
Supranuclear Disorders of Gaze
What Is the Anatomy of Horizontal Conjugate Gaze?
Supranuclear structures coordinate the action of muscle groups and control two types of eye movements: conjugate movements (both eyes move in the same direction) and vergence movements (both eyes move in opposite directions). The vergence movements can either turn in (converge) or turn out (diverge) (Cassidy 2000). All of the supranuclear components act through a “final common pathway” for horizontal conjugate gaze. This final common pathway starts in the abducens nucleus (composed of two types of intermingled neurons: motor neurons and internuclear neurons). The axons of the internuclear neurons cross to the contralateral side in the lower pons, ascend in the medial longitudinal fasciculus (MLF), and synapse in the portion of the oculomotor nucleus that innervates the medial rectus muscle. The final common pathway is modulated by several inputs: the vestibular, optokinetic, smooth pursuit, and saccadic systems. As an example, an excitatory horizontal vestibulo-ocular impulse originating in the horizontal canal is relayed from the ipsilateral medial vestibular nucleus to the contralateral abducens nucleus, resulting in conjugate horizontal deviation of the eyes to the contralateral side (Johnston, 1992; Pierrot-Deseilligny, 1995; Tusa, 1998).
Where Are Lesions Causing Horizontal Gaze Palsies Located?
A lesion located anywhere along the supranuclear, nuclear, and infranuclear pathways that control horizontal eye movements may cause a horizontal gaze palsy. Depending on the location of the lesion, horizontal pursuit or saccades, or both, may be impaired.
Lesions causing defects in horizontal smooth pursuit are summarized in Table 14–1. Lesions causing defects in horizontal saccadic eye movements are summarized Table 14–2.
Frontal lobe: impaired ipsilateral horizontal smooth pursuit Posterior parietal cortex or temporo-occipito-parietal region decrease the amplitude and velocity of smooth pursuit toward lesion (Barton, 1996; Lekwuwa, 1996; Morrow, 1995) Occipitotemporal areas posteriorly, through the internal sagittal stratum, the posterior and anterior limbs of the internal capsule with adjacent striatum, to the dorsomedial frontal cortex anteriorly; ipsilesional pursuit deficits (Lekwuwa, 1996) Posterior thalamic hemorrhage: deficit in smooth pursuit toward lesion (Brigell, 1984) Unilateral midbrain or pontine lesion: ipsilateral pursuit defects (Furman, 1991; Gaymard, 1993; Johkura, 1998; Johnston, 1992; Kato, 1990; Thier, 1991; Waespe, 1992) Unilateral cerebellar damage: transient impairment of pursuit in direction of involved side Bilateral cerebellar damage: permanent impairment of smooth pursuit eye movements Posterior vermal lesion: may impair pursuit (Pierrot-Deseilligny, 1990) Middle cerebellar peduncle lesions or floccular lesions; ipsilateral pursuit defect (Kato, 1990; Waespe, 1992) |
What Studies Are Indicated in a Patient with a Horizontal Gaze Palsy?
In general, unilateral restriction of voluntary horizontal conjugate gaze to one side is usually due to contralateral frontal or ipsilateral pontine damage. At the bedside, pontine lesions can usually be differentiated from supranuclear lesions by associated neurologic findings and by the oculocephalic (doll’s-eyes) maneuver or caloric stimulation. These latter procedures will overcome gaze deviations induced by supranuclear lesions but will not overcome gaze deviations caused by pontine lesions. Structural lesions, such as infarction, hemorrhage, vascular malformations, tumors, demyelination, trauma, or infections, are the usual causes of horizontal gaze palsies. As all processes causing horizontal gaze palsies directly or indirectly damage intraparenchymal brain pathways, neuroimaging studies are necessary in all patients. In the acute setting, in patients with altered levels of consciousness, or in patients in whom magnetic resonance imaging (MRI) is contraindicated (e.g., patients with pacemakers), computed tomography (CT) is appropriate. Otherwise, MRI is the procedure of choice in evaluating patients with horizontal gaze palsies (class III, level B). In patients with evidence of clinical seizure activity, in patients with intermittent conjugate gaze deviation, or in obtunded or comatose patients with horizontal gaze palsies and evidence for possible contralateral cortical lesions, an electroencephalogram is indicted to evaluate the possibility a seizure disorder (e.g., status epilepticus) (class III, level C). The evaluation of patients with horizontal gaze palsies is outlined in Figure 14–1.
What Is the Anatomy of the Abducens Nucleus and Medial Longitudinal Fasciculus (MLF)?
The abducens nucleus has two types of intermingled neurons: motor neurons and internuclear neurons. The axons of the internuclear neurons cross to the contralateral side in the lower pons and ascend in the MLF to synapse in the portion of the oculomotor nucleus that innervates the medial rectus muscle (Leigh, 1999). In pontine lesions affecting the abducens nucleus and/or the paramedian pontine reticular formation (PPRF), a conjugate horizontal gaze palsy to the ipsilateral side occurs. Lesions of the MLF result in internuclear ophthalmoplegia (INO), whereas lesions of the MLF plus the ipsilateral abducens nucleus and/or PPRF result in the one-and-a-half syndrome. The clinical characteristics of these latter two syndromes and their evaluation are reviewed below.
Frontal lobe lesions Transient neglect contralaterally Defect in generating voluntary saccades Transient horizontal gaze deviation ipsilaterally acutely Gaze palsy overcome with the oculocephalic maneuver or caloric stimulation Late disorders of saccades (contralateral more than ipsilateral) due to frontal eye field (FEF) lesions (Tijssen, 1993, 1994) Prolonged eye deviation after stroke implies large stroke or preexisting damage to the contralateral frontal region (Steiner, 1984) Impaired ability to make a remembered sequence of saccades to visible targets (supplementary eye field lesions) Impaired performance of antisaccade tasks (dorsolateral prefrontal lesions) Epileptogenic lesions in the frontal eye fields Transient deviation of the eyes and head to the contralateral side (Godoy, 1990) Ipsiversive head and eye movements during a seizure may also occur Initial forced turning (versive) head and eye movements usually correspond to a contralateral epileptiform focus, but these initial contraversive movements may be followed by late ipsiversive or contraversive nonforced movements during the secondary generalization (Kernan, 1993) Unilateral parietal lesions Ipsilateral horizontal gaze preference with acute lesions contralateral inattention with right-sided lesions Unilateral or bilateral increased saccade latencies Hypometria for contralateral saccades Saccadic slowing Bilateral parietal lesions Acquired ocular motor apraxia (Dehaene, 1991; Harris, 1996; Prasad, 1994; Shawkat, 1996) Lesions in the corona radiata adjacent to the genu of the internal capsule Contralateral selective saccadic palsy (Fukutake, 1993) Lesion (e.g., hemorrhage) deep in a cerebral hemisphere, particularly the thalamus Eye deviation to the side of the hemiparesis (“wrong-way eyes”) (Tijssen, 1994) Paresis of contralateral saccades Supranuclear contralateral gaze palsies associated with ipsilateral oculomotor palsies Pontine lesions affecting the abducens nucleus and/or the paramedian pontine reticular formation (PPRF) Ipsilateral conjugate gaze palsy (Kataoka, 1997) Ipsilateral horizontal gaze palsy with ipsilateral esotropia (Coats, 1998) Acutely, eyes deviated contralaterally Doll’s-eye maneuver or cold caloric stimulation usually does not overcome gaze palsy Saccades toward side of lesion are present in contralateral hemifield but are slow with abducens nuclear lesions; ipsilaterally directed saccades from opposite field are small and slow or absent with PPRF lesions (Leigh, 1999) Horizontal gaze-evoked nystagmus on looking contralaterally Bilateral horizontal gaze palsies with bilateral lesions (Shimura, 1997) Selective saccadic palsy (bilateral lesions of pons) Voluntary saccades in both horizontal and vertical planes slow Smooth pursuit, the vestibulo-ocular reflex, the ability to hold steady eccentric gaze, and vergence eye movements preserved Paraneoplastic loss of horizontal voluntary eye movements or slow horizontal saccades (Baloh, 1993) Associated with persistent muscle spasms of the face, jaw, and pharynx Associated prostate carcinoma Congenital and familial bilateral horizontal gaze palsy (Aribal, 1998; Harris, 1996; Shawkat, 1996; Stavrou, 1999) May occur in isolation May be associated with kyphoscoliosis and facial contraction with myokymia MRI may reveal absence of abducens nuclei bilaterally (Aribal, 1998) Pseudo–horizontal gaze palsy with pontine lesions Damaging the medial longitudinal fasciculus on one side and the contralateral abducens nerve fascicle Bilateral internuclear ophthalmoplegia with the process subsequently extending laterally to the regions of the abducens fascicles bilaterally, resulting in complete horizontal gaze paralysis due to multiple sclerosis (Milea, 2001) |
What Are the Clinical Features of INO?
Clinically, an INO is characterized by adduction weakness on the side of the MLF lesion and monocular horizontal nystagmus of the opposite abducting eye. Convergence is usually preserved unless the responsible lesion is high in the midbrain. Often patients with INO have no visual symptoms, but some complain of diplopia (due to skew deviation or limitation of adduction) or oscillopsia. INO is most evident during horizontal saccadic eye movements, and the “adduction lag” is best detected during optokinetic testing using a tape or drum. For example, with a right INO when the drum is rotated to the right the amplitude and velocity of the adducting quick phase of the right eye is smaller and slower than that of the abducting saccades in the left eye. The pathogenesis of the nystagmus in the abducting eye is unclear but is likely a normal adaptive process that helps overcome the adducting weakness of the fellow eye (Getenet, 1993; Thömke, 1996). Other clinical findings noted with unilateral and bilateral INO are outlined in Table 14–3.
Vitamin E deficiency syndrome (abetalipoproteinemia) may cause an eye movement disorder that superficially resembles the wall-eyed bilateral internuclear ophthalmoplegia (WEBINO) syndrome (Yee, 1976). In both of these syndromes, patients demonstrate exotropia with associated adduction limitation and dissociated horizontal nystagmus on lateral gaze. In vitamin E deficiency, however, saccades are slower in the abducting eye than in the adducting eye, and the dissociated nystagmus is of greater amplitude in the adducting eye. This motility impairment is especially noted with abetalipoproteinemia, with other findings including ataxia, weakness, posterior column dysfunction, and pigmentary retinopathy.
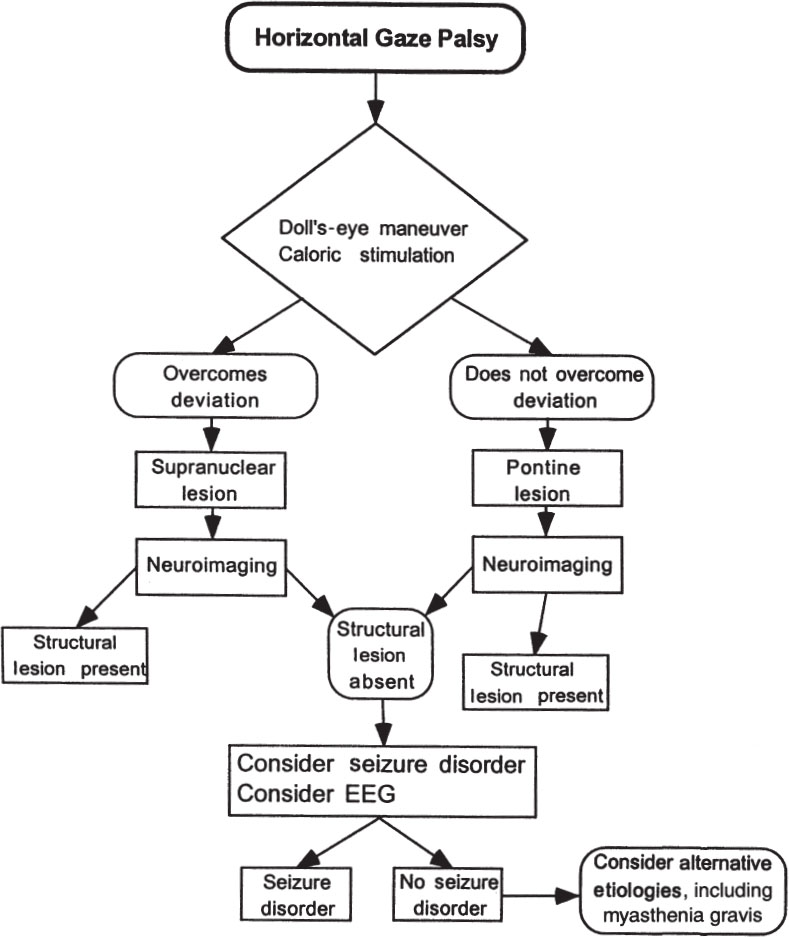
Figure 14–1. Evaluation of horizontal gaze palsy.
What used to be called Lutz posterior INO is now known as INO of abduction (Oliveri, 1997; Thömke, 1992b). In this rare syndrome, abduction is restricted on volition but can be fully effected by reflex maneuvers, such as cold caloric stimulation. Unilateral or bilateral INO of abduction, occasionally associated with adduction nystagmus of the contralateral eye, has been described with ipsilateral rostral pontine or mesencephalic lesions (Thömke, 1992b).
Unilateral INO Ipsilateral adduction weakness, especially slow or fractionated adducting saccades (“adduction lag”), and monocular nystagmus in contralateral abducting eye May have esophoria acutely, suggesting increased vergence tone Convergence usually spared Skew deviation with the higher eye on the side of the lesion Vertical gaze-evoked nystagmus and impaired vestibular and pursuit vertical eye movements (i.e., dissociated vertical nystagmus) (Leigh, 1999) Ipsilateral downbeat nystagmus and contralateral incyclorotatory (torsional) nystagmus (Marshall, 1991) Transient (disappearing within 3 days) torsional nystagmus, which is clockwise (examiner’s view) in cases of left INO and counterclockwise in right INO (Fantin, 1995) Normal vertical saccades Rare exotropia (wall-eyed monocular internuclear ophthalmoplegia [WEMINO] syndrome) (Johnston, 1994) Rare exotropia in contralateral eye due to overexcitation of contralateral PPRF when fixating with paretic eye (Komiyama, 1998) Bilateral INO Bilateral adduction paresis or lag with the eyes generally aligned in primary gaze Exotropia, with both eyes deviated laterally (wall-eyed-bilateral internuclear ophthalmoplegia [WEBINO] syndrome) (Fay, 1999; Flitcroft, 1996) Vertical gaze-evoked nystagmus (on looking up or down) and impaired vestibular and pursuit vertical eye movements (Leigh, 1999) Impaired vertical gaze holding |
What Is the Cause of the INO?
INO is due to pathologic processes affecting the medial pontine or midbrain parenchyma. Often there are associated brainstem symptoms and signs, although occasionally unilateral or bilateral INO may occur in isolation. The nature of the responsible pathologic process is suggested by the temporal mode of onset of the INO, the general clinical circumstances, and associated signs on neurologic and neuro-ophthalmologic examination. Etiologies for INO are listed in Table 14–1.
Although bilateral INO is more common with multiple sclerosis than with vascular insults, bilateral INO may occur with stroke as well as many other pathologic processes, and thus the presence of a unilateral or bilateral INO cannot be used as a differential feature for etiologic diagnosis (class III–IV, level C). For example, in a series of 100 patients with multiple sclerosis, 34 had INO, which was bilateral in 14 and unilateral in 20 (Muri, 1985). In another study of 51 patients with INO, 28 had multiple sclerosis and 23 had infarction; INO was bilateral in 33 patients and unilateral in 28 (Hopf, 1991). Most patients with nutritional, metabolic, degenerative, and drug-induced intoxication have bilateral INOs. Bilateral MLF involvement with the pathologic process subsequently extending laterally to the region of the two abducens fascicles has been described as explaining complete bilateral horizontal gaze paralysis in two patients with multiple sclerosis (Milea, 2001). The pattern of extraocular muscle weakness with myasthenia gravis (including penicillamine-induced myasthenia) can mimic INO (pseudo-INO) (Ito, 1997). Myasthenic pseudo-INO is not uncommon and may be associated with downshoot in the adducting eye (Ito, 1997). Other etiologies of pseudo-INO are listed in Table 14–4.
Multiple sclerosis (Flitcroft, 1996; Frohman, 2001a; Gass, 1997; Hopf, 1991; Milea, 2001) Brainstem infarction (Alexander, 1991; Getenet, 1993; Hopf, 1991; Kataoka, 1997; Marshall, 1991; Nagasaka, 1999; Okuda, 1993) Giant cell arteritis (Ahmad, 1999; Askari, 1993; Hughes, 1994; Johnston, 1990; Trend, 1990) Systemic lupus erythematosus Neuro-Behçet’s disease (Masai, 1995) Sickle cell trait (Leavitt, 1994) Polyarteritis nodosa (Kirkali, 1991) Eales’ disease (Ataby, 1992) Pyoderma gangrenosum (Lana, 1990) Sneddon’s syndrome (Rehany, 1998) Complication of angiography Complication of cardiac catheterization (Mihaescu, 2000) D-penicillamine–induced cerebral vasculitis (Pless, 1997) Brainstem hemorrhage, including hemorrhage due to “crack” cocaine use (Diaz-Calderon, 1991) Brainstem and fourth ventricular tumors (Arnold, 1990) Infections Cryptococcal meningitis (Fay, 1999; Sung, 1991) Tuberculosis (granuloma or infectious vasculitis) Viral and bacterial meningoencephalitis (Luis Guerrero-Peral, 2001) Syphilis Poliomyelitis (Wasserstrom, 1992) AIDS (Cacciatori, 1997) Jakob-Creutzfeldt disease (Billette de Villemeur, 1996) Head trauma (Chan, 2001; Haller, 1990; Hsu, 2001; Mueller, 1993; Strauss, 1995) Cervical injury by hyperextension or manipulation Cancer-related Carcinomatous meningitis Remote effect of cancer Brainstem demyelination due to chemotherapy/radiation therapy Nutritional and metabolic disorders Wernicke’s encephalopathy (De la Paz, 1992) Pernicious anemia Hepatic encephalopathy Maple syrup urine disease Abetalipoproteinemia Fabry’s disease Hexosaminidase A deficiency (Barnes, 1991) Degenerative diseases Progressive supranuclear palsy (Friedman, 1992) Familial spinocerebellar degeneration (Senanayake, 1992) Arnold-Chiari malformation and associated hydrocephalus or syringobulbia (Arnold, 1990b; Chavis, 1998; Lewis, 1996) Drug intoxications Narcotics Phenothiazines Tricyclic antidepressants Propranolol Barbiturates Lithium Antiobesity treatments (Lledo Carreres, 1992) Toluene (Hunnewell, 1998) Miscellaneous causes Hydrocephalus Pseudotumor cerebri (Friedman, 1998) Mesencephalic midline clefts (Lagreze, 1996) Subdural hematoma Subdural hygroma with an arachnoid cyst in the middle cranial fossa (Minamori, 1992) After external ventricular drainage of a benign aqueductal cyst (Shin, 2000) Supratentorial arteriovenous malformations Partial seizures Pseudo-INO Myasthenia gravis (may be associated with downshoot of adducting eye) (Ito, 1997) Fisher syndrome (Al-Din, 1994; Mori, 2001; Ortuno, 1990) Guillain-Barré syndrome Myotonic muscular dystrophy (Azuara-Bianco, 1997; Verhagen, 1998) Surgical paresis of the medial rectus muscle |
What Studies Should Be Ordered in a Patient with INO?
In general, the investigation of a patient with INO depends on the clinical circumstances. For example, in a patient with known multiple sclerosis, the appearance of INO as part of an exacerbation of the disease may not require neuroimaging (class IV, level C), whereas INO in isolation or with associated unexplained brainstem signs and symptoms usually requires neuroimaging (class III–IV, level C). If there is variability of the adduction deficit, associated fluctuating ptosis, or other variable ocular motor signs suggestive of myasthenia gravis, a myasthenic pseudo-INO should be considered (class III–IV, level C). If the evaluation including a Tensilon test is normal or if the clinical situation does not suggest myasthenia and there are no signs of an associated degenerative process (e.g., progressive supranuclear palsy) on clinical examination, neuroimaging is usually warranted (class III–IV, level C).
MRI is superior to CT scan in evaluating patients with INO (Alexander, 1991; Atlas, 1987; Frohman, 2001a; Haller, 1990; Hopf, 1991; Strauss, 1995). For example, in a study of 11 patients with INO (nine with multiple sclerosis and two with infarct), CT in all nine tested failed to show a responsible lesion, whereas appropriate MRI abnormalities were documented in 10 of 11 patients (Atlas, 1987). In another study, CT did not detect abnormalities of the MLF in two patients with INO who had abnormal brainstem hypersignals on MRI (Awerbach, 1990). In a study of chronic INO in 58 multiple sclerosis patients, proton density imaging (PDI) revealed a hyperintensity in the MLF in all patients, whereas T2-weighted imaging and fluid-attenuated inversion recovery (FLAIR) imaging showed these lesions in 88% and 48% of patients, respectively (Frohman, 2001a). Thus, PDI imaging may show the MLF lesion in patients with multiple sclerosis and INO. CT imaging in a patient with INO is reserved only for acute situations (e.g., brainstem hemorrhage) or for patients in whom MRI is contraindicated (e.g., pacemakers, etc.). MRI may give useful diagnostic data by also giving information about supratentorial processes likely to be involved in the etiology of the INO, such as multiple sclerosis, multiple cerebral infarcts, etc. If an infarct is detected as the cause of INO in a patient older than 50 years of age, giant cell arteritis should be considered as an etiology, especially if other stroke risk factors are not evident (class III, level C). Thus, a sedimentation rate is warranted, and if it is elevated or the patient has other systemic symptoms of giant cell arteritis (e.g., jaw claudications, headache, polymyalgia rheumatic symptoms, etc.), a temporal artery biopsy should be obtained. There is insufficient evidence to recommend an evaluation for giant cell arteritis in every INO in the elderly (class III–IV, level U).
If MRI in nontraumatic cases is normal, then rarer etiologies for the INO should be considered (class III–IV, level C). If the INO is bilateral, drug intoxication should be suspected. Because pernicious anemia has rarely been reported to cause INO, a B12 level should be considered (class IV, level C). Syphilis may rarely cause INO, so serology for syphilis should be considered (class III–IV, level U). If MRI reveals meningeal enhancement or if meningeal signs or symptoms are present, spinal fluid examination is warranted to search for infectious or carcinomatous meningitis (class III– IV, level C). The suggested evaluation of a patient with INO is outlined in Figure 14–2.
What Is the One-and-a-Half Syndrome?
In the one-and-a-half syndrome, there is a conjugate gaze palsy to one side (“one”) and impaired adduction on looking to the other side (“and-a-half”) (Bronstein, 1990; Celebisoy, 1996; Wall, 1983). As a result, the only horizontal movement remaining is abduction of one eye, which may exhibit nystagmus in abduction. The responsible lesion involves the PPRF or abducens nucleus and the adjacent MLF on the side of the complete gaze palsy. Patients with the one-and-a-half syndrome often have exotropia of the eye opposite the side of the lesion (paralytic pontine exotropia). Rarely, a primary position esotropia occurs, most likely due to involvement of the abducens nerve fascicle superimposed upon lesions of the PPRF and MLF (Wall, 1983).
The one-and-a-half syndrome may be associated with ocular bobbing and, more often, facial nerve palsy (the “eight-and-a-half syndrome”) (Eggenberger, 1998). Patients with the one-and-a-half syndrome and facial nerve palsies may develop oculopalatal myoclonus months to years after the onset of the ocular motility problem (Wolin, 1996). The one-and-a-half syndrome may also be associated with supranuclear facial weakness on the same side as the gaze palsy and an INO. The lesion is in the paramedian aspect of the dorsal pontine tegmentum, providing evidence for the existence of corticofugal fibers that extend to the facial nucleus in the dorsal paramedian pontine tegmentum (Anderson, 1999).
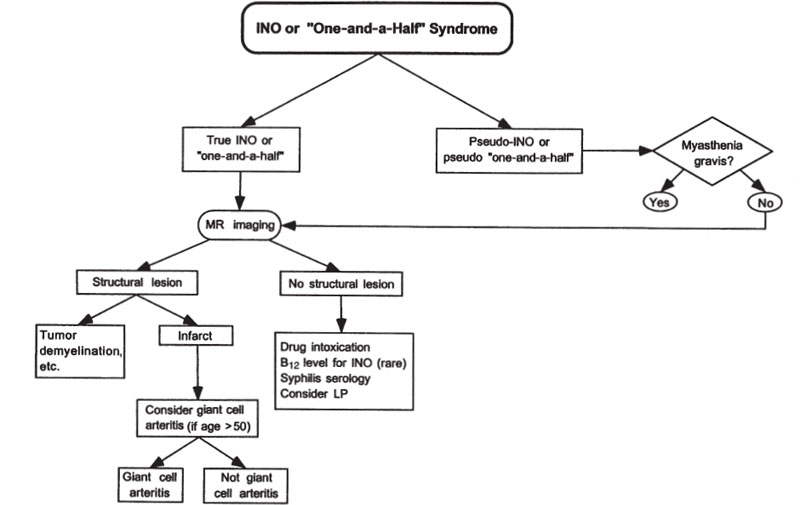
Figure 14–2. Evaluation of internuclear ophthalmoplegia (INO) and the one-and-a-half syndrome.
Another type of one-and-a-half syndrome may result from two separate lesions involving both MLFs and the fascicles of the abducens nerve on the side of the unilateral horizontal “gaze” palsy. In this case, however, if the “gaze” palsy is incomplete, the eyes would move disconjugately in the direction of the gaze palsy (Pierrot-Deseilligny, 1981). A true gaze palsy due to unilateral PPRF damage causes concomitant paresis of both eyes. Another form of one-and-a-half syndrome consists of a horizontal conjugate gaze palsy to one side (e.g., to the left). This is associated with abduction paralysis of the eye on the side opposite to the gaze palsy (e.g., the right eye) and adduction nystagmus in the eye on the side of the gaze palsy (e.g., in left eye) (Çelebisoy, 1996). This form of the one-and-a-half syndrome may be seen with rostral brainstem infarction. A fourth type of (pseudo) one-and-a-half syndrome has been described in a patient with mucormycosis of the cavernous sinus (Carter, 1994). The patient had an ipsilateral sixth nerve palsy due to cavernous sinus involvement and a contralateral horizontal gaze palsy due to simultaneous carotid artery occlusion with infarction of the frontal lobe. Contrary to the pontine one-and-a-half syndrome, in which abduction in one eye is the preserved horizontal movement, this patient had only preserved adduction in one eye (contralateral to the sixth nerve palsy). Myasthenia gravis or the Miller Fisher syndrome may produce findings that mimic a one-and-a-half syndrome (“pseudo-one-and-a-half syndrome”).
What Etiologies Should Be Considered as a Cause of the One-and-a-Half Syndrome and What Evaluation Is Indicated?
Etiologies of the one-and-a-half syndrome are listed in Table 14–5. Brainstem infarction is the most common cause of the one-and-a-half syndrome in the elderly, whereas multiple sclerosis is the most common cause of the one-and-a-half syndrome in young adults. In fact, the one-and-a-half syndrome may be the presenting sign of multiple sclerosis (Martyn, 1988).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


