Chapter 23 Sporadic Medullary Thyroid Carcinoma
Introduction
Medullary thyroid carcinoma (MTC) comprises 5% to 10% of all thyroid cancers. MTC arises from the parafollicular or C-cells of neuroendocrine origin. It occurs in hereditary (25% of cases) and sporadic (75%) forms. Sporadic MTCs tend to be more aggressive than hereditary MTCs, and they frequently metastasize to cervical lymph nodes (see Chapters 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, 25, Sporadic Medullary Thyroid Microcarcinoma, 67 Parathyroid management in the MEN syndromes, and 44, Surgical Pathology of the Thyroid Gland). Thorough surgical extirpation of the primary tumor and nodal metastases by compartment-oriented resection has been the mainstay of treatment. Surgical resection of residual and recurrent disease effectively reduces calcitonin levels and controls complications of central neck disease. Radioactive iodine, external beam radiation therapy, and conventional chemotherapy have not been effective. Newer systemic treatments, with small molecule inhibitors, hold promise and are being tested in clinical trials for patients with metastatic disease.
Pathology
C-cells are so named for their secretion of the hormone calcitonin and make up 1% of thyroid cells. C-cells are neuroectodermal in origin and belong to the amine precursor uptake decarboxylation (APUD) cell family. They are primarily concentrated in the posterior upper third of the gland. C-cells normally exist in clusters of six to eight cells at the periphery of thyroid follicles. These cells secrete calcitonin and other substances, including carcinoembryonic antigen (CEA), histaminase, neuron-specific enolase, calcitonin gene-related peptide, somatostatin, thyroglobulin, thyrotropin-stimulating hormone, adrenocortical stimulating hormone, gastrin-related peptide, serotonin, chromogranin, and substance P.1 Calcitonin has been shown to be integral in calcium homeostasis in other vertebrate species, but its role in humans is unclear. C-cell replacement is not necessary in humans following thyroidectomy.
MTC was first described in the beginning of the 20th century in the German medical literature as a malignant goiter with amyloid stroma. For decades, MTC was considered to be a variant of anaplastic thyroid carcinoma, and it was not recognized as a unique entity until 1959 when Hazard, Hawk, and Crile first described its characteristics.2 Grossly, the tumor is well demarcated, firm, gray-white, and gritty. Microscopically, the cells are uniform polygonal cells with finely granular eosinophilic cytoplasm with central nuclei. Varying numbers of spindle cells are present in nearly all tumors. The presence of amyloid is considered to be a distinctive feature of MTC, although it may not necessarily be found in all cases. The amyloid differs from that of other tumors in that it is formed from calcitonin or procalcitonin molecules. In sporadic tumors, approximately 68% are solitary, and 32% are bilateral or multifocal. In familial forms, 94% are bilateral or multifocal, and 6% are solitary. Histologically, the tumors can be classified according to dominant histologic patterns. These groups include the classic, amyloid-rich, insular, trabecular, and epithelial variants. Classic variants are most common (48.9%), followed by the amyloid-rich variants (38.3%).3,4 C-cell hyperplasia is associated with MTC, particularly in the familial forms. C-cell hyperplasia is thought to be a precursor in the malignant transformation to MTC.5,6
Clinical Settings
Medullary thyroid carcinomas occur in two clinical settings: sporadic and hereditary. Familial forms of MTC are usually multifocal and bilateral, and they are discussed elsewhere in this book (see Chapter 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B). Sporadic micromedullary thyroid carcinoma (SmMTC) is also discussed elsewhere in this book (see Chapter 25, Sporadic Medullary Thyroid Microcarcinoma). Much of the MTC literature lumps data on hereditary and sporadic tumors together, making it difficult to compare sporadic and hereditary MTC. Overall, sporadic medullary thyroid carcinomas behave in a similar fashion to hereditary medullary thyroid carcinomas with similiar prognosis when adjusted for stage, although hereditary MTCs are more likely to be bilateral and to present at a less advanced stage.
Sporadic MTC is usually unilateral. As discussed in Chapter 25, Sporadic Medullary Thyroid Microcarcinoma, sporadic MTC may be discovered after detection of elevated calcitonin or CEA levels in patients with a nodular thyroid,7 but it usually presents as a mass in the neck from the thyroid tumor or cervical nodal metastases. Some patients present with distant metastases or with diarrhea secondary to substances (described earlier) secreted by the MTC tumor cells. Rarely, MTC is discovered to be the cause of an elevated CEA level—for example, in a patient being followed after treatment of colon cancer.
Calcitonin Testing
Peripheral serum levels of calcitonin are measured by radioimmunoassay. In patients with MTC, a direct correlation exists between basal plasma calcitonin levels and tumor mass. Provocative calcitonin stimulation tests were routinely performed in the United States in the past (until approximately 2005). The gastric hormone pentagastrin stimulates calcitonin secretion and was widely used (with or without additional calcium infusion) as a secretagogue in the so-called provocative calcitonin stimulation test. In this test, an intravenous infusion of calcium gluconate (2 mg/kg over 1 min) followed by pentagastrin (0.5 mg/kg over 5 secs) is given, and blood samples are obtained before and at 1, 2, 3, and 5 minutes after the infusion. Peak plasma calcitonin values generally occur at 1 to 2 minutes.8,9 Improvements in the sensitivity of immunoradiometric basal calcitonin assays (to < 3 pg/mL), however, and the lack of availability of pentagastrin in the United States have precluded the use of such provocative testing. In addition, the test is very uncomfortable for the patients; they often complain of nausea, diaphoresis, agitation, and urinary urgency. Measurement of basal serum calcitonin levels is adequate for following patients with sporadic MTC.
Routine measurement of calcitonin in patients with nodular thyroid disease has been used in parts of Europe to screen for MTC. This subject is discussed in more detail in Chapter 25, Sporadic Medullary Thyroid Microcarcinoma.
Genetics
The possibility that an apparently sporadic case of MTC is familial should be considered preoperatively. Despite the absence of a family history of MTC, germline mutations in the RET proto-oncogene have been identified in < 10% of cases of apparently sporadic MTC (index cases).10,11 Testing for RET mutations in these patients has several benefits. First, if a patient with apparently sporadic MTC is found to have a germline RET mutation, other family members harboring the same mutation may be identified by genetic testing, starting with first-degree relatives, and appropriate therapy or preventative surgery can be initiated. Second, if a patient is found to have a germline RET mutation, he or she may then be screened for hyperparathyroidism and pheochromocytoma. Third, the type of RET mutation present has prognostic significance, relative to the clinical behavior of MTC. The specific RET mutation predicts age of onset, approach to surgical management, risk of recurrence, and association of other endocrine neoplasms. We recommend therefore that all patients with a personal medical history of primary C-cell hyperplasia, MTC, or MEN 2 should be screened for germline RET mutations (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia).
Somatic mutations are mutations that are identified in tumor cells only and not in all cells of the body, as are germ line mutations. It is common for sporadic cancers to have somatic mutations in the same gene that is mutated as a germline mutation in the hereditary form of that cancer (e.g., APC mutations in sporadic colon cancers). Somatic mutations or rearrangements involving RET have been identified in 40% to 50% of sporadic MTCs and up to 70% of sporadic papillary thyroid carcinomas (PTCs).12,13 Most of the mutations identified in sporadic MTCs are point mutations involving the same codons associated with the MEN 2 syndromes, including 918, 634, and 883.12 Of sporadic MTCs with alterations of RET, 60% to 80% are found to have the M918T mutation.12,14–16 Patients with sporadic MTCs bearing a RET mutation (particularly M918T) have a more advanced stage at diagnosis, increased rates of recurrent or persistent disease after resection, and poorer long-term survival (10 to 20 years) than those without this RET mutation.12,15,16 In addition, sporadic MTCs are heterogeneous with regard to the distribution of RET mutations. Molecular studies of sporadic MTCs demonstrate that some cell subpopulations within a given primary tumor or its metastases have a RET mutation, whereas others do not.17,18 This suggests that the RET mutation may be a later event in tumorigenesis, occurring in subclonal populations of an already established MTC. It is unclear what survival advantage, if any, is conferred on these subgroups of cells by the presence of a RET mutation, and it is unclear whether RET mutations are actually responsible for the poorer prognosis seen clinically.
Clinical Course
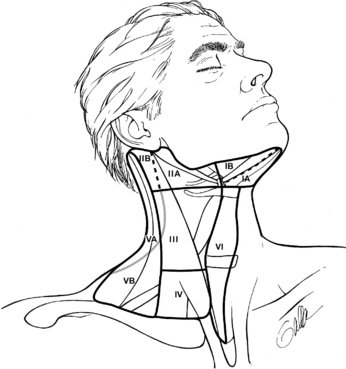
Patients with sporadic MTC usually present with a palpable thyroid or neck mass. Symptoms of dysphagia, shortness of breath, or hoarseness are present in approximately 15% of cases. MTC frequently metastasizes to regional lymph nodes, and in patients with palpable disease, these metastases are often apparent at the time of diagnosis. The frequency of nodal metastases has been reported to be over 50% in patients who present with palpable, established primary tumors.19–23 Spread is most frequent to the central compartment (level VI), followed by the ipsilateral jugular chain of nodes (levels II through V) and the contralateral cervical nodes. Spread is also seen to the upper and anterior mediastinum (Figure 23-1).19,24 Hematogenous spread may occur to the lungs, liver, bones, brain, and soft tissues. Metastatic spread may be difficult to assess by imaging because of the fine miliary pattern of these deposits. Laparoscopy with direct imaging of the liver may identify small metastatic deposits not visible by conventional imaging in 25% of patients with elevated calcitonin levels following therapeutic surgery.25
MTC is a relatively indolent malignancy, with reported 10-year survival rates from 69% to 89%.26–28 Higher long-term survival rates are reported in patients who have normalization of serum calcitonin levels after surgery. The American Joint Committee on Cancer defines four stages of disease in MTC (Table 23-1, parts A and B). The different stages take into account tumor size, evidence of regional lymph node or distant metastases, and tumor invasion. The prognosis for MTC includes age and stage. In one of the earlier studies, 10-year cause-specific survival was 71%. Of the 53 patients with MTC, the mean age at diagnosis was 46 and distribution of familial and sporadic MTC was 17% and 83%, respectively. Prognosis was most influenced by stage and postoperative basal calcitonin levels correlated most strongly with survival.29 In a later review of 104 patients of which 44% had hereditary MTC, cause-specific survival was 89%. By univariate analysis, age, stage, gender, distant metastases, extent of surgery were all significant prognostic factors. Only age and stage, however, were statistically significant by multivariate analysis.30 This was confirmed in a more recent review by Rendel et al. where again the most sensitive predictors of survival were age at diagnosis and tumor stage.31 In this report, 32 patients with MTC treated over a 42-year period (with a median follow-up of 9.5 years) had a cause-specific survival of 84.4%. The estimated mean survival time from the time of diagnosis was 31years. There was, however, a difference in survival time based on whether patients achieved biochemical and radiographic remission. In those who did not achieve remission, 10-year survival was slightly reduced to 73%. This observation demonstrates the indolent nature of the disease, the appropriateness of reoperative surgery when technically possible, and the usefulness of cytostatic agents that keep clinically occult disease under control.
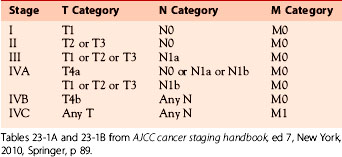
Table 23-1A American Joint Committee on Cancer TMN Criteria for Medullary Thyroid Cancer
| TMN | Criteria |
|---|---|
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Tumor ≤ 2 cm, limited to the thyroid; T1a: tumor ≤ 1 cm; T1b: tumor > 1 cm but ≤ 2 cm |
| T2 | Tumor > 2 to ≤ 4 cm, limited to the thyroid |
| T3 | Tumor > 4 cm, limited to the thyroid or minimal extrathyroidal extension |
| T4 | T4a: tumor of any size extending beyond the thyroid capsule to invade subcutaneous soft tissues, larynx, trachea, esophagus, or recurrent laryngeal nerve; T4b: tumor invades prevertebral fascia or encases carotid artery or mediastinal vessels |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Regional lymph node metastasis; N1a: metastasis to level VI (pretracheal, paratracheal, and prelaryngeal lymph nodes); N1b: metastasis to unilateral, bilateral, or contralateral cervical or retropharyngeal or superior mediastinal lymph nodes |
| MX | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
Diagnosis
Familial forms of MTC usually present with multifocal, bilateral disease. Patients with sporadic MTC (sMTC) more commonly have unifocal disease, later age of onset, and absence of C-cell hyperplasia.32–34 Every patient presenting with newly diagnosed MTC should be considered to have familial MTC until proved otherwise. Thus, MTC patients should have a full family history taken at the time of initial consultation. Physical exam should take note of size of palpable neck nodules, fixation to surrounding structures, and the presence of cervical lymphadenopathy. Characteristic features of the MEN 2B phenotype should also be noted. Symptoms of patients with extensive localized involvement include dysphagia, hoarseness, dyspnea, and coughing. Direct examination of the vocal cords prior to surgical intervention is warranted to document possible involvement of the recurrent laryngeal nerve. Patients presenting with elevated levels of calcitonin may exhibit diarrhea as the initial symptom of their disease. Lichen planus amyloidosis may be an early indicator of MEN 2A and should prompt genetic testing and appropriate clinical workup. The dominant nodule should be evaluated first with fine-needle aspiration (FNA) aided by immunocytochemical staining for calcitonin. FNA for the diagnosis of MTC has certain pitfalls. FNA is generally successful in diagnosing MTC in approximately > 80% of patients. In the remaining patients, the cytopathologic diagnosis assigned still typically warrants surgical intervention (i.e., follicular neoplasm, suspicious for MTC).
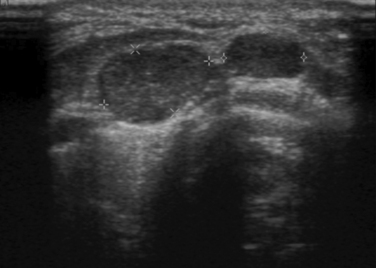
Preoperative evaluation of patients with suspected MTC (i.e., FNA or elevated calcitonin level) should include serum calcitonin measurement, carcinogenic embryonic antigen (CEA), serum calcium, and RET proto-oncogene analyses. Biochemical screening for pheochromocytoma should be conducted in any MTC patient more than 10 years of age. Preoperative imaging includes a neck ultrasound with lymph node mapping of the cervical and superior mediastinal nodal compartments (Figure 23-2). The sensitivity of intraoperative palpation to detect lymph node metastases by experienced surgeons is only 64%.35 In patients presenting with a palpable thyroid nodule, cervical lymph node metastases are common (> 75%), with 10% to 15% of these patients also having evidence of distant metastases.35 MTC most commonly metastasizes to bone, liver, and lung. Detection of distant disease begins with computed tomography (CT) of neck, chest, and abdomen. CT is the most sensitive test to detect lung and mediastinal lymph node metastases.36 Contrast enhanced magnetic resonance imaging (MRI) is most sensitive for the detection of liver metastases and bone metastases are seen best on either axial MRI or bone scan. In one series, CT has been found to be superior to FDG-PET for lung, liver, and bone metastases.37 However, FDG-PET was more sensitive than CT in detecting neck and mediastinal disease. Imaging to identify possible distant metastasis is not indicated in every MTC patient. Current evidence suggests that imaging for evidence of metastatic disease should be reserved for patients with either local lymph node metastases or those with a basal serum calcitonin of 150 to 400 pg/mL or greater.38
On computed tomographic imaging, MTCs appear as nodules with calcifications and may demonstrate extrathyroidal disease. Fine-needle aspiration of the palpable thyroid nodule or cervical lymph node metastasis is a sensitive means for establishing the diagnosis of MTC. Ultrasound examination of the neck is a sensitive technique for identifying cervical lymph node metastases.39
Surgical Treatment
The surgical strategy for MTC has evolved significantly since the early 1990s. In the late 1990s, the most common procedure performed for sporadic MTC was total thyroidectomy without lymph node dissection (based on cohort data on patients with thyroid cancer treated in 1996 collected by the American Cancer Society’s Commission on Cancer).28 It is now clear that a systematic lymph node clearance is optimal in all MTC cases (except very early familial cases in a screening setting with low calcitonin levels).40 It is the author’s perception that management of MTC in the United States has changed significantly since the 1990s, and now most patients are treated by total thyroidectomy and some form of node dissection.
The surgical treatment of MTC is influenced by several factors: (1) the clinical course of MTC is usually more aggressive than that of differentiated thyroid cancer, with higher rates of recurrence and mortality, especially in young patients; (2) MTC cells do not take up radioactive iodine; (3) hormone suppression is ineffective; (4) MTC is multifocal and bilateral in 90% of patients with hereditary forms of the disease and in 20% of patients with the sporadic form; (5) nodal metastases are present in more than 70% of patients with palpable disease; and (6) the ability to measure postoperative calcitonin levels allows assessment of the adequacy of surgical extirpation. Recently published guidelines for clinicians contain recommendations for management of MTC in multiple relevant clinical settings that will be useful to clinicians treating these patients.41
In patients with palpable MTC (sporadic or hereditary), total thyroidectomy is the appropriate treatment of the primary tumor, accompanied by a central node dissection. Calcitonin levels are virtually always elevated in these patients (see Chapter 38, Central Neck Dissection: Technique). Classification and methods of central neck lymph node dissection have been described by several groups.35,40,42–44 In this operation, all thyroid and nodal tissue is removed from the level of the hyoid bone superiorly to the innominate vessels inferiorly. A systematic (compartment-oriented) approach to the removal of all nodal tissue in the central neck has been reported to improve recurrence and survival rates when compared retrospectively with procedures in which only grossly involved nodes were removed.42 Preoperative ultrasonographic evaluation of neck nodes is helpful. Suspicious nodes are marked (skin marking is usually adequate), and identified nodal compartments are removed at operation. CT and fluorodeoxyglucose positron emission tomography may be useful in these patients; however, ultrasound allows a more detailed evaluation of nodal architecture and allows for easier localization, biopsy, and marking in the perioperative setting.39,45
The likelihood of lateral compartment lymph node involvement (levels 2 to 4) is related to the presence and extent of nodal disease in the central compartment, though “skip metastases” have been reported.35,46 In one study, the presence of 0, 1 to 3, or ≥ 4 central lymph node metastases was correlated with 10.1%, 77%, and 98% risk of metastatic involvement of ipsilateral levels 2 to 4 nodes, respectively. For contralateral lateral compartment involvement, the rates were 4.9%, 28%, and 77% with no central lymph node metastases, 1 to 9, and ≥ 10 respectively.47 In a report from our institution, the ability of the surgeon to detect lymph node metastases intraoperatively was evaluated in 73 patients.35 In this series, metastases were present in 80% of central nodes, 75% of ipsilateral jugular nodes (levels 2, 3, and 4), and 47% of contralateral jugular nodes. Of note in this series, intraoperative palpation of nodes was not an accurate predictor of the presence or absence of metastases in a nodal compartment. The sensitivity of intraoperative assessment by the surgeon was only 64%, and the specificity was 71%. Therefore, reliance on intraoperative assessment would miss involved nodes 36% of the time. Preoperative imaging, with ultrasound in particular, is helpful in delineating the extent of nodal spread and in planning the extent of node dissection, as has been described in differentiated thyroid carcinoma.48 Thus, in patients with preoperative imaging suggesting central lymph node metastases, serious consideration should be given to doing a prophylactic ipsilateral lymph node dissection (LND). In patients with clinically apparent, palpable sporadic MTC, total thyroidectomy with compartment-oriented neck dissection appears to be most effective at achieving long-term control and cure of disease.40,49
Preservation of parathyroid function is critically important. Most surgeons preserve the blood supply of some parathyroids and leave those glands in situ. Preservation of devascularized glands may be accomplished by removal and autotransplantation of the minced parathyroid fragments to the sternocleidomastoid muscle or to a forearm muscle (all parathyroid tissue should be left in the patient.)50–53 In the past, our group routinely performed four-gland parathyroidectomy with autotransplantation, arguing that adequate bilateral central node dissection is not possible if the parathyroid glands are left in place with sufficient blood supply. Reported rates of long-term hypoparathyroidism are quite low with this approach.9 Our recent approach has been to resect and autotransplant the two parathyroids on the side of the primary tumor, as well as the contralateral lower parathyroid, leaving the contralateral upper parathyroid in situ on a vascular pedicle if possible. All removed parathyroids are minced into 1 × 3 mm fragments, and the fragments are transplanted into individual muscle pockets (two to three fragments per pocket), which are then closed with a suture.54 Transplantation of whole minced glands into a single pocket is discouraged. Parathyroid fragments may be transplanted into individual muscle pockets in the sternocleidomastoid muscle in cases of sporadic disease, FMTC, or MEN 2B. They may be transplanted into the nondominant forearm in cases of MEN 2A when there is a risk of future hyperparathyroidism (e.g., codon 634 RET mutation carriers). The reason for this difference is the risk of subsequent graft-dependent hyperparathyroidism in some MEN 2A patients, which is more easily localized and treated if the grafts are in the forearm (see Chapters 67, Parathyroid Management in the MEN Syndromes, and 68, Reoperation for Sporadic Primary Hyperparathyroidism).
Overall risk of recurrent laryngeal nerve injury during thyroid cancer operations is approximately 1.3%, per the most recent cohort data from the American College of Surgeons Commission on Cancer Patient Care Evaluation Study. This ranges from 0.7% without lymph node dissection (LND) to 2.7% for total thyroidectomy with LND. The risk of temporary hypocalcemia after total thyroidectomy ranged from 12% without LND to 14% with LND.28 This study did not measure the risk of long-term hypocalcemia, but it is low in experienced hands.
Table 23-2 summarizes several series of compartment-oriented lymphadenectomy for MTC. The reports provide level IV evidence, and a grade C recommendation is made for compartment-oriented dissection with total thyroidectomy for clinically established, palpable sporadic MTC.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree