Chapter 108 Retinotomies and Retinectomies
Drainage retinotomy
A drainage retinotomy is a retinal hole created to allow removal of subretinal fluid.1,2 A drainage retinotomy is most often used in conjunction with treatment of a retinal detachment with vitrectomy techniques. Because of the potential for complications and the availability of other techniques, posterior drainage retinotomy is less frequently used today; if a drainage retinotomy is needed, a peripheral drainage retinotomy is more commonly used. A drainage retinotomy may be necessary in proliferative vitreoretinopathy (PVR), other complicated retinal detachments, proliferative vascular retinopathy, and primary rhegmatogenous retinal detachment treated with vitrectomy. The drainage retinotomy can be used when a pre-existing retinal tear is not present or not adequate for drainage or egress of the subretinal fluid.
General principles
The drainage retinotomy is made after as complete a removal as possible of periretinal membranes, and should not be created in an area with residual membranes or traction. The drainage retinotomy is most easily created with endodiathermy.3 Taking care not to close large vessels, the unimanual, bipolar endodiathermy probe is set on a continuous mode and held against the retina in the selected area. A necrotic retinal hole with a well-marked white edge is usually produced. The advantages of creating the retinotomy with diathermy are: (1) complete hemostasis of the retinal blood vessels; and (2) whitening of the edge of the hole, which allows easy identification of the retinotomy when flat against the retinal pigment epithelium for laser treatment. If the diathermy probe does not perforate the necrotic area, the central necrotic area is perforated with a sharp blade, or by suction with the blunt tip of the vacuum needle.
Surgical technique in conjunction with perfluorocarbon liquid (PFCL)
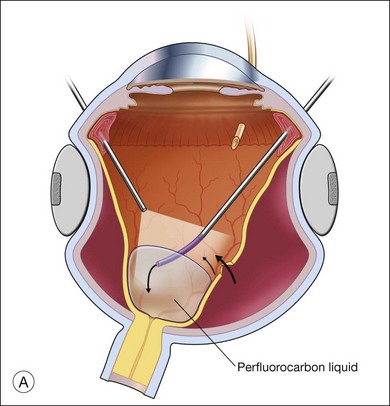
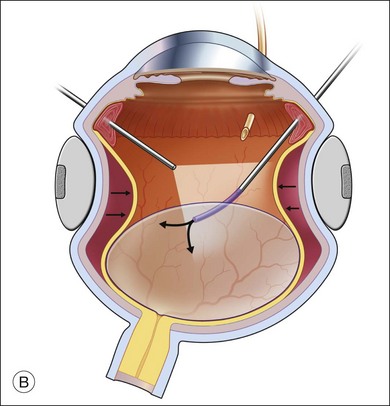
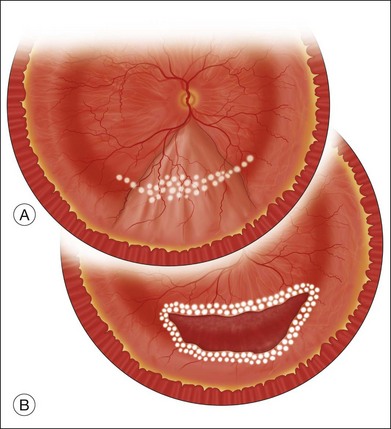
The preferred technique for retinal reattachment is to use PFCL. PFCL, heavier than water or saline, can be used to reattach the retina from posteriorly to anteriorly.4 In addition, PFCL can fixate and stabilize the posterior retina, thereby facilitating removal of peripheral vitreous and membranes.5 A peripheral retinal break is necessary to allow egress of subretinal fluid when PFCL is injected over the posterior pole. A pre-existing retinal break is usually present, but if the break is not located anteriorly enough, subretinal fluid may become loculated beneath peripheral retina anterior to the anterior-most retinal break (Fig. 108.1A,B). Anterior subretinal fluid can usually be drained through posteriorly located retinal breaks during PFCL–air exchange. However, most surgeons prefer to do laser treatment through PFCL rather than air, so it is desirable to flatten the retina with PFCL. If anteriorly loculated subretinal fluid is present, one of several approaches can be taken to remove it. With PFCL filling the eye to just posterior to the most anteriorly located retinal break, a partial fluid–air exchange can be done to remove the subretinal fluid through the break. Then, if the desire is to do laser treatment throughout the PFCL, additional PFCL is injected to bring the level anteriorly; anterior air can then be replaced with infusion fluid in order to have a clear view. There is a risk of corneal striate keratopathy or pupillary constriction associated with the air in the aphakic eye that can reduce visualization and interfere with laser treatment and subsequent fluid–air exchange. In the phakic eye, there is the possibility of posterior subcapsular lens opacity induced by the air that could interfere with visualization.
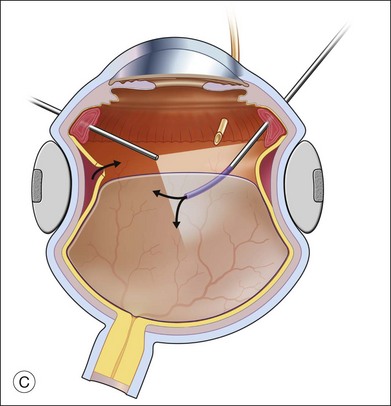
An alternative approach that is less time-consuming and more likely to maintain good visualization is to create an anterior drainage retinotomy with endodiathermy in the area where subretinal fluid is loculated in order to allow egress of the fluid into the vitreous cavity (Fig. 108.1C). If a scleral buckle is not present and will not be used, the retinotomy can be made just posterior to the ora serrata. If a scleral buckle is present, the retinotomy should be made in an area of retina supported by the buckle rather than anterior to the scleral buckle. After the drainage retinotomy is created, PFCL is injected to reattach the retina. If the retinotomy is made in an area under traction, there is a risk that the PFCL could go through the retinal break and course beneath the retina; however, if no traction is present, the level of the PFCL can be safely brought anterior to the break to allow laser treatment. The PFCL is usually removed by performing a PFCL–air exchange. Alternatively, a direct PFCL–silicone oil exchange can be done. It is preferable to drain any residual subretinal fluid, if present, through the anterior-most retinal break or retinotomy during the early portion of the exchange. This will prevent trapping of subretinal fluid that can be forced posteriorly as the eye is filled with air or silicone oil.
Surgical technique without PFCL
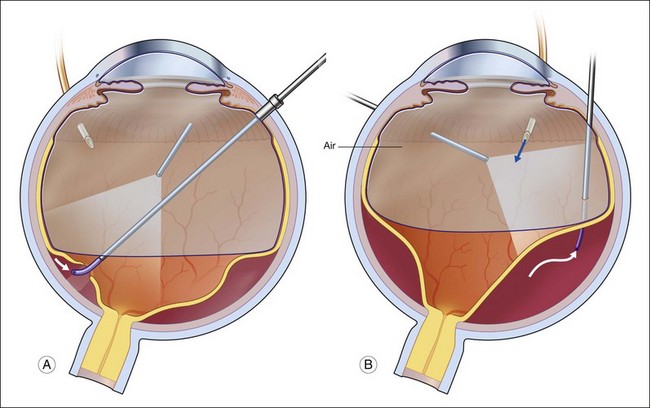
Fluid–air exchange was the most commonly used technique to reattach the retina during vitrectomy before introduction of PFCL. In the absence of a posterior retinal break for internal drainage of subretinal fluid, a posterior drainage retinotomy was usually done. The technique was to make a posterior drainage retinotomy with diathermy, taking care to make it in a superior quadrant at least 1.5 disc diameters from the optic nerve and avoiding the macula and large blood vessels in order to avoid complications. The retina was reattached by suctioning subretinal fluid through the retinotomy while the eye was filled with air supplied by a continuous infusion air pump (Fig. 108.2A). However, posterior drainage retinotomy can be associated with significant complications, including hemorrhage, fibrosis with traction, choroidal neovascularization (CNV), and visual field loss.6 In most instances, a peripheral drainage retinotomy can be used instead of a posterior retinotomy. Although the same complications can occur in the periphery, peripheral complications are usually less visually significant than posterior complications.
Fluid–air exchange can be done through a peripheral break or drainage retinotomy. A needle with an attached fixed-length silicone tube can be used for drainage in the periphery. One available needle has a 6 mm flexible silicone tube that is longer and more flexible than the silicone tube on standard silicone-tip needles (Beaver Visitec International Inc., Waltham, MA). The silicone tip can be inserted through a retinotomy for drainage, and the extra length allows drainage significantly posterior to the level of the retinotomy (Fig. 108.2B).
For fluid–air exchange through a drainage retinotomy, intraocular air pressure is usually set at approximately 30 mmHg. Because of the peripheral location of the drainage retinotomy, a wide angle contact or noncontact viewing system or prism contact lens will improve visualization.7 Before air is insufflated, fluid–fluid exchange (internal drainage of fluid through a retinal break or retinotomy in a fluid-filled eye) will sometimes partially flatten the retina. Air is then insufflated and the air bubble is enlarged to fill the anterior vitreous cavity.
In an eye that is partially filled with air, the view is minimized, and it is necessary to refocus the operating microscope on the drainage retinotomy. The silicone tube is inserted through the drainage retinotomy, and low suction is applied to aspirate subretinal fluid.8 If the tip is not obstructed, subretinal fluid will be removed as the eye fills with air. The retina may not flatten initially because fluid from the vitreous cavity often goes through retinal breaks or through the retinotomy into the subretinal space. When the air reaches the level of the drainage retinotomy, the orifice is obstructed by the air, and no further vitreous fluid can go through the retinotomy into the subretinal space as suction is applied. The retina will flatten unless the tip of the silicone tube is obstructed. Slightly withdrawing the tube will usually initiate further drainage of subretinal fluid if the cannula obstructs. It is rare to aspirate and incarcerate retina into the silicone tip during this maneuver. If difficulty is encountered in the initial part of the exchange and fluid cannot be aspirated while the silicone tube is in the subretinal space, the tube is withdrawn from the subretinal space and aspiration is applied in the vitreous cavity just anterior to the retinotomy. When the air level meets the drainage retinotomy, the silicone tip is gently slipped obliquely into the subretinal space, and aspiration is once more applied. Care must be taken not to push the needle tip forcibly against the retinal pigment epithelium (RPE) because choroidal hemorrhage may occur. When the retina flattens posteriorly over the silicone tube, it is slowly withdrawn from the subretinal space while gently aspirating subretinal fluid. At this point all or most of the subretinal fluid has been removed.
Complications
Postoperative complications of the drainage retinotomy are few. McDonald et al.6 first described cellular proliferation and CNV from drainage retinotomies. Richards and Maberley9 found that subretinal neovascularization was associated with the retinotomy site in 1% of 287 cases and focal PVR was associated with the retinotomy site in 2%. Retinal detachment from the retinotomy site is extremely rare. The retinotomy site may even remain attached if the rest of the retina detaches postoperatively. Cellular proliferation from the retinotomy site may lead to periretinal proliferation. In gas-filled eyes, the proliferation usually remains in the local area of the retinotomy site and rarely leads to complications. In silicone-oil-filled eyes, the proliferation may be more extensive and lead to tractional complications. CNV probably results from damage to the underlying choroid and RPE.
An extensive visual field defect may result from a large retinotomy too close to the optic disc or macula. Bourke et al.10 found visual field defects within 30° of fixation in 12 of 14 eyes with drainage retinotomies. Visual field defects were found in all eyes in which the retinotomy was created within 5 disc diameters of fixation. The researchers recommended that retinotomies be placed more than 5 disc diameters from the fovea and in the superotemporal quadrant to minimize visual field loss.
Retinotomy to gain access to the subretinal space
It is sometimes necessary to create a retinotomy to gain access to the subretinal space. A retinotomy may be necessary to remove a subretinal band or membrane (see Chapter 107, Proliferative vitreoretinopathy); a subretinal hemorrhage; a subretinal neovascular membrane (see both in Chapter 119, Surgical management of choroidal neovascularization and subretinal hemorrhage); a subretinal foreign body, or, rarely, a subretinal mass such as a disciform scar, abscess, or retinal or subretinal tumor.
