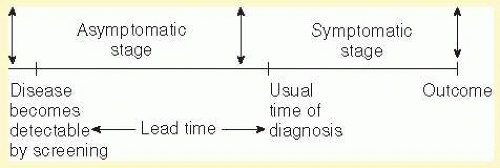
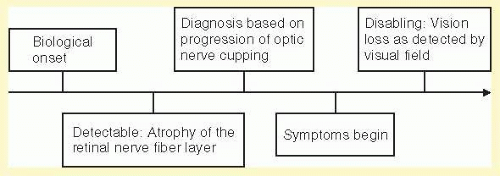
Population |
Number and Race/Ethnicity |
Screening Methodsa |
Referral IOP Level (mm Hg) |
Prevalence of High IOP/Ocular Hypertension (%) |
Prevalence of Glaucoma (%) |
Ferndale, Wales, 196615 |
4,231 White |
H, T, O |
>20 |
9.4% |
0.5% |
P(l/3) |
|
|
|
Dalby, Sweden, 198116 |
1,511 White |
H, T, O, P |
>20.5 |
7.3% |
0.9% |
Framingham, MA, USA, 198017 |
5,262 White (eyes) |
H, T, O |
>21 |
7.6% |
0.8%b |
Beaver Dam, WI, USA, 199218 |
4,926 White |
H, T, FP, P |
>21 |
4.7% |
2.1% |
Baltimore, MD, USA,199119 |
2,913 White 2,395 Black |
H, T, FP, P |
>21 |
7.4% |
1.1% |
4.2% |
St. Lucia, West Indies, 198920 |
1,667 Black |
H, T, O |
>21 |
18.4% |
8.6% |
Barbados, West Indies, 199421 |
4,631 Black |
H, T, O, FP, P |
>21 |
17.8% |
6.7% |
Japan, 199122 |
8,126 Asian |
H, T, FP |
>21 |
2.0% |
2.6% |
Rotterdam Eye Study, Rotterdam, The Netherlands, 200023 |
6,281 White |
O, P, T, FP |
None (high IOP = >21 mm Hg) |
5.6% |
0.8% |
Blue Mountains Eye Study, Sydney, Australia, 199624 |
3,654 White |
H, P, FP |
None |
3.7% |
2.4% |
Ponza, Italy, 199725 |
1,034 White |
H, T, O |
>20 |
6.0% |
2.5% |
Melbourne Visual Impairment Project, Australia26 |
3,271 White |
T, P, O, FP |
>21 |
1.6% |
1.7% |
Proyecto VER, Arizona, USA, 200127 |
4,774 Hispanic |
P, FP, H, T, O |
>22 |
2.3% |
2.0% |
Aravind Comprehensive Eye Survey, Tamil Nadu, India, 200328 |
5,150 Asian |
T, O, P, FP |
None (high IOP = >21 mm Hg) |
1.1% |
1.2% |
Tanzania, East Africa, 200029 |
3,268 Black |
T, O, P |
>23 |
2.7%c |
3.1% |
a H, history of glaucoma; T, tonometry; O, ophthalmoscopy; P, perimetry, FP, fundus photography.
b Excluding blind spot enlargement.
c Includes high IOP without glaucoma. |
|