This chapter covers the broad category of diagnoses that fall under the diagnosis of stomatitis. The term “stomatitis” indicates an inflammation and potential breakdown of the oral cavity mucosal surfaces and is typically symptomatic. The clinical presentation may extend to the oropharynx or the external perioral region. The etiologies are multiple and include treatment-related, infectious, immunologic, systemic, nutritional, and idiopathic causes.
The diagnoses that fall under this classification can be challenging to identify and require a detailed assessment of a patient’s history of presentation and associated conditions. Additionally, these presentations are not solely seen in the adult population and some forms of stomatitis are unique to the pediatric population.
Given that many of these diagnoses are localized manifestations of systemic diseases, collaborative management of patients with our Dermatology, Infectious Disease, Oral Medicine, and/or Rheumatology colleagues is expected.
Interestingly, despite being encountered daily in general practice, the etiology of many of these oral lesions remains unclear. Many of the advocated treatment options vary dramatically within the medical literature and may only offer symptom control and not affect cure of the underlying diagnosis. As such, this chapter attempts to summarize some of the common perspectives relative to these diagnoses; however, for those seeking more detail on any one condition, the reader is encouraged to review the articles cited within the chapter’s reference list.
CLINICAL ASSESSMENT
The clinical appearance of stomatitis is variable. Presentations can differ from simple localized to fulminant manifestations and may include components of mucosal erythema, hyperkeratosis, ulceration, pseudomembrane, and/or vesicle formation.
The rate of epithelial turnover in the oral cavity ranges from 3 to 7 days. As a result, this anatomic location is quick to demonstrate visible signs of a metabolic disturbance from a systemic condition or in relation to treatment (
1).
Throughout this chapter, a theme should develop relative to the importance of a thorough history of presentation and the assessment for associated diagnoses. Some of the conditions that are discussed are diagnoses of exclusion, while others may represent the earliest presentation of a more significant systemic process.
Pertinent issues that should be addressed in the history include the circumstances and the time course surrounding the onset of symptoms, recent changes in medications, aggressiveness of oral hygiene and product use, palliating/provoking factors, quality of pain and any prodromal symptoms.
A full head and neck examination is necessary in these cases in an effort to assess for synchronous findings beyond the oral presentation. Dentures should be removed and assessed for quality of fit while asking the patient about the original date of manufacture in addition to visits for modification. In cases of buccal mucosal lesions, the relationship of the ipsilateral dentition and any amalgam that contacts the mucosa in a closed mouth position should be identified. Additionally, the quality of salivary production should be assessed.
Exudative lesions should be considered for gram stain and culture (with fungal cultures additionally requested). Tzanck smear and viral cultures should also be considered in patients with vesicular lesions.
Laboratory assay, in cases where a wide differential diagnosis continues to exist after history and physical examination, can include a complete blood count (CBC) with differential, erythrocyte sedimentation rate (ESR), antinuclear antibody (ANA), and cytoplasmic-antineutrophil cytoplasmic antibodies (c-ANCA) in addition to levels
for iron, ferritin, total iron-binding capacity, folate, thiamine (B1), riboflavin (B2), pyridoxine (B6), cobalamin (B12), zinc, and magnesium (
2). In xerostomic patients, anti-SSA and anti-SSB can be drawn to assess for Sjogren disease.
Biopsy remains a powerful tool for establishing a definitive diagnosis in patients with stomatitis of unclear etiology. Patients with vesiculobullous lesions should have perilesional tissue sent for direct immunofluorescence (DIF) for deposition of IgG, IgA, and/or complement C3. Biopsy should be considered in patients with suspected recurrent aphthous stomatitis (RAS), oral lichen planus (OLP), or graft-versus-host disease (GVHD) to rule out oral squamous cell carcinoma because of the potential for similarity in appearance.
Patients with a history and findings highly suggestive of an inflammatory bowel condition, such as Crohn disease or ulcerative colitis (UC), should be considered candidates for colonoscopy.
ETIOLOGY
Our understanding of the etiology of treatment-related mucositis at the cellular level continues to evolve. Some aspects may be genetically predetermined and explain the heterogeneity of response between patients to different treatments.
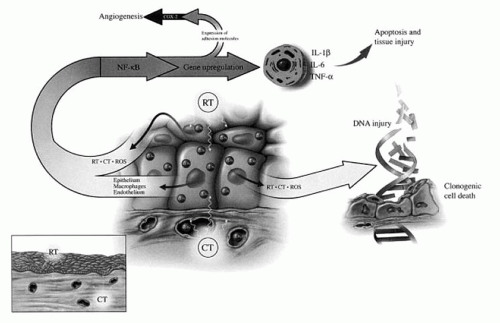
Theories on the development of mucositis have traditionally stated that it was the result of direct damage to the basal epithelial cell layer leading to an impairment of the renewal capacity of the surface epithelium. The resultant clonogenic cell death would result in atrophy and consequent ulceration with the clinical presentation of mucositis.
Additional evidence has suggested that an acute microvascular injury resulting from endothelial apoptosis may play a more significant role in the etiology of mucositis than previously considered. Electron microscopy has demonstrated that endothelial and connective tissue damage precede epithelial changes in irradiated oral mucosa. This finding suggests that the development of treatment-related mucositis is not just an epithelial process.
Sonis et al. (
3) describe the five phases of mucosal barrier injury as a result of chemotherapy and/or radiation therapy as:
Initiation with generation of reactive oxygen species
Up-regulation with generation of messengers including nuclear factor-κB
Signaling and amplification with proinflammatory cytokines including tumor necrosis factor-alpha (TNF-α)
Ulceration with inflammation with bacterial colonization and pain
Healing with epithelial proliferation and the return of local microbial flora
TREATMENT-RELATED STOMATITIS
Stomatitis can occur as a result of multiple forms of therapy; however, it is most evident and has the greatest impact in the treatment of cancer. It is expected that with chemotherapy and radiation therapy some degree of stomatitis, also known as mucositis, development will be experienced. Strategies for prevention, assessment, and treatment of treatment-related mucositis have been well documented; however, variation in findings and recommendations exist.
Assessment tools typically categorize the severity of a presentation by assessing a patient’s well-being, utilizing mouth and throat soreness (MTS) scores, and defining the patient’s functional capacity. Oral function can be assessed through dietary capacity, the ability to eat in public, and overall speech quality. An example of a quality-of-life instrument used in this setting is The Oral Mucositis Weekly Questionnaire—Head and Neck Cancer. It has been shown to be a reliable and valid means for assessing the impact of mucositis on patients undergoing treatment (
4).
Administration of 5-fluorouracil, with or without leucovorin, is associated with oral mucositis in as many as 40% of patients with grade 3 and 4 presentations occurring in 10% to 15% of patients (
5). In a double-blind randomized study by Sorensen et al., patients undergoing chemotherapy with 5-fluorouracil and leucovorin received prophylactic chlorhexidine or cryotherapy in attempt to decrease treatment-associated mucositis. Prophylactic chlorhexidine (10 mL at 0.1% concentration) was utilized three times per day with 1-minute oral rinses. Cryotherapy was performed with crushed oral ice 10 minutes before and
35 minutes after administration of chemotherapy. The frequency and duration of oral mucositis were significantly improved with both treatment options. Combined use of cryotherapy and chlorhexidine were not examined during this study (
6).
Another preventative treatment option, frequently reported in patients with hematologic malignancies, is palifermin. Palifermin is a recombinant human keratinocyte growth factor-1 (hKGF-1) that is considered to offer mucosal protection by inducing epithelial hyperplasia. Typical administration is in multiple pre- and posttreatment administrations of 60 µg/kg. Use of single-dose palifermin was studied in sarcoma patients receiving doxorubicin chemotherapy in a randomized, double-blind, placebocontrolled trial. Single-dose intravenous palifermin (180 µg/kg body weight), administered before each cycle, was noted to significantly reduce the incidence and severity of mucositis experienced. During the trial, the drug was well tolerated and majority of patients experienced thickening of oral mucosa and tongue. Biopsies of the affected areas noted increased levels of proliferative marker Ki-67 suggesting a palifermin-associated etiology (
7).
The medications associated with treatment-associated mucositis are not limited to the conventional chemotherapeutic options but also include agents like hydroxyurea (
8) and the newer targeted therapies such as imatinib mesylate (
9), cetuximab, panitumumab, gefitinib, erlotinib, rapamycin, sorafenib tosylate, and lapatinib (
10).
Mucositis associated with radiation therapy is a common finding that tends to be self-limited after the completion of treatment. Mucositis typically peaks between weeks 5 to 6 during treatment as assessed by MTS scores (
11). Radiation-associated mucositis is worse in patients undergoing radiation with accelerated regimens (
5). Grade 3 and 4 mucositis requiring gastrotomy tube placement is more significant with chemoradiation protocols (38%) than compared to radiation alone (18%). The risk of developing mucositis is the same for patients undergoing treatment of an oral cavity and oropharyngeal cancer as those treated for a laryngeal and hypopharyngeal cancer.
Prophylactic pilocarpine has been shown to decrease the incidence and severity of treatment-associated mucositis that is presumed through the up-regulation of salivary function during treatment (
12). However in contrast, in a randomized phase III prospective trial of bethanechol, which has a similar ability to stimulate salivary function, prophylactic treatment did not prevent mucositis or candidiasis in head and neck cancer patients undergoing radiotherapy (
13).
In 2004, the Multinational Association of Supportive Care in Cancer and the International Society for Oral Oncology (MASCC/ISOO) offered a consensus statement with recommendations on treatment-associated mucositis.
In the setting of radiation therapy, the MASCC/ISOO advocated the use of benzydamine, a nonsteroidal antiinflammatory that inhibits TNF-α (not available in the United States), and did not support the use of chlorhexidine as a prophylactic for mucositis (
5).
In patients treated with chemotherapy, the prophylactic use of acyclovir was not recommended. Cryotherapy was not advocated in the setting of treatment with 5-fluorouracil, with or without leucovorin. Additionally, the panel did not support the prophylactic use of pentoxifylline or low-level laser therapy in patients preparing for stem cell transplant (
5).
Novel agents requiring additional evaluation include Gelclair, AES-14, and human keratinocyte growth factor-1 (hKGF-1) and hKGF-2. Gelclair, a mixture of polyvinylpyrrolidone, sodium hyaluronate, and glycyrrhetinic acid, has demonstrated improved pain scores during treatment yet clinical trial data were lacking. AES-14, a proprietary form of administering L-glutamine, demonstrated the capacity to decrease grade 2 or greater mucositis when compared to placebo. Repifermin (hKGF-2) was shown to reduce grade 2 to 4 mucositis in clinical trials, and head and neck cancer patients receiving palifermin (hKGF-1) experienced a lower incidence and shorter duration of mucositis when compared to placebo (
5).
The Cochrane Collaboration reviewed 131 studies with over 10,000 randomized patients in assessing treatment options for the prevention of oral mucositis in patients receiving cancer treatment. Less than 10% of the studies were considered to have a low risk of bias associated with their performance. The therapies shown to offer some benefit compared to placebo included: aloe vera, amifostine, cryotherapy, granulocyte-colony stimulating factor, intravenous glutamine, honey, keratinocyte growth factor, polymyxin/amphotericin antibiotic paste, and sucralfate. The strength of the evidence was described as variable (
14).
Graft-Versus-Host Disease
Acute GVHD is dependent upon type I cytokine-driven CD8 effectors, while chronic graft-versus-host disease (cGVHD) has traditionally been associated with type II CD4 T cells (
15). The median onset of GVHD is 6 months after transplantation with oral manifestations seen in up to 80% of patients.
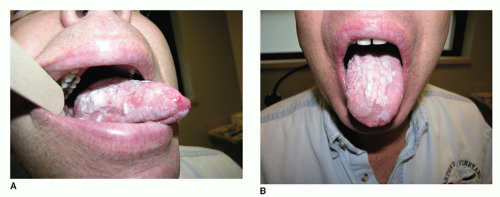
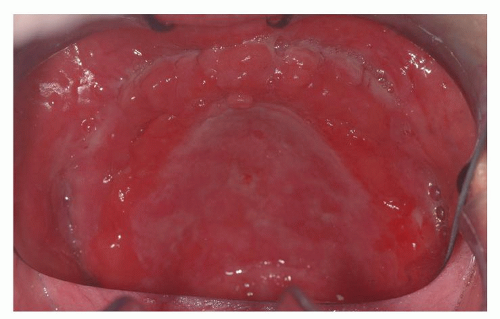
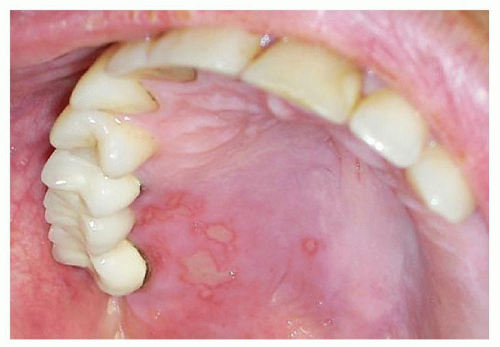
The clinical presentation includes xerostomia, oral ulceration, altered capacity for taste, and recurrent candidal infections (
16) (
Fig. 51.1A and B). The clinical appearance can mimic oral squamous carcinoma. Of note, patients who have undergone hematopoietic stem cell transplant are at increased risk of developing oral squamous cell carcinoma and should have regular surveillance exams with biopsy performed for suspicious lesions (
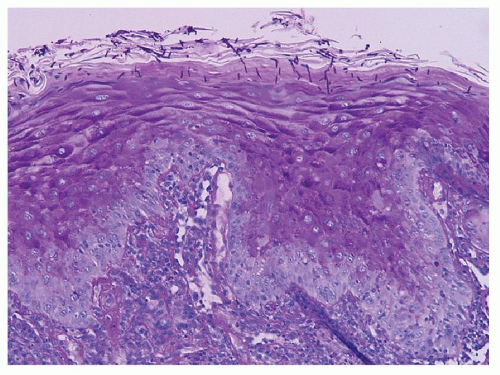
17). Histologic findings of GVHD on biopsy include hydropic degeneration of the basal layer of the epithelium, intracellular edema, apoptotic bodies, fibrosis, and lymphocytic infiltrate in the submucosa (
18).
The current therapy for cGVHD is nonspecific immunosuppression.
Contrary to conventional belief, Imanguli et al. believe that cGVHD is not a type II cytokine driven disorder. They advocate a model that oral cGVHD results from type I interferon (IFN)-driven proliferation and differentiation of Th1/Tc1 differentiated effectors. This concept was supported by the finding of T cells in cGVHD infiltrates expressing T-bet, a transcription factor critical for Th1/Tc1 polarization and the production of IFN-γ and granzyme B. With these new findings, the authors suggest that new treatment approaches for cGVHD should focus on affecting the IFN-mediated inflammatory axis (
15).
Lichenoid Reactions
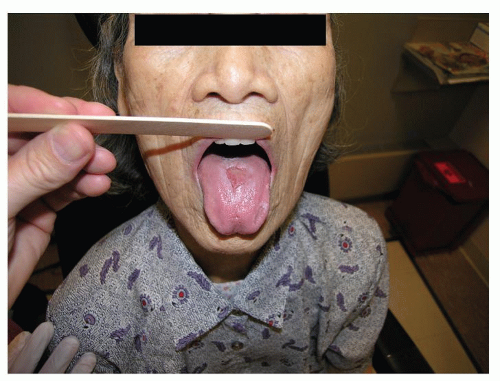
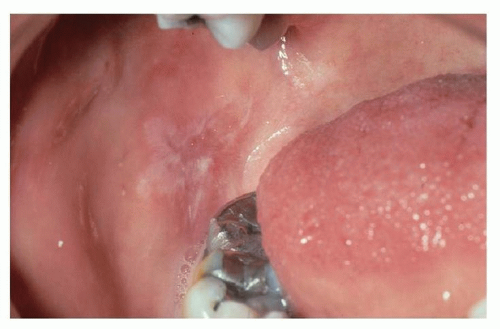
Lichenoid reactions of the oral cavity represent an immunologic response to a systemic or local stimulus. Reactions can present as a contact stomatitis, as seen in the buccal mucosa opposing a dental amalgam (
Fig. 51.2), or may represent a drug reaction from the initiation of a common medication. An extensive number of oral medications have been associated with lichenoid drug reactions (
Table 51.1). The most common agents responsible for oral lichenoid drug reactions are nonsteroidal anti-inflammatories, antihypertensives, and HIV antiretrovirals. Presentation of a lichenoid reaction can vary widely but typically occurs within 2 to 3 months after initiation of a medication (
19).
In addition to dental amalgam, contact lichenoid stomatitis has been reported with cinnamon flavoring (as a result of cinnamic aldehyde) and other flavorings commonly found in mints and mouthwashes. A history of a recent change in the use of oral hygiene products or variation in the habitual use of a mint or gum brand may help elucidate this risk factor.
Individuals with contact allergy to inorganic mercury amalgam present with an oral mucosal lesion within 1.0 cm of the suspected offending restoration, and they lack associated cutaneous lichen planus (CLP). Patch testing to inorganic mercury has been advocated for recalcitrant presentations and should be read on days 7, 10, and 14 in an effort to identify late reactions. In the setting of a positive patch test, subsequent improvement of oral lesions is highly likely with removal and replacement of the restoration. Contact lichenoid reactions are not unique to mercury and may also occur to gold and palladium (
20,
21).