Secondary Intraocular Lens Implantation
Neil J. Friedman
Timothy T. Khater
Douglas D. Koch
The role of secondary intraocular lens (IOL) implantation has evolved with advances in cataract surgery. Traditionally, most patients who required secondary IOL implantation were either elderly aphakes who had previously undergone intracapsular cataract extraction, or aphakes who had lensectomy during early childhood for congenital cataracts. Individuals also may be left aphakic after complicated cataract surgery, lensectomy after trauma, or removal of a dislocated crystalline lens. For these patients, secondary implantation of an IOL generally is recommended when traditional spectacle or contact lens correction of aphakia is unsuccessful. For bilateral aphakia corrected with aphakic spectacles, surgery is indicated when the patient cannot readily cope with the optical distortions produced by the glasses. For the unilateral aphake, spectacle correction usually is intolerable because of the large amount of anisometropia. Contact lenses can reduce the aberrations and aniseikonia produced by aphakic spectacles. However, many patients are unable to wear contact lenses because of an inability to handle or care for the contact lens, difficulty in fitting the lens, discomfort, contact lens-related complications such as giant papillary conjunctivitis or poor motivation for proper use.
For patients unable to use these devices, various surgical procedures have been investigated, including secondary IOL implantation, epikeratophakia,1 and intracorneal implants.2 The major concern with all of these approaches is the risk of vision-threatening complications. Epikeratophakia and corneal inlays produced disappointing results because of irregular corneal surface changes and poor refractive predictability.3,4,5,6
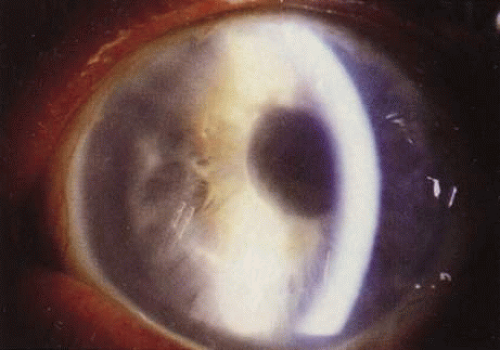
Early results of secondary IOL implantation were mixed. Several reports documented excellent outcomes after secondary posterior chamber intraocular lens (PC IOL) implantation in patients with intact posterior capsules.7 Good short-term results were initially reported with early closed-loop anterior chamber intraocular lenses (AC IOLs), but unfortunately, numerous complications, including uveitis, glaucoma, hyphema, cystoid macular edema, endothelial cell loss, and corneal decompensation (Fig. 1), later developed in many eyes receiving lenses of this type.7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32 The newer, open-loop, one-piece flexible design (i.e., Kelman multiflex style) has eliminated nearly all of these complications, and, as a result, has gained growing acceptance.33,34,35,36,37 As an alternative option for eyes without sufficient capsular support, various lenses and surgical techniques have been developed for implanting iris-supported (e.g., lobster-claw lens)38,39,40,41,42,43,44 and sulcus-sutured posterior chamber lenses.
Because of advances in primary cataract surgery, surgical aphakia is becoming an increasingly rare condition, and secondary intraocular lens insertion is now most commonly performed as part of an IOL exchange procedure. This is indicated for mechanical, inflammatory, or optical IOL-related complications.45 In addition, secondary IOLs are sometimes implanted in combination with other procedures, such as pars plana lensectomy for the treatment of a dislocated crystalline lens, penetrating keratoplasty, or trabeculectomy.
For patients with adequate posterior capsular support, we believe that implantation of a PC IOL into the capsular bag or ciliary sulcus most often is the preferred approach. However, debate continues regarding which of several methods of secondary IOL implantation is superior for eyes lacking sufficient capsular support. Because a clear-cut answer does not exist, we compare the advantages and disadvantages, surgical techniques, and results of studies of IOL implantation with each of these methods.
PREOPERATIVE SCREENING AND SELECTION OF INTRAOCULAR LENS TYPE
The health of the eye is critical in deciding which patients are candidates and, in some instances, which type of lens is to be implanted. Conditions such as corneal endothelial abnormalities, glaucoma, and diabetes require thorough investigation before the insertion of any type of lens. Cystoid macular edema (CME) is generally a contraindication for secondary IOL implantation, unless the CME is caused by a problematic IOL and the problematic IOL is to be repositioned or exchanged.
The peer-reviewed literature suggests that otherwise healthy eyes without capsular support can receive an anterior chamber, iris-supported, or sutured posterior chamber lens with equally satisfactory results.33,46,47,48,49 The surgeon’s experience and comfort with each procedure usually determines which one is chosen; however, some surgeons believe that patients younger than 50 years of age generally should not receive an AC IOL. This age limit is arbitrary and varies among surgeons. However, the long-term biostability of the polypropylene sutures required for sutured PC IOL implantation is uncertain.50
During the preoperative evaluation, it is important to perform meticulous slit-lamp biomicroscopy, funduscopy, and, where indicated, gonioscopy to evaluate any ocular pathology that might affect surgical planning. In addition, aphakes are commonly chronic contact lens users, and an extended period of contact lens disuse is indicated for optimal refractive results. We currently recommend discontinuing contact lenses for at least 1 to 2 weeks for soft contact lens wearers, and 2 to 4 weeks or longer for rigid gas permeable contact lens (RGP) wearers. A more conservative approach is to discontinue lens wear 1 month for each decade of RGP wear. Corneal stability is then verified by serial topography, keratometry, and refraction. Factors to assess include the following:
Best spectacle-corrected visual acuity
Corneal astigmatism
Corneal endothelial status
Anterior chamber (depth, cell, and flare)
Anterior chamber angle (peripheral anterior synechiae, recession, and neovascularization)
Iris and pupil (size, reactivity, peripheral iridectomies, and anterior and posterior synechiae)
Posterior capsule (adequacy of support and clarity)
Presence of vitreous in anterior segment
Macula (cystoid macular edema, macular degeneration)
Peripheral retina
In some cases, specialized testing such as corneal pachymetry, specular micrography, optical coherence tomography, and fluorescein angiography may be indicated.
The timing of secondary IOL surgery also must be considered. In one study,51 the risk for CME was increased if secondary IOL implantation was performed within 1 year of intracapsular cataract extraction; unfortunately, there are no comparable data on the timing of surgery after extracapsular procedures.
The status of the patient’s fellow eye is obviously important. Does it have good vision? Does it have any sight-threatening conditions? Either of these factors might make one more cautious in performing intraocular surgery in the fellow eye. What is the refractive error of the fellow eye?
Furthermore, the patient’s overall health may contribute to the surgical decision. Because implanting an anterior chamber lens generally is faster than suturing an IOL into the sulcus, an AC IOL may be chosen for a patient who may not be able to tolerate a lengthy procedure. Similarly, an anticoagulated patient may be at greater risk for intraocular hemorrhage from needle passes through the iris or ciliary sulcus and sclera; therefore, a safer approach might be to insert an AC IOL.
Finally, the technique of choice is often determined at the time of surgery and is often a matter of clinical judgment. Intraoperative findings that alter the surgical approach include the adequacy of zonular support, posterior iridocapsular adhesions, status of the anterior and posterior capsular remnants, and the presence of lenticular pieces that were not visible during the preoperative examination. The surgeon should be well versed in multiple techniques, in case one or more techniques are surgically contraindicated.
POSTERIOR CHAMBER INTRAOCULAR LENSES
Clearly, if the preoperative eye has adequate capsular support, the preferred site for secondary IOL implantation is the capsular bag or the ciliary sulcus. We believe that the optimal lens for secure sulcus fixation is a large optic (6.5 mm or more), one-piece polymethyl methacrylate (PMMA) IOL with an overall diameter of 13.5 to 14 mm. However, a foldable IOL with a 6-mm optic and 13-mm overall length allows insertion through a smaller wound and is acceptable if the IOL is stable. Unfortunately, many patients requiring a secondary IOL lack adequate zonular or capsular support for either of these procedures. Sulcus implantation may be contraindicated in patients with a history of pigment dispersion from previous posterior chamber lens implantation or with an excessively large sulcus that might not permit firm haptic fixation. Options in these patients might include suture fixation of the posterior chamber lens to the sclera or iris, an iris-supported lens, or an anterior chamber IOL. In rare instances, if the capsular bag is intact and can be reopened but lacks adequate zonular support, a capsular tension ring (CTR) with or without fixation sutures can be inserted to enable implantation of an unsutured posterior chamber lens.52,53,54,55,56
Various techniques of posterior chamber IOL fixation have been tried in patients who were poor candidates for an anterior chamber lens and who lacked sufficient capsular support. There has been a recent resurgence of interest in the use of iris-suture lenses. Advantages of this approach include: (a) Avoidance of the risks associated with needle passes through the sclera and tend to spare the anterior chamber angle,39,40,41,44 (b) ability to use a foldable IOL and small incision, and (c) greater ease compared to suturing a lens into the ciliary sulcus. Fixation to the peripheral iris is most commonly accomplished with a modified McCannel retrievable suture, although a number of closed and open-sky techniques have been described.57,58,59,60,61 Complications reported with this method of IOL fixation include pigment dispersion, uveitis, glaucoma, pupillary block, iris/pupil distortion, peripheral anterior synechiae (PAS), hyphema, CME, and retinal detachment (RD).62,63 However, comparative studies suggest that the results with iris-fixation match or exceed those with anterior chamber or scleral-sutured IOLs.
A more popular method of suture fixation has been transscleral suturing. A variety of techniques and lens modifications have been reported for transsclerally suturing PC IOLs. Gess64 first described scleral fixation of one haptic of a posterior chamber lens. In 1986, Malbran and colleagues65 described an open-sky technique for sutured PC IOLs, and in 1988, Cowden and Hu66 reported secondary PC lens implantation with scleral fixation of both haptics through scleral stab incisions.
Advantages of transsclerally sutured IOL fixation include the elimination of corneal and angle trauma associated with anterior chamber lenses, decreased risk of pupillary block and secondary glaucoma, and little or no IOL contact with the iris, thereby decreasing the risk of iritis, pigment dispersion, and CME.9,13,24,25,29,67,68,69,70 The sulcus location most closely approximates the normal anatomic position of the crystalline lens, and this method of PC IOL fixation minimizes the risk of pseudophakodonesis.20 In addition, transsclerally sulcus–sutured posterior chamber IOLs theoretically can be used in any age group, including children,71,72,73 because the lenses are more likely than anterior chamber lenses to accommodate to the growing eye; however, long-term results are unknown.
Important drawbacks to this type of lens fixation need to be considered. Compared with AC IOL insertion and iris-sutured PC IOLs, the procedure is technically more difficult, requiring longer surgical time and a thorough anterior vitrectomy, both of which might increase the risk of intraoperative and postoperative complications. In addition, it is difficult to precisely and symmetrically fixate both haptics within the ciliary sulcus.74,75,76 The anatomy of this space, which averages 11.0 ± 0.37 mm in diameter77 and is located approximately 0.83 mm and 0.46 mm posterior to the surgical limbus in the vertical and horizontal meridians,78 respectively, may be altered in long-standing aphakia.75 In Pavlin’s ultrasound biomicroscopy study of 34 transsclerally fixated PC IOLs, 13 IOLs were in the ciliary sulcus, 8 were posterior, and 13 were anterior.74 Manabe also used the ultrasound biomicroscope to identify haptic placement and found only 32 of 86 haptics sutured at the sulcus; there were 29 at the ciliary processes, 25 posterior to the pars plicata, and 41 with vitreous incarceration.76 Additionally, in a clinical study comparing complications of AC IOL versus transsclerally fixated PC IOL implantation, Bellucci showed that six of 32 PC IOLs were in the sulcus, 24 were in the pars plana, and 2 were in the iris root.75 We suspect that in many instances one or both haptics may not be properly positioned in the ciliary sulcus, but this is not noted clinically because of the excellent stabilization provided by the sutures.
Despite its anatomic advantages, PC IOL implantation into the ciliary sulcus is associated with complications, primarily because of haptic contact with uveal tissue and the need for haptic fibrosis to ensure long-term stability. Even without suture fixation, sulcus placement of a lens implant carries the risks of lens decentration (Fig. 2), pigment dispersion, uveitis, recurrent hemorrhage (Fig. 3), ciliary body erosion,79,80,81,82,83,84 and, in one reported case, occlusion of the major arterial circle of the iris (located in the ciliary body), with devastating results.70 Transscleral fixation introduces additional risks caused by needle penetration of uveal and scleral tissue, abnormal positioning of the haptics, and external suture exposure; these risks include lens tilt and decentration (Fig. 4), lens subluxation, episcleritis, corneal decompensation, hypotony, PAS formation, secondary glaucoma, hyphema, vitreous hemorrhage, suprachoroidal hemorrhage, choroidal effusion, CME, RD, external suture erosion (Fig. 5), and endophthalmitis.43,47,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101
 Fig. 2. Inferior subluxation (sunset syndrome) of a sulcus-fixated posterior chamber intraocular lens. |
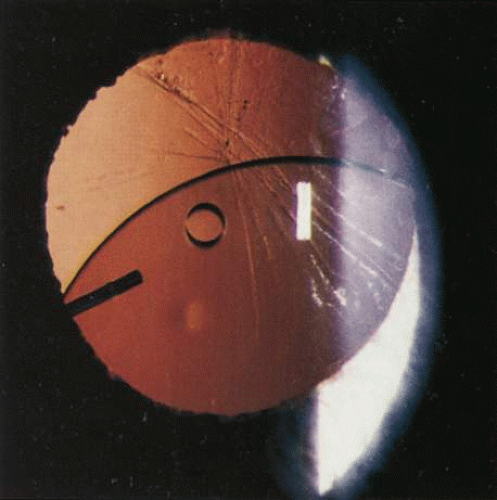 Fig. 3. Recurrent iritis and hyphema associated with a loose posterior chamber intraocular lens in the ciliary sulcus. Note fine dusting of red blood cells on endothelial surface. |
 Fig. 4. Mild inferior decentration of a transsclerally sutured intraocular lens; the fixation sutures were not placed 180 degrees apart. |
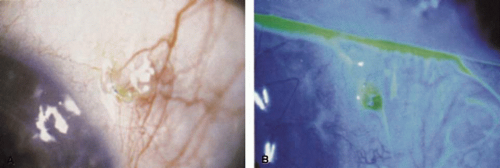 Fig. 5. A. External suture erosion and exposure after placement of a transsclerally fixated posterior chamber intraocular lens. B. Note fluorescein pooling around exposed suture ends. |
A variety of modifications of surgical technique and lens design intended to reduce some of these risks have been described.39,47,63,86,93,94,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119 Goals have included limiting the time the eye is open (to reduce the risk of intraoperative complications), decreasing the number of needle passes through the sclera (to reduce the risk of intraocular bleeding), burying the suture knots (to reduce the risk of suture erosion and subsequent endophthalmitis), using direct visualization (endoscopic or mirror needle holder to ensure more accurate placement of the haptics in the sulcus), and, perhaps most importantly, suturing with stronger material and double fixation (9-0 Prolene or 8-0 Gore-Tex to reduce the risk of suture breakage and lens subluxation).102,120,121,122,123
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree