Superficial Keratectomy
Kenneth R. Kenyon
Dasa V. Gangadhar
Leslie T. L. Pham
Kenneth M. Goins
Michael D. Wagoner
Superficial keratectomy is the microsurgical removal of subepithelial fibrous membranes and/or anterior corneal opacities that occur in a variety of corneal degenerative and dystrophic conditions. Although most commonly performed with simple manual microdissection, the technique has more recently evolved to employ technology including the excimer laser, femtosecond laser, and microkeratome. Here we present the indications, surgical technique, and postoperative management for manual superficial keratectomy, as well as automated therapeutic alternatives and our personal experience with this remarkably useful and noninvasive operative procedure.
INDICATIONS
A host of corneal dystrophies, degenerations, and other ocular surface disorders have pathologic changes limited to the anterior cornea.1 The most common general indications for performing superficial keratectomy are either decreased vision from corneal opacification and irregular astigmatism or pain due to recurrent epithelial erosions.2,3,4,5,6,7,8,9,10 As will be detailed subsequently, those conditions affecting the epithelium, basement membrane complex, and Bowman’s layer are especially appropriate for superficial keratectomy, while those of the superficial stroma may also benefit.
PREOPERATIVE ASSESSMENT, SURGICAL TECHNIQUE AND POSTOPERATIVE MANAGEMENT
The fundamental theme of manual superficial keratectomy technique has been described by numerous authors.8,9,10 Our variations thereof are as follows11,12,13,14: treatment should be limited to the areas of superficial corneal abnormality and is seldom extended broadly over the entire corneal surface. Hence it is necessary to perform careful slit lamp examination and then to determine and diagram the specific areas to be treated. This examination is facilitated by utilizing fluorescein (Fluoress) to highlight abnormal areas of tear film break up, stain uptake, and “negative” staining, the latter denoting subepithelial material elevations that thin the tear film. Automated corneal topography, especially with more sensitive instruments such as the Orbscan or Pentacam, can also detect and depict irregular astigmatism related to focal lesions.
If recurrent epithelial erosion is also a component of the particular case, then the so-called Weck-Cel adhesion test is of diagnostic aid and of assistance to define the areas requiring debridement as well as to ascertain if concomitant anterior stromal puncture or diamond burr polishing is also required. Performed at the slit lamp, this adhesion test simply involves topically anesthetizing the cornea and then gently dragging the tip of a dry cellulose surgical sponge across the epithelium in areas suspect for loose epithelial-stromal adhesion. If the epithelial adherence is normal, then the epithelial sheet does not shift position (negative test). However, if the epithelium is loosely adherent and at high risk for recurrent erosion, then the epithelial sheet can be easily moved over the Bowman’s and stromal substrate (positive test).
Finally, it is also essential to assess corneal sensation preoperatively. This is most readily performed with the drawn out end of a cotton applicator, or alternatively with the more sophisticated Cochet-Bonet esthesiometer. If corneal sensation is intact, then there is almost never a risk of delayed epithelial healing. However, in the absence of corneal sensation (e.g., following Herpes simplex or zoster keratitis), then the increased postoperative risk of delayed reepithelialization or even a persistent epithelial defect must be considered. The management of such neurotrophic situations is beyond the scope of this discussion but might include additional strategies such as amnion membrane overlay and/or lateral tarsorrhaphy.
The superficial keratectomy procedure itself is then typically performed in a minor surgical setting where sterile instrumentation and technique plus an operating microscope are available. Topical proparacaine or tetracaine is administered followed by a drop of povidone iodine and lid skin prep with Betadine or alcohol. Subconjunctival or peribulbar anesthetic injection is rarely required.
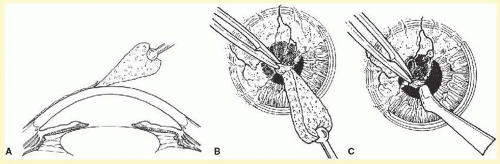
The corneal surface is dried with a cellulose surgical sponge and importantly must remain dry to facilitate identification of the abnormal fibrocellular deposits to be removed. As epithelial-stromal adhesion is also frequently defective in the areas of abnormality, scrubbing lightly over the epithelial surface with a dry sponge or a perpendicularly oriented scarifier or crescent blade will very often easily accomplish removal of the epithelium overlying the abnormal deposits. Following epithelial removal and still maintaining a dry corneal surface, the subepithelial fibrous plaques are readily identified against the background of the otherwise smooth Bowman’s layer. Slightly more vigorous scraping with sponge or blade (e.g., Beaver 57, BVI) allows the margins of the focal deposits to be identified and thereby reveals the naturally occurring cleavage planes that can then be readily exploited, as the edges of these often cellophane-like fibrocellular membranes (as, e.g., in epithelial basement membrane dystrophy (EBMD) or Reis-Bücklers dystrophy) or dense collagenous plaques (as in Salzmann nodular degeneration) can then be grasped with jeweler’s or fine tissue forceps and stripped, frequently as a continuous sheet, often to disclose a smooth and glistening Bowman’s layer or an intact the felt-like stromal surface. After removal of discrete focal pannus and membranes, the perpendicularly oriented blade may again be gently used to scrape or dissect more firmly adherent tissue. Importantly, sharp incision or dissection should be specifically avoided to prevent damage to the underlying Bowman’s and stromal layers. Once cleared in this manner, the corneal surface can be again polished with a dry cellulose sponge (Fig. 28.1).11 In cases having a history of recurrent epithelial erosion and/or demonstrating a positive Weck-Cel adhesion test, then anterior stromal puncture is additionally performed utilizing an 18G needle in the areas of aberrant adhesion, excluding the central visual axis.14,15 Alternatively, careful polishing with a diamond burr can be applied to the area of keratectomy and can also be safely extended into the visual axis if needed (see Videos).
Once completed, a therapeutic soft contact lens (SCL), usually with a flat base curve (8.6 to 9.0), is inserted to facilitate reepithelialization and to provide comfort. Given the universal availability of appropriate SCLs, it is seldom necessary to resort to pressure patching. Prophylactic topical antibiotic (usually a fluoroquinolone) and topical corticosteroid (usually prednisolone acetate 1%) are used four times daily until reepithelialization is complete and then reduced for the duration of SCL use. Topical nonsteroidal anti-inflammatory drug (NSAID) can be used frequently during the first few days but should be discontinued once its analgesic effect is no longer required as prolonged use might slow epithelial healing. Hence major oral analgesics are seldom required. Depending on the size of the epithelial defect, resurfacing is usually complete within 5 to 7 days. The SCL should be retained for as long 2 weeks in cases not involving recurrent erosions; however, in cases with erosive propensity, 6 weeks or more is recommended. Although topical antibiotic, steroid, and NSAID can be tapered over this interval and then discontinued once the SCL therapy is finished, frequent use of lubricant drops by day and ointment at bedtime is essential to prevent erosive exacerbations. In selected situations, systemic doxycycline and a weak topical steroid (e.g., prednisolone acetate 0.125% or fluorometholone 0.1%) are of use to minimize postoperative subepithelial collagenase production and its deleterious effect on redevelopment of epithelial-stromal adhesion.16
Patients wearing refractive contact lenses can cautiously return to lens wear at the completion of the postoperative recovery interval. Patients desiring refractive surgery or requiring cataract surgery should delay 2 to 3 months until the corneal surface and refraction have again stabilized.
THERAPEUTIC ALTERNATIVES
The excimer laser has now been used for two decades to perform a variant of superficial keratectomy, termed phototherapeutic keratectomy (PTK), to manage superficial corneal scars and opacities17,18,19,20,21,22,23 as well as erosions.24,25,26,27 As such, precise amounts of superficial corneal tissue can be removed to restore an optically clear and smooth surface, thereby avoiding anterior lamellar or penetrating keratoplasty. Refinements of microkeratome instrumentation consequent to the development of laser-assisted in situ keratomileusis (LASIK) have also found application for precise lamellar resection of anterior corneal stroma,28 affording, for example, superior results for irregular climatic droplet degeneration29 than the use of excimer PTK alone.30 Most recently, application of the femtosecond laser (e.g., AMO, Technolas or Zeiss instruments) has further advanced the precision of excising a superficial plane of anterior stromal tissue, as we have utilized for primary and recurrent superficial corneal dystrophies.
The main advantage of these automated techniques lies in their ability to remove deeper corneal opacities (into the anterior one-third of the stroma) than are readily accessible by mechanical superficial keratectomy.19,20 Their principle disadvantages are the high capital and utilization costs of the instrumentation, the induction of refractive errors (typically hyperopia and irregular astigmatism) if substantial depth of tissue removal is required, and the potential to develop visually significant haze as a consequence of the treatment itself (specifically with excimer PTK). For these reasons, although such automated techniques have definite utility in specific situations, the straightforward, noninvasive, and economical superficial keratectomy remains our overall procedure of choice.
It must also be cautioned that excimer PTK is not simply “point-and-shoot” surgery. In particular, as scar tissue ablates at a different rate than adjacent normal stroma, masking with viscous agents and/or blending techniques is necessary to avoid induction of a lesion free but optical aberration-abundant stromal surface.
A detailed therapeutic algorithm to guide decision making among superficial keratectomy, excimer PTK, and combinations thereof has been devised by Wagoner19,20,31 based on the key principles that
1. The primary diagnosis does not necessarily dictate the treatment of choice, as various clinical presentations of the same disorder may dictate different therapeutic approaches. For example, early primary stromal dystrophies in previously treated eyes may be readily amenable to manual superficial keratectomy, whereas more advanced disease might require excimer PTK or anterior lamellar keratoplasty for adequate clearing of the visual axis.
2. Identical clinical presentation does not dictate the treatment of choice, as functional requirements of the patient may also require different therapeutic approaches. For example, excimer PTK might be more appropriate for ultimate visual rehabilitation of a myopic eye with Reis-Bücklers dystrophy, whereas a hyperopic eye with the same condition might be better served by manual superficial keratectomy.
3. Combined approaches may be required in certain situations. For example, in conditions with higher surface elevations, such as Salzmann degeneration, superficial keratectomy is initially necessary to truncate elevated nodules before proceeding with PTK.32 However, even in these cases, manual superficial keratectomy with diamond burr polishing generally affords sufficient treatment.
Apart from the major dichotomy of superficial keratectomy versus excimer PTK versus combinations thereof, additional adjunctive techniques have been devised to facilitate the overall goals of superficial keratectomy. In situations involving epithelial erosion, various techniques to reinforce epithelial-stromal adhesion have been advocated, including iodine cauterization of Bowman’s layer (no longer recommended),4 focal diathermy, diamond burr polishing,33,34,35,36,37 and neodymium:YAG laser application.38 In this scenario, however, our usual preference remains focal anterior stromal puncture, as described by McLean,15 as it is brief, safe, effective, and economical. Thus we conclude every case of superficial keratectomy also involving a recurrent erosion-associated disorder with application of anterior stromal puncture or diamond burr polishing.
Other potentially useful techniques include the adjunctive application of mitomycin C (usually 0.02% locally applied using surgical sponge disc for 30 seconds or less) generally to reduce recurrent scar formation (e.g., for stromal haze following excimer photorefractive keratectomy, PRK) or specifically to prevent recurrence of ocular surface dysplasia and neoplasia.39,40 Amniotic membrane overlay grafts41 are especially beneficial for neurotrophic situations with propensity for persistent epithelial defects.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree