Chapter 78 Sarcoidosis
Sarcoidosis is a multisystem granulomatous disorder of unknown etiology characterized by intrathoracic involvement. The disease was first described as early as 1869, but its protean presentation and clinical course still make sarcoidosis a diagnostic and therapeutic challenge for modern-day physicians. Ocular involvement is common and has been previously reported to occur in approximately 15–25% of patients with sarcoidosis.1,2 However, some series have reported higher rates. Posterior-segment manifestations may account for up to 28% of the lesions seen in patients with ocular sarcoid. Most large case series of patients with uveitis report that approximately 5% of patients with uveitis have biopsy-confirmed systemic sarcoidosis.3–6
General considerations
Epidemiology
Sarcoidosis is worldwide in its distribution, but it is most frequently recognized in developed countries where adequate diagnostic facilities are available. Although all races are affected, series in the USA generally show that the disease is more prevalent in blacks than in whites. It has also been noted that the prevalence of sarcoidosis in black populations of Africa and South America is less than what has been observed in the African American population.7–9 Both sexes are affected, with the overall frequency showing a very slight excess of females (approximately 60%). Sarcoidosis is a disease of young adults, with almost three-fourths of cases occurring in those younger than 40 years of age. Children may be affected, but this is uncommon.6–10 The clinical course of childhood sarcoidosis is often atypical; that is, there is less frequent pulmonary involvement and more frequent extrathoracic disease.11,12 A recent study of 46 caucasian children from Denmark reported the four most frequent presenting symptoms to be erythema nodosum (22%), iridocyclitis (22%), peripheral lymphadenopathy (15%), and cutaneous sarcoidosis (7%). Overall, a good prognosis was observed. Moreover, erythema nodosum was generally associated with good outcomes while central nervous system (CNS) involvement was associated with a poor prognosis.13 These children must be differentiated from children with juvenile rheumatoid arthritis and those with familial juvenile systemic granulomatosis14–17 because of the similarity of ocular and articular involvement. Many cases of familial juvenile systemic granulomatosis are misdiagnosed as childhood sarcoidosis.18
Etiology and pathogenesis
The etiology of sarcoidosis is unknown. Multiple etiologies have been proposed, including a variety of infectious agents, allergy to pine pollen and peanut dust, chewing pine pitch, and hypersensitivity to chemicals such as beryllium or zirconium. To date, there is no conclusive evidence to implicate any of these as an etiologic agent. Familial studies and human leukocyte antigen (HLA) typing have suggested a possible genetic predisposition, but these studies are far from conclusive.19 A cooperative multicenter study, A Case Control Etiologic Study of Sarcoidosis (ACCESS), enrolled 736 biopsy-confirmed cases from 10 centers in the USA and suggested a genetic predisposition for sarcoidosis, presenting evidence for the allelic variation at the HLA-DRB1 locus as a contributing factor to the disease. The most significant finding was the relationship between DRB1*0401 and eye involvement (P ≤ 0.0008; odds ratio (OR) 3.49) in the study population. Although only 2% of blacks had a DRB1*0401 allele, a similar OR was present in both blacks and whites.19
Bronchoalveolar lavage has enabled investigators to determine the immunologic events at the area of active disease in the lungs. These studies have shown entirely different results from peripheral blood lymphocytes. In the lungs, there is an excess of helper T lymphocytes (CD4+). These activated helper T cells spontaneously secrete lymphokines, including interleukin-2 (IL-2), and will polyclonally activate B cells to produce immunoglobulins. These studies have been interpreted to show that an active T-cell-driven immunologic response occurs at the target organ site and eventually leads to granuloma formation.21,22 Studies of bronchoalveolar lavage fluids have suggested that macrophages may also play a role in the pathogenesis of pulmonary sarcoidosis by inducing changes in the pulmonary microvasculature.23
Immunohistologic studies of biopsy tissue from patients with sarcoidosis have demonstrated the presence of cells of macrophage lineage and activated T cells in the granuloma. The vast majority of lymphocytes are T cells of the helper subset (CD4+) and express activation markers, including class II antigens and the IL-2 receptor.24–26
Clinical features
The organs most frequently involved in sarcoidosis are the lungs, lymph nodes and spleen, skin, eyes, nervous system, and bones and joints1,19,27–29 (Table 78.1).
Table 78.1 Organ system involvement in sarcoidosis
| Organ system | Frequency (%) |
|---|---|
| Intrathoracic | 84–93 |
| Hilar nodes | 60–77 |
| Lung parenchyma | 40–56 |
| Lymph nodes | 23–37 |
| Eyes | 11–32 |
| Skin | 12–27 |
| Erythema nodosum | 4–31 |
| Spleen | 1–18 |
| Bones | 2–9 |
| Parotid | 5–8 |
| Central nervous system | 2–7 |
Intrathoracic sarcoidosis
Several series have demonstrated that intrathoracic involvement is the most common manifestation of sarcoidosis and occurs in 90% of patients. An abnormality on chest radiograph examination is evident at the onset of sarcoidosis in almost all patients. Chest radiograph abnormalities have been classified according to a simple staging system, which closely correlates with the eventual outcome. Stage 0 is characterized by a normal chest radiograph; stage 1 is characterized by bilateral hilar lymphadenopathy without pulmonary infiltration and is seen in 65% of patients with pulmonary sarcoidosis; stage 2 is characterized by hilar lymphadenopathy associated with pulmonary infiltration and is seen in 22% of patients with sarcoid; stage 3 sarcoid is characterized by pulmonary infiltration with fibrosis but without bilateral hilar adenopathy and occurs in 13% of patients. The overall rates of radiographic resolution are 59%, 39%, and 38% for stage 1, 2, and 3 disease, respectively.1
Extrapulmonary lesions
Involvement of the reticuloendothelial system, particularly the extrapulmonary lymph nodes or spleen, or both, is common and occurs in 23–37% of patients with sarcoidosis. Biopsy of a palpable lymph node is often used for histologic confirmation of the diagnosis of sarcoidosis (see below). Skin lesions occur frequently in sarcoidosis and include erythema nodosum, lupus pernio, maculopapular rashes, cutaneous plaques, and subcutaneous nodules. Lupus pernio (dusky-purple infiltration of the skin of the nose) and sarcoid plaques are typically associated with chronic disease, whereas erythema nodosum (especially in the presence of polyarthritis) is typically associated with acute sarcoid or Lofgren syndrome.30 Neurosarcoidosis occurs in 2–7% of patients with sarcoid. Facial palsy is the most frequent manifestation of neurosarcoidosis. Other presentations include other cranial nerve palsies, papilledema, peripheral neuropathy, meningitis, space-occupying cerebral lesions, cavernous sinus syndrome,31 and endocrine disorders such as hypopituitarism or diabetes insipidus resulting from space-occupying lesions. Musculoskeletal involvement includes bone cysts in patients with chronic sarcoid, polyarthralgias, and periarthritis in patients with acute sarcoid, and, less commonly, myopathy from granulomatous lesions within muscles.1,19,29,31–35
Investigations
Radiological evaluation
High-resolution computed tomography (CT)
The CT findings in sarcoidosis can be divided into three main categories:
The presence of diffuse nodular opacities (1–5 mm) with irregular borders in the perilymphatic distribution typically in the upper and middle lung zones is the most common and almost universal finding in the lung parenchyma.36 The presence of architectural distortion is also exclusive to sarcoidosis as opposed to other diseases with perilymphatic distribution.37 “Sarcoid galaxy sign,” which is a collection of multiple granulomas and gives the impression of an opacity, is present in 10–20% of patients with pulmonary sarcoidosis.38 Extensive fibrosis, mostly distributed in the upper and middle zones associated with architectural distortion, is also seen in 20–25% of cases. The presence of air trapping at end expiration has been reported frequently. Terasaki et al.39 reported air trapping on expiration in 45 (98%) of their study patients. Devies et al.40 demonstrated that the extent of air trapping on expiration was correlated with the pulmonary function of the patient.
Large-airway involvement is seen in 1–3% of patients with pulmonary sarcoidosis. This typically presents with tracheal stenosis due to granuloma formation within the tracheal mucosa or submucosa or secondary to lymph node compression from the outside. More than 60% of patients with sarcoidosis are reported to have smooth thickening of the smaller airways.36
The classic radiologic finding in the mediastinum of a sarcoid patient is symmetric bilateral hilar adenopathy with some form of paratracheal adenopathy. Symmetry is an important diagnostic feature of the sarcoid hilar adenopathy as it is uncommon in the major diagnostic alternatives, including tuberculosis and lymphoma.36
Gallium scan
Gallium-67 is a radioactive isotope with a half-life of 78 hours and is administered intravenously to patients as Ga-citrate. In the vascular system, the half-life of Ga drops to 12 hours. Ga is taken up by activated macrophages in the epithelioid cell granulomas. Such uptake is detected by gamma cameras 48 hours after injection and is assessed in the liver, spleen, thorax, eyes, and lacrimal and salivary glands. Scanning has also been suggested as a useful diagnostic test for sarcoidosis. The test is again nonspecific, and gallium uptake is seen in other diseases, including Sjögren syndrome, tuberculosis, radiation therapy, and lymphoma. It has been proposed that the combination of gallium scanning and an elevated angiotensin-converting enzyme (ACE) level is highly specific for sarcoidosis, but these studies have employed patients specifically chosen for very active sarcoid.41 As such, these tests may be of less utility in a patient with presumed sarcoid uveitis and no obvious evidence of systemic sarcoidosis. The best use of these tests may be in following patients with active disease.42,43
In a study of 22 patients with sarcoid uveitis compared to 70 patients with uveitis secondary to other disorders, Power et al.44 reported that the sensitivity and specificity of an elevated ACE alone in diagnosing sarcoidosis were 73% and 83%, respectively, and that the sensitivity and specificity of the gallium scan alone were 91% and 84%, respectively. Using the combination of a gallium scan and an elevated serum ACE, the specificity for diagnosing sarcoidosis was 100% and the sensitivity was 73%. The authors concluded that the combination of serum ACE level and whole-body gallium scan might be useful for diagnosing sarcoidosis in patients with uveitis. However, because of the study design inherent in investigating the values of these tests, their actual utility in patients with normal chest radiographs and without clinical evidence of sarcoid remains uncertain. Furthermore, because the reported prevalence of sarcoid uveitis is approximately 5% among patients with biopsy-confirmed systemic sarcoidosis,3–6 routine screening of all patients with uveitis by both ACE levels and gallium scan may have a low positive predictive value and, therefore, may be misleading. Nevertheless, in selected patients in whom sarcoidosis is highly likely, these tests may be useful.
Magnetic resonance imaging (MRI) and positive emission tomography scan (PET)
Fluorodeoxyglucose PET (18F-FDG PET) scanning has become of great importance in the study and follow-up of patients with cancer. However, in recent years, its role in the diagnosis and management of other conditions such as sarcoidosis has been explored. Although the sensitivity of a whole-body PET scan to detect sarcoid lesions is 80–100%, the findings are nonspecific and histology is required to confirm or rule out sarcoidosis. MRI has been successfully used in the evaluation of organ-specific damage in patients with sarcoidosis, especially cardiac and musculoskeletal tissue.41,44–48
Histology
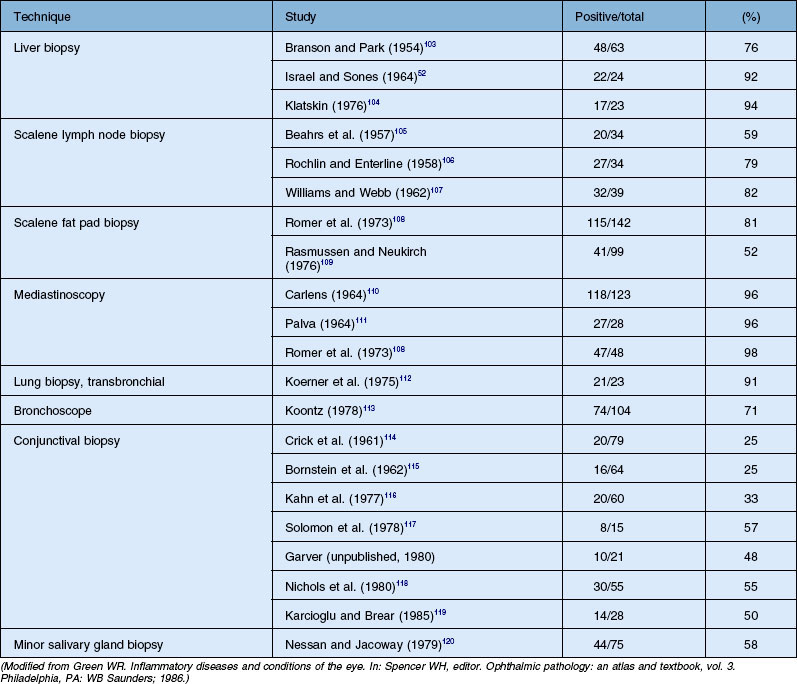
Histologic confirmation (Table 78.2) is generally required to establish the diagnosis of sarcoid. The only clinical situation when diagnosis of sarcoidosis can be strongly considered reliably without biopsy is Löfgren syndrome (see below). Otherwise, biopsy should be done. Sites most often biopsied include the lungs, mediastinal lymph nodes, skin, peripheral lymph nodes, liver, and conjunctiva. Biopsy of clinically evident skin lesions or palpable lymph nodes is frequently performed because of the high yield and low morbidity. Fiberoptic bronchoscopy with transbronchial lung biopsy is positive in 80% of patients with intrapulmonary sarcoidosis. This procedure is routinely performed by pulmonary physicians and has a relatively low morbidity. The next step in such a scenario is cervical mediastinoscopy, which is both highly invasive and an expensive procedure. The requirement of general anesthesia further increases the chances of complications from the procedure. From the patient’s perspective it is important that further possibilities of minimally invasive techniques for the diagnosis of sarcoidosis are explored. Development of linear echoendoscopes and subsequent procedures (endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) and endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) ) has opened new diagnostic possibilities for sarcoidosis. Both techniques allow real-time monitoring of the needle. Several investigators have reported a high sensitivity of 72–85% for EBUS-TBNA and minimal complications. In a randomized control trial (RCT) Tremblay et al.49 have shown that the diagnostic yield of EBUS-guided TBNA (95.8%) versus the conventional TBNA (73.1%) was 22.7% greater. Sensitivity and specificity were 60.9% and 100%, respectively, in the standard TBNA group, and 83.3% and 100%, respectively, in the EBUS-guided TBNA group (absolute increase in sensitivity of 22.5%). EUS-FNA was used for diagnosis of sarcoidosis and had a yield of 82% and sensitivity of 89–94% by assessing noncaseating granulomas in mediastinal nodes.50 The liver biopsy is often positive in patients with sarcoidosis, but the finding of granulomatous lesions on liver biopsy must be interpreted with caution, as they can be produced by other disorders. Other potential biopsy sites include peripheral lymph nodes and minor salivary glands.
Conjunctival biopsies are positive in 25–57% of patients with histologically documented sarcoidosis. Variations in these reports are the result of whether clinically evident lesions are biopsied or whether a “blind” conjunctival biopsy is performed. However, the yield can be increased by techniques such as bilateral conjunctival biopsies and serial sectioning of the specimens, and careful inspection of the conjunctiva for any visibly evident nodules which can be biopsied. Transconjunctival lacrimal gland biopsy can also be used for histologic diagnosis, but the procedure is not performed routinely.42,51
Immunology
The Kveim skin test was a simple, specific, outpatient skin test using human sarcoid tissue. It was positive in 78% of patients with sarcoidosis and was helpful in delineating multisystemic sarcoidosis from other granulomatous disorders. The antigen was a saline suspension of human sarcoid tissue prepared from the spleen of a patient suffering from active sarcoidosis. This material was injected intradermally, and the site inspected for nodule formation after 3–6 weeks. A palpable nodule was biopsied, and the finding of noncaseating granulomas on biopsy established the diagnosis of sarcoid.19,52,53 Concerns about the injection of human tissue, with its potential for disease transmission, have essentially eliminated the use of the Kveim test.
Noninvasive tests
Multiple attempts have been made to find noninvasive tests that could be both sensitive and specific in the diagnosis of sarcoidosis. These have included measurement of serum calcium, urinary calcium, serum lysozyme, and serum immunoglobulins. Although all these may be abnormal in patients with sarcoid, they are nonspecific and nondiagnostic. The serum ACE level has been touted as a useful measurement in the diagnosis of sarcoidosis. The ACE level is frequently abnormal in patients with active sarcoidosis and appears to reflect the total-body granuloma content in such patients. As such, it may be useful in following patients with active sarcoid.43,52,54 However, it is not diagnostic of sarcoidosis and appears to be of limited utility in the diagnostic dilemma of patients with possible sarcoid uveitis but a normal chest film.
For following patients with active intrapulmonary sarcoid, pulmonary function tests, particularly forced vital capacity, forced expiratory volume, and diffusing capacity, are far more useful. Changes in pulmonary function tests are often used to follow patients with sarcoidosis and to adjust the corticosteroid dosage.27
Jabs and Johns55 reported that over 80% of patients with ocular sarcoid had their ocular lesions at the time of diagnosis of sarcoidosis, and Hunter and Foster48 reported that 3% of patients with uveitis were diagnosed as having sarcoid after the initial evaluation for a systemic disease revealed no diagnosable systemic disorder. Although patients who present with uveitis should be evaluated for sarcoid, repetitive workups appear to have limited value unless new symptoms arise.
Course and prognosis
There appear to be two distinct paradigms of sarcoidosis, acute and chronic, with differences in onset, natural history, course, prognosis, and response to treatment. Acute sarcoidosis tends to have an abrupt, explosive onset in young patients and to go into spontaneous remission within 2 years of onset. Acute iritis is often seen in acute sarcoidosis. The response to systemic corticosteroids is generally quite good, and the long-term complications are minimal. Löfgren’s syndrome comprises erythema nodosum, bilateral hilar adenopathy, and acute iritis; it generally has a good long-term prognosis.1,19,29
Chronic sarcoidosis is defined as disease persistence of greater than 2 years. The disease may have a more insidious onset and generally has intrapulmonary involvement with chronic pulmonary disease as a major source of morbidity. Corticosteroid therapy is generally required and may be prolonged. Chronic ocular disease, particularly chronic uveitis, may be a feature of chronic sarcoidosis.1,19,27,29,55
The overall mortality from sarcoidosis is 3–5%, but neurosarcoid is associated with a mortality of 10%.47 Corticosteroids are the mainstay of treatment, although antimalarial agents can be used for patients with mucocutaneous lesions. Patients with hilar adenopathy without abnormalities of pulmonary function and without intrapulmonary infiltration may not need systemic corticosteroid treatment.
Ocular manifestations
Multiple studies have documented the common occurrence of ocular involvement in sarcoidosis and the various ocular manifestations of sarcoid. Frequency estimates vary and have ranged as high as 50%.47 However, most series report rates that are generally closer to 15–28%.1,19,29,32,55,56 These differences undoubtedly relate to case ascertainment methods, the patient population studied, definitions of ophthalmic involvement, and the nature of the evaluation conducted. Racial differences influence not only the mode of presentation for ocular sarcoidosis but also the frequency of patients with ocular involvement. When the same diagnostic criteria were applied, Japanese patients with sarcoidosis were found to have ocular disease six times more frequently than Finnish patients, and they were also more likely to present with ocular symptoms of sarcoidosis. In the USA, the African American population is twice as likely to have ocular disease as opposed to caucasian patients. Furthermore, high frequencies of ocular involvement are reported when keratoconjunctivitis sicca is sought carefully and included as evidence of lacrimal involvement in sarcoidosis.47
Sarcoidosis may affect most of the ocular structures, as well as the orbit and adnexa. Ocular lesions described in sarcoidosis include: anterior uveitis, iris nodules, conjunctival nodules, scleral nodule,57 and corneal disease with either band keratopathy or interstitial keratitis; posterior segment disease, including chorioretinitis, periphlebitis, chorioretinal nodules, vitreous inflammation, and retinal neovascularization; and orbital disease, including involvement of the lacrimal gland, nasolacrimal duct, optic nerve, and orbital granulomas. The various ocular lesions along with the prevalence estimates are outlined in Table 78.3.
Table 78.3 Ocular manifestations of sarcoidosis
| Ocular manifestations | Frequency in patients with ocular sarcoid (%) |
|---|---|
| Anterior segment disease | |
| Anterior uveitis | 66–70 |
| Acute | 15–32 |
| Chronic | 39–53 |
| Iris nodules | 11–16 |
| Conjunctival lesions | 7–47 |
| Cornea | |
| Band keratopathy | 5–14 |
| Interstitial keratitis | 1 |
| Posterior segment disease | |
| Vitritis | 3–25 |
| Periphlebitis | 10–17 |
| Chorioretinitis | 11 |
| Choroidal nodules | 4–5 |
| Retinal neovascularization | 1–5 |
| Orbital and other disease | 26 |
| Lacrimal gland | 7–60 |
| Keratoconjunctivitis sicca | 5–60 |
| Enlargement | 7–28 |
| Orbital granuloma | 1 |
| Optic nerve granuloma | <1–7 |
The frequency of orbital, and particularly lacrimal gland, lesions varies widely among series and depends on the patient selection and investigations used. Clinical enlargement is present in less than one-third of patients with ocular sarcoid, but when sought, keratoconjunctivitis sicca may be present in a greater percentage.29,32,47,55–57 Orbital granuloma, independent of the lacrimal gland, occurs infrequently.58–60 Massive lacrimal gland enlargement simulating a lacrimal gland tumor may occur and require biopsy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree