Secondary Angle-Closure Glaucoma
Asha Yancy Okorie
Eydie Miller-Ellis
Husam Ansari
In contrast to primary angle closure, in which angle closure can be thought to result from a congenital but normal anatomic variant or the natural history of said anatomic variant, secondary angle closure (SAC) results from an acquired, identifiable disease state. SAC can be devastating as patients can suffer vision loss not just from glaucomatous optic neuropathy (GON), but also from the other sequelae of the underlying disease that is causing the angle closure.
SECONDARY ANGLE-CLOSURE DEFINITIONS
As with primary angle closure, SAC suffers from a history of inconsistent and inadequate nomenclature. It can be helpful to subtype SAC phenomenon similarly to their primary counterparts (Table 54.1). The secondary angle-closure suspect (SACS) is a patient with likely appositional contact between the peripheral iris and the trabecular meshwork, but without the stigmata of angle damage (i.e., elevated intraocular pressure (IOP) or peripheral anterior synechiae (PAS). This can be seen in uveitic patients with angle closure due to pupillary block resulting from posterior synechiae between the pupil margin and the anterior lens surface. Another example is the patient with a peripheral iris or ciliary body mass causing appositional contact. The patient with SAC has an underlying disease state that has resulted in PAS formation or appositional contact with elevated IOP. Finally, the patient with secondary angle-closure glaucoma (SACG) has an underlying disease that has resulted in SAC and GON.
TABLE 54-1 Secondary Angle-Closure Definitions | ||||||
|---|---|---|---|---|---|---|
|
SECONDARY ANGLE CLOSURE MECHANISMS
SAC is often divided into anterior and posterior mechanisms that pull or push, respectively, the peripheral iris toward the trabecular meshwork. Table 54.2 lists these anterior “pulling” and posterior “pushing” mechanisms.
NEOVASCULAR GLAUCOMA
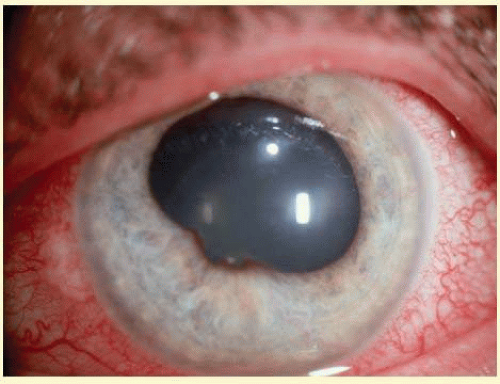
Neovascular glaucoma (NVG) arises when new vessels form on the iris, then grow over the trabecular meshwork, and obstruct aqueous humor outflow (Fig. 54.1). Early on, this condition is an open-angle glaucoma, but over time the fibrovascular tissue grows to cover the angle in a sheetlike manner then contracts leading to synechial closure of the anterior chamber angle. This is usually followed by a dramatic increase in IOP, ocular pain, and decreased vision. There are several causes of neovascularization of the iris (NVI). NVG is most commonly related to posterior segment disease, specifically conditions involving a hypoxic retina. In hypoxic states, angiogenic factors such as VEGF are produced which stimulate new blood vessel growth in the anterior segment. Chronic uveitis may also cause neovascularization.
TABLE 54-2 Mechanisms of Secondary-Angle Closure | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Patients with proliferative diabetic retinopathy are at risk for NVI. The reported incidence of NVI in this population is 50%.1 Surgery affects onset of NVI in diabetics with retinopathy. Studies have shown the incidence of NVI in diabetics to be higher after pars plana vitrectomy for diabetic retinopathy when compared to the incidence of NVG in the same population.2
Treatment of NVG requires not only lowering the IOP, but also treating the underlying cause of neovascularization. For retinal diseases, this usually involves pan-retinal photocoagulation. It may also include intravitreal injection of anti-VEGF medication. Other modalities have been described, including transscleral diode laser retinopexy, panretinal cryotherapy, and panretinal diathermy.
Wakabayashi and associates reported a retrospective consecutive case series of 41 eyes and examined the efficacy of intravitreal bevacizumab (IVB) for NVI, NVG with an open angle, and NVG with a closed angle. Patients were given 1 mg of IVB as initial treatment and followed for at least 6 months. They found that IVB was effective in decreasing neovascularization in NVI and NVG with an open angle.3 In these eyes, IOP was controlled and the treatment was well tolerated. In patients with closed angle NVG, IOP was not improved by IVB despite resolution of NVI. These findings remained true even with additional panretinal photocoagulation (PRP). They proposed that IVB could also be used in NVG with closed angle to improve surgical results. While these results are encouraging, more studies need to be done, particularly randomized controlled trials.
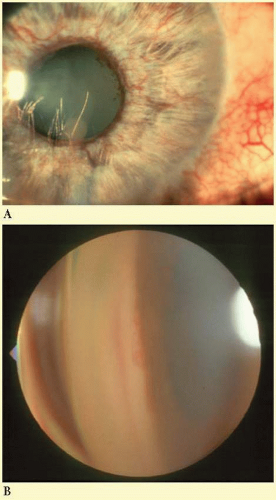 FIG. 54.1 Left: Neovascularization of the iris in neovascular glaucoma. Right: Neovascularization of the angle in neovascular glaucoma. (Photos courtesy of B.J. Shingleton.) |
For eyes with poor visual potential, another use of IVB has been proposed. Although diode laser cyclophotocoagulation (CPC) may effectively lower IOP in eyes with NVG, it is not known to induce regression of NVI. Ghosh et al.4 studied the efficacy of combining IVB with diode laser CPC. This case series of 14 consecutively diagnosed eyes with NVG involved concurrent treatment of CPC and IVB (1.25 mg) and follow-up of at least 6 months. All eyes in this series had closed angle NVG. At 6 months, 9 of 14 eyes achieved a 30% reduction of IOP, 13 of 14 eyes were pain-free, and 12 of 14 eyes had a dramatic regression in anterior segment neovascularization. These results are promising for eyes with NVG that are resistant to PRP or bevacizumab alone.
Medical management of NVG is often insufficient, but includes the use of topical beta blockers, alpha agonists, and topical and oral carbonic anhydrase inhibitors. Cycloplegics and corticosteroids may also be useful in reducing IOP and controlling inflammation. Medical treatment may serve as a temporizing measure prior to surgery. Filtering surgery is more successful after PRP treatment for neovascularization. The adjunctive use of antimetabolites, 5-flurouracil, or mitomycin C, has been shown to increase success rates of trabeculectomy. Glaucoma drainage devices are preferentially employed for IOP control in NVG.
UVEITIC GLAUCOMA
Several mechanisms of uveitic glaucoma exist, including clogging of the trabecular meshwork with inflammatory material, neovascularization, formation of posterior synechiae with subsequent iris bombe, formation of PAS, anterior rotation of the lens-iris diaphragm, and trabeculitis.5 Understanding the pathogenesis of uveitic glaucoma is expanding with more research into aqueous humor dynamics. There are multiple mechanisms involved in this type of glaucoma from simple mechanical obstruction of aqueous outflow by PAS to more complex inflammatory mechanisms.6
In angle closure related to uveitis, the peripheral iris becomes apposed to the trabecular meshwork resulting in the formation of PAS (Fig. 54.2). The disruption of the blood-aqueous barrier and presence of cells and debris in the angle cause the surfaces of the iris and trabecular meshwork to become sticky and more likely to form PAS. These PAS are more prominent in the inferior angle due to the settling of inflammatory debris in this area. In addition, there can be formation of posterior synechiae involving the iris adhering to the anterior lens capsule, the intraocular lens in pseudophakic patients or the vitreous face in aphakic patients (Fig. 54.3). If there are 360 degrees of posterior synechiae, the pupil is then said to be secluded since aqueous humor can no longer flow from the posterior to the anterior chamber. This condition leads to iris bombe—bowing forward of the iris with shallowing of the anterior chamber due to posterior pressure of aqueous humor—and acute angle closure glaucoma (Fig. 54.4).
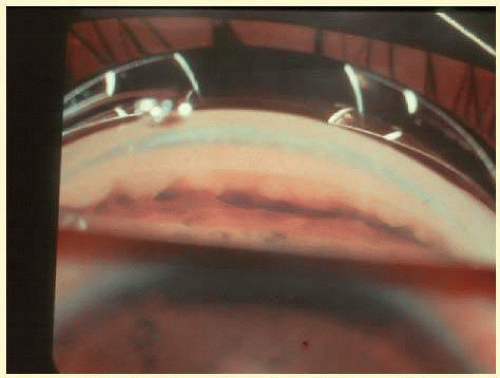 FIG. 54.2 Peripheral anterior synechiae in uveitic angle closure. (Photo courtesy of B.J. Shingleton.) |
As with other secondary glaucomas, management of uveitic glaucoma begins with treatment of the underlying condition then lowering the IOP. In most cases, first line anti-inflammatory treatment is a corticosteroid, including topical, sub-Tenon’s, intravitreal, and systemic administration. Nonsteroidal anti-inflammatory agents may be helpful in cases where corticosteroids are inadequate or contraindicated. Immunosuppressive drugs are sometimes used as steroid-sparing agents to aid in avoiding corticosteroid side effects, especially elevated IOP. Cycloplegia is also used to stabilize the blood-aqueous barrier, control pain, and to prevent a secluded pupil.
If the angle is mostly open, adequate control of inflammation may lead to IOP control. When more treatment is needed for IOP control, agents that may be used include topical beta blockers, alpha agonists, and topical and oral carbonic anhydrase inhibitors. Prostaglandin analogs are avoided due to their proinflammatory properties.
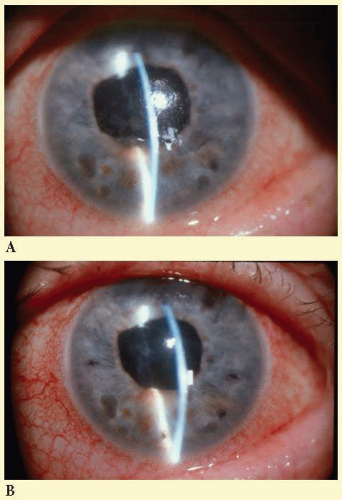 FIG. 54.4 Iris bombe in uveitic angle closure before (left) and after (right) laser iridotomy. (Photos courtesy of B.J. Shingleton.) |
The first line of treatment for uveitic angle closure is to lower the IOP. This is accomplished with medical treatment using aqueous suppressants. Choices include beta blockers, alpha agonists, and carbonic anhydrase inhibitors. Metipranolol is a beta blocker that has been associated with granulomatous uveitis and should be avoided. Likewise, prostaglandin analogs have been associated with increased ocular inflammation so would not be helpful for uveitic angle closure. Oral carbonic anhydrase inhibitors may also be used for IOP control. Cycloplegic and mydriatic agents such as atropine 1% and phenylephrine 10% may be used to break posterior synechiae. This is most effective with recently formed posterior synechiae.
Goniosynechialysis is a surgical method for treating PAS. In this technique, the anterior chamber is deepened with viscoelastic and the synechiae are broken with an instrument, such as an iris sweep, to tease the iris adhesions away from the trabecular meshwork by moving the instrument in an anterior to posterior direction. For iris bombe, since 360 degree posterior synechiae block the movement of aqueous from the posterior to the anterior chamber, a peripheral iridotomy (PI) should be used to create another route for aqueous flow. An inflamed eye will require a large iridotomy and may need more than one due to the tendency for closure. Iridotomies performed with an argon laser have higher failure rates than those created with the Nd:YAG laser.7 In some cases a surgical iridectomy is necessary to obtain an opening large enough to remain patent during future episodes of uveitis.
If medical treatment is inadequate for IOP control, surgical approaches may be considered.8 Trabeculectomy success can be improved with the use of antifibrotic agents such as mitomycin C and 5-FU. Tube shunt procedures may also be performed in these patients. There is no role for laser trabeculoplasty in uveitic eyes since this procedure may exacerbate inflammation and lead to formation of PAS. In cases where HSV is suspected as the cause of uveitis, antiviral agents should also be used.
EPITHELIAL DOWNGROWTH/FIBROUS INGROWTH
Epithelial downgrowth is a rare cause of secondary angle-closure glaucoma. It occurs after intraocular surgery such as cataract extraction, penetrating keratoplasty, removal of anterior chamber epithelial or iris inclusion cysts, and McCannel suture. This condition may also occur after pterygium excision. In addition to angle structures, the membrane may grow over the corneal endothelium, iris, and vitreous face.
The epithelium gains access to the interior of the eye through an open wound or a fistula. These conditions are precipitated by complicated surgery which usually involves hemorrhage, vitreous loss, Descemet’s membrane tear, or tissue incarceration.9 Signs and symptoms include conjunctival injection, photophobia, ocular discomfort, and epiphora. On slit lamp exam, cells and aqueous flare are seen. In cases of wound gape or fistula, a Seidel test will be positive and the eye may be hypotonus. The characteristic finding is an irregular, gray-white scalloped membrane of the posterior corneal surface. The membrane may be associated with a vascularized corneal stroma. There are no keratic precipitates. The cornea may be thickened. If the IOP is high, bullous keratopathy may ensue. The membrane preferentially grows over iris and ciliary body leading to distortion of the normal architecture. Gonioscopic examination reveals PAS.
Clinically, a white membrane representing a sheet of epithelium is seen covering the angle structures. This membrane obstructs the drainage of aqueous and leads to increase of intraocular pressure. If there is doubt as to the etiology of the membrane, an argon laser may be used to identify the tissue as epithelium. Argon laser (500 microns, 100 to 200 mW, 0.1 seconds) aimed at the membrane on the iris will cause a white burn. Other methods of identification include specular microscopy, fluorophotometry, and more invasive methods such as aqueous humor aspiration, posterior corneal curettage, and iris or posterior corneal biopsy.9
The mechanism of glaucoma in epithelial downgrowth is multifaceted. In the presence of wound gape or fistula, hypotony occurs as long as an opening is present. If the fistula closes spontaneously or iatrogenically, glaucoma often results.9 Early on, inflammation associated with epithelial invasion leads to PAS and partial angle closure. Trabeculitis may also be present and associated with decreased aqueous humor outflow in parts of the angle that remain open. Neovascularization and hemorrhage may also occur. Patients with a history of long-term topical steroid use may have a less clear clinical picture in the early stages. Without treatment, angle-closure glaucoma develops from an obstructed trabecular meshwork.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree