelial rejection.
There is no risk of endothelial rejection.
Disadvantages
 Technical complexity of the procedure
Technical complexity of the procedure
 Opacification of the lamellar interface, which may reduce visual acuity
Opacification of the lamellar interface, which may reduce visual acuity
Indications
 Keratoconus
Keratoconus
 Anterior corneal scars and dystrophies
Anterior corneal scars and dystrophies
 Recurrent pterygium
Recurrent pterygium
 Limbal dermoid
Limbal dermoid
 Peripheral ulcerative keratitis
Peripheral ulcerative keratitis
 Terrien’s marginal degeneration
Terrien’s marginal degeneration
 Rarely, infectious keratitis, superficial corneal tumors
Rarely, infectious keratitis, superficial corneal tumors
Surgical Technique
 Local or general anesthesia
Local or general anesthesia
 Partial-thickness corneal trephination (encompassing the pathology) is performed on the recipient cornea with a blade or a femtosecond laser.
Partial-thickness corneal trephination (encompassing the pathology) is performed on the recipient cornea with a blade or a femtosecond laser.
 Lamellar dissection is then carried out on the recipient cornea using a blunt dissecting blade or a femtosecond laser. Alternatively, Descemet’s membrane can be separated from the posterior stroma (e.g., with air as in the Anwar big bubble technique or with viscoelastic), which is termed deep anterior lamellar keratoplasty (DALK).
Lamellar dissection is then carried out on the recipient cornea using a blunt dissecting blade or a femtosecond laser. Alternatively, Descemet’s membrane can be separated from the posterior stroma (e.g., with air as in the Anwar big bubble technique or with viscoelastic), which is termed deep anterior lamellar keratoplasty (DALK).
 Donor lamellar cornea is dissected and trephined with a blade or femtosecond laser. It is usually 0.25 to 0.5 mm larger than the recipient bed. Either a whole eye or a corneoscleral button fixated in an artificial chamber can be used. In DALK, a full-thickness corneal button, usually denuded of its Descemet’s membrane, is used.
Donor lamellar cornea is dissected and trephined with a blade or femtosecond laser. It is usually 0.25 to 0.5 mm larger than the recipient bed. Either a whole eye or a corneoscleral button fixated in an artificial chamber can be used. In DALK, a full-thickness corneal button, usually denuded of its Descemet’s membrane, is used.
 Alternatively, an automated microkeratome can be used to dissect both the donor and recipient cornea.
Alternatively, an automated microkeratome can be used to dissect both the donor and recipient cornea.
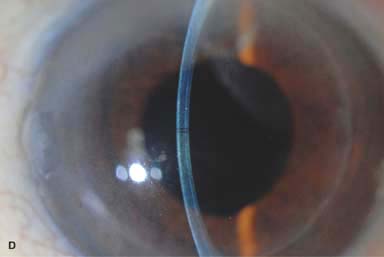
 The recipient bed is irrigated to remove epithelium, debris, and blood, and the donor cornea is sutured with 10-0 nylon using either interrupted or running sutures (Fig. 10-4A and B) or, occasionally, fibrin glue.
The recipient bed is irrigated to remove epithelium, debris, and blood, and the donor cornea is sutured with 10-0 nylon using either interrupted or running sutures (Fig. 10-4A and B) or, occasionally, fibrin glue.
 Postoperative management: topical corticosteroid and antibiotics
Postoperative management: topical corticosteroid and antibiotics
Complications
 Perforation of recipient or donor cornea during dissection, with possible need to convert to a penetrating keratoplasty
Perforation of recipient or donor cornea during dissection, with possible need to convert to a penetrating keratoplasty
 Double anterior chamber (Fig. 10-4C)
Double anterior chamber (Fig. 10-4C)
 Opacification of lamellar interface
Opacification of lamellar interface
 Irregular astigmatism
Irregular astigmatism
 Recurrence of disease: dystrophies, infection
Recurrence of disease: dystrophies, infection
 Persistent epithelial defect
Persistent epithelial defect
 Fibrovascular ingrowth into lamellar interface
Fibrovascular ingrowth into lamellar interface
 Broken suture
Broken suture
 Infectious keratitis
Infectious keratitis
 Stromal rejection (rare)
Stromal rejection (rare)
Success Rate
 Poor to excellent, depending on the indication for surgery
Poor to excellent, depending on the indication for surgery
Figure 10-4. Lamellar keratoplasty. A. A lamellar keratoplasty was performed in this eye with a large recurrent pterygium associated with significant corneal scarring. The slit beam shows the deep lamellar dissection. Multiple 10-0 nylon sutures are still in place. B. A half-thickness lamellar keratoplasty was performed for anterior corneal scarring. The recipient cornea was removed with mechanical dissection with a blade, whereas the donor button was fashioned with a microkeratome. On postoperative day 1 the slit-beam view demonstrates a relatively clear cornea; the graft–host interface is noted to be about half-thickness. Lamellar keratoplasty. C. This eye underwent a DALK procedure for deep corneal scarring resulting from HSV keratitis. A “double anterior chamber” was noted on postoperative day 1. It did not resolve with medical therapy, but it did resolve after filling the anterior chamber with air and then leaving a 40% air bubble. Although the corneal edema has completely resolved, note the folds in Descemet’s membrane where it does not lay down perfectly flat.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