Subretinal foreign body
If the retina is detached, the foreign body may be mobile. In this case, after vitrectomy, a retinotomy is made with endodiathermy. The retinotomy is made in an area where forceps or the rare-earth magnet will have access to the foreign body. The posterior pole or areas of fibrous proliferation should be avoided when the retinotomy is made. It is often possible to manipulate a subretinal foreign body to the area of the retinotomy before the foreign body is grasped. Joondeph and Flynn11 described moving the foreign body to the retinotomy site with the soft flexible tip of the cannulated extrusion needle. The foreign body is then grasped and pulled into the vitreous cavity through the retinotomy.
Removal of subretinal PFCL
An occasional need for retinotomy is the removal of subretinal PFCL. This indication may arise intraoperatively or postoperatively, subsequent to vitrectomy with use of PFCL. Insufficient release of retinal traction or misdirected injection of preretinal PFCL into a retinal break can result in intraoperative subretinal PFCL. Postoperative retention of subretinal PFCL occurs in up to 12% of cases and is most often associated with large peripheral retinectomies.12
Small extramacular bubbles of subretinal PFCL are usually visually insignificant, and should not be removed unless they show migration toward the subfoveal region. This complication is more likely to occur with bubbles trapped subretinally superior to the macula, and in the presence of epiretinal membranes. If the PFCL bubble is located subfoveally, or is found migrating in that direction, repeat vitrectomy with small retinotomy is indicated, given PFCL’s known toxicity to the photoreceptors and pigment epithelium.12 Subretinal PFCL has been found to produce a dense scotoma on microperimetry.13 Once PFCL is removed, the affected retina tends to regain partial function; the degree of recovery is probably dependent on the duration of its subretinal presence. Large amounts of subretinal PFCL, either intra- or postoperatively, should be removed regardless of location. If the retina is attached, a PFCL bubble can be removed through a small retinotomy given its high fluidity.14–17 Active suction with a 33G subretinal cannula, or smaller, can be used. Light laser photocoagulation of the retinotomy may not be needed, although it may be beneficial if the retina has been recently detached.
Retinal or subretinal mass
Pars plana vitrectomy and retinectomy were used to remove a retinal vasoproliferative tumor, thereby providing a specimen for histopathologic diagnosis and treatment for recurrent hemorrhages and exudation.18 Gaudric et al. reported retinectomy to treat nine eyes with retinal capillary hemangiomas in von Hippel–Landau disease.19
Removal of a subretinal mass is an uncommon indication for retinotomy, although a retinotomy may be used for excision of a subretinal mass such as a disciform scar.20,21 The surgical results of disciform scar removal have been disappointing; more recently, surgery has been directed at removal of choroidal neovascular membranes (see Chapter 119, Surgical management of choroidal neovascularization and subretinal hemorrhage), an earlier form of the disease, or macular translocation away from the area of neovascularization or scar (see Chapter 120, Macular translocation). Vitrectomy removal or biopsy of choroidal melanomas has been popularized in some centers,22,23 Karkhaneh et al.24 reported the outcome on 20 cases followed for 24–132 months (mean, 89.55 months; SD±38.4 months). Only one patient had died of metastasis (liver) and 75% of the eyes were stable, with no evidence of tumor or severe complications. Harris et al. described successfully using extensive retinectomy to remove a subretinal abscess due to Klebsiella.25
Retinotomies to mobilize retina: macular translocation
Macular translocation developed as a complex surgical approach to save central vision in patients affected by exudative and atrophic macular degeneration. It uses extensive peripheral retinectomy to rotate the macula away from the degenerating subretinal structures. Although its popularity has decreased as anti-VEGF therapy has proven to be highly effective, it is still advocated by some retinal surgeons. (See Chapter 120, Macular translocation, for indications and technique.)
Retinotomies to obtain abnormal retinal tissue: retinal biopsy
Retinal and choroidal biopsies use retinectomies and choroidectomies to obtain tissue for pathologic diagnosis in difficult diagnostic problems, such as retinitis, uveitis, and neoplastic diseases. (This topic is covered in Chapter 124, Vitreous, retinal, and choroidal biopsy.)
Retinectomy for treatment of intractable glaucoma
Retinectomy has been advocated for treatment of intractable glaucoma. Kirchhof26 and Joussen et al.27 described vitrectomy and anterior retinectomy to utilize the pressure-lowering effect of retinectomy to treat glaucomas resistant to other forms of therapy. Joussen et al. reviewed 44 eyes treated with retinectomy. Patients included: 12 with neovascular glaucoma; three with infantile and juvenile glaucoma; 13 with aphakic glaucoma; seven with glaucoma secondary to trauma; seven with glaucoma due to uveitis, and two with Ehlers–Danlos syndrome. With at least 5 years’ follow-up, 52.3% had long-term control of intraocular pressure without complications. Among the remaining eyes, complications were frequent, with a high rate of PVR, hypotony, and phthisis bulbi. Eyes with neovascular glaucoma due to central retinal vein occlusion and eyes with uveitis had a particularly poor prognosis.
Relaxing retinotomy and retinectomy
Relaxing retinotomies and retinectomies are used in the presence of retinal shortening as a result of retinal incarceration or fibrous proliferation and contraction that prevents contact of the retina with the RPE.28–33 Usually, the peripheral retina is cut or removed to preserve function of the posterior, more visually significant retina. If the retina is cut and not removed, the procedure is technically a retinotomy; however, in discussing relaxing retinotomies and retinectomies, we will refer here to all retinotomies and retinectomies as “retinectomies”.
The indications for relaxing retinectomies are listed in Box 108.1. Except for retinal incarceration in traumatic or surgical wounds and excision of the inner wall of congenital retinoschisis, all the indications involve PVR or proliferative vascular retinopathy, with fibrous proliferation causing contraction and shortening of the retina. Relaxing retinectomies should only be done if other methods have failed or have no chance of success. A scleral buckle will sometimes adequately relieve traction to avoid cutting the retina. Michels et al.34 have shown that a scleral buckle will change the vector force of contraction of proliferative membranes so the force is no longer applied to pull the retina away from the pigment epithelium. A buckle should be considered in the presence of residual contraction.
Box 108.1
Indications for relaxing retinotomies and retinectomies
General surgical principles and techniques
The relaxing retinectomy can be performed after either 20G or smaller-gauge vitrectomy. Newer instrumentation and the ability to inject silicone oil into eyes using 23G technology makes the use of smaller-gauge technology possible.35 The retinectomy should only be performed after complete membrane removal. If the retina is cut or excised before complete membrane removal, further membrane removal will be more difficult and may result in unnecessarily large retinal defects or residual membranes that may lead to redetachment of the retina. Larger peripheral retinectomies are less functionally significant than are smaller posterior retinectomies. Although a large peripheral retinectomy may be more difficult to manage, the greater preservation of retinal function obtained is usually worthwhile. Circumferential relaxing retinectomies are usually preferred to radial retinectomies. In the face of circumferential traction, a radial retinectomy that adequately relieves traction may extend too far posteriorly into the central retina.
It is useful to be able to see the full extent of the retina to be cut or excised during creation of a retinectomy. With full visualization, it is easier to assess the best location and the necessary extent of the retinectomy. A wide angle viewing system is ideal for visualization of the retina during this maneuver. Either a contact or noncontact system can be used. Use of a wide angle system may reduce the time necessary to do the procedure, improve the ability to apply laser photocoagulation, and reduce the need for scleral depression.36 Before a relaxing retinectomy is created, diathermy should be applied to the entire area to be cut (Fig. 108.3A). Blood vessels in the area should be occluded. The retina can be cut with scissors or a vitrectomy instrument. Scissors will make a more precise, controlled cut, whereas larger areas of retina are more easily excised with the vitrectomy instrument. For folded retina, sequential cutting and reapplication of diathermy, as described later for release of retinal incarceration, is the preferred method. PFCL may be necessary to stabilize the retina during retinectomy.5 As a general principle, the retinectomy should extend into normal retina on each end of the area of contraction. With shorter retinectomies, the extension into normal retina needs to be only a few degrees in length. With very large retinectomies, extension into the normal retina may need to be up to 30°. If the normal retina is attached, care must be taken not to damage the choroid during retinectomy, because bleeding may occur. After diathermy, the retina should be gently pulled away from the pigment epithelium by the scissors tips or a pick before cutting.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree