Retinopathy of Blood Dyscrasias
Gavin W. Roddy
Robert H. Rosa Jr.
The word dyscrasia comes from the Greek language and means “bad temperament.” In the older medical literature, the term dyscrasia was used to indicate disease. Currently, we use the phrase blood dyscrasia to indicate a pathologic condition of the blood, usually when referring to disorders of the cellular elements of the blood. An increase or decrease in the total number of red blood cells in a given patient is referred to as polycythemia or anemia, respectively. A large population of atypical or neoplastic white blood cells within the blood constitutes leukemia. A subnormal number of platelets in the circulating blood (thrombocytopenia) or loss of normal platelet function can lead to bleeding disorders or coagulopathies. Interestingly, disorders of the various cellular components of the blood often overlap (i.e., leukemia, anemia, and thrombocytopenia).
Clinical and pathologic studies indicate that the ocular manifestations of blood dyscrasias are frequent. A cross-sectional study of 226 anemic and/or thrombocytopenic patients revealed fundus lesions as a frequent finding. Retinopathy was observed in 28% of patients with either an anemia or a thrombocytopenia and in 38% of patients with both blood disorders.1
Herein, the typical ophthalmoscopic findings in blood dyscrasias are reviewed, and general information regarding the most common blood dyscrasias is presented.
OPHTHALMOSCOPIC FINDINGS
Apart from the hemorrhagic and infiltrative complications observed in the skin and mucous membranes, the ocular fundus provides an unequaled direct view of the hematologic effects of blood dyscrasias in the living patient. The ophthalmologist may be the first member of the medical team to identify the hematologic effects of a blood dyscrasia by observing the characteristic changes in the retina. Indeed, recent cases of patients with life-threatening hematologic disorders that first presented to an ophthalmologist have been described.2,3,4,5,6
The typical ophthalmoscopic findings in the various blood dyscrasias are not pathognomonic and may actually be observed in many different local and systemic diseases involving the eye (i.e., diabetes, hypertension, collagen vascular disease). However, the pattern and distribution of the retinal findings in blood dyscrasias are characteristic. If one identifies these characteristic findings on ophthalmoscopy, then ancillary testing may reveal a blood dyscrasia and allow for early referral for treatment by the appropriate medical subspecialist. Therefore, the benefit of a careful ophthalmic examination in the context of a blood dyscrasia is twofold: The ophthalmologist may play a critical role in the early diagnosis of a systemic disease as well as monitor the potential ocular complications of a previously diagnosed condition.
Retinal Hemorrhages
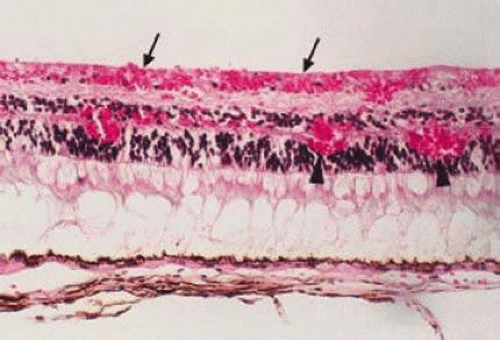
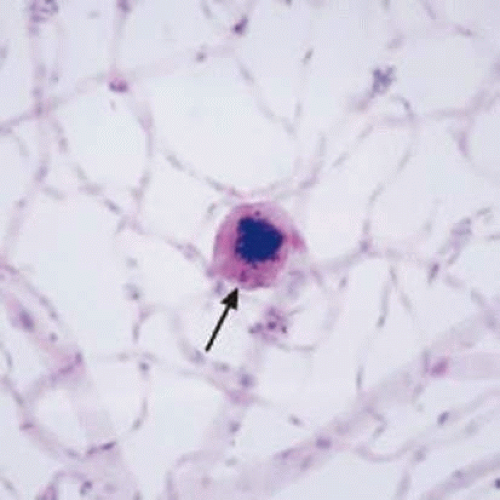
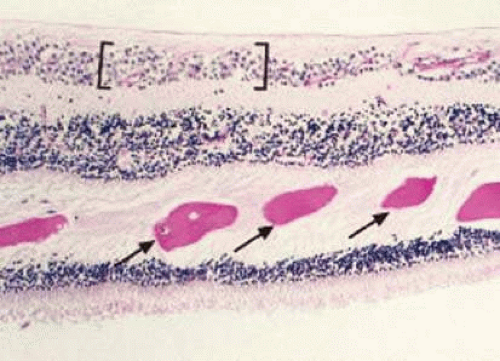
Retinal hemorrhages may take various configurations based on their location within the retina. Flame-shaped and dot-blot hemorrhages are the most commonly encountered intraretinal hemorrhages in blood dyscrasias (Fig. 18-1). Flame-shaped hemorrhages are located in the nerve fiber layer of the retina, and dot-blot hemorrhages are typically located in the inner nuclear and outer plexiform layers (Fig. 18-2). Large blot or boat-shaped hemorrhages may be observed and are present beneath the internal limiting membrane (ILM) of the retina (i.e., sub-ILM hemorrhage) (Fig. 18-3). These large superficial retinal hemorrhages may break through the ILM and extend into the vitreous cavity (Fig. 18-4). White-centered hemorrhages (Fig. 18-1) and, much less commonly, red-centered infiltrates (Fig. 18-5) may be seen in the retinae of patients with a blood dyscrasia. The white center may be associated with a leukemic embolus (Fig. 18-6) or more commonly a platelet-fibrin thrombus.7 The red center is blood, which may be associated with a cotton-wool spot (CWS) or a leukemic retinal infiltrate.
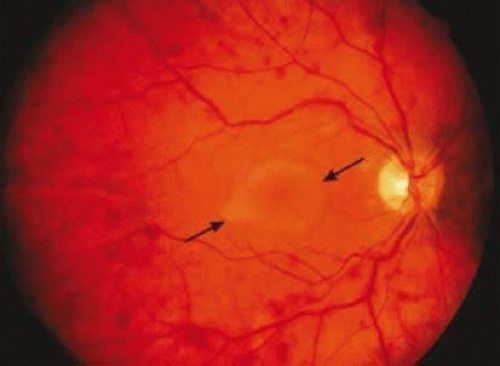
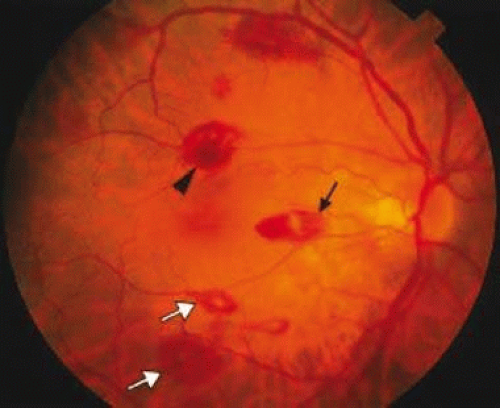 Figure 18-1. Intraretinal hemorrhages in acute myelocytic leukemia (AML) and anemia. Note the flame-shaped (arrow), blot (arrowhead), and white-centered (open arrows) hemorrhages. |
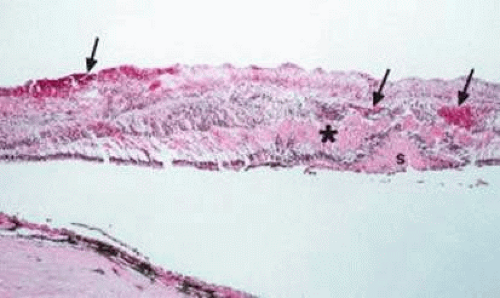
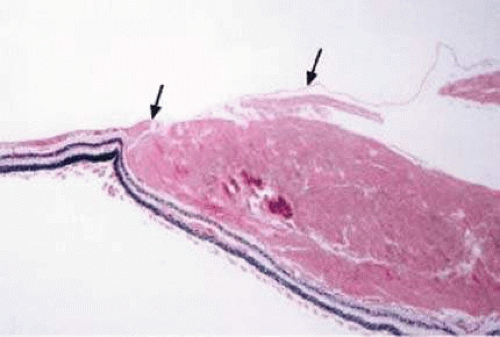 Figure 18-3. Histopathology of subinternal limiting membrane (sub-ILM) hemorrhage. Note the dense blood beneath the ILM (arrows) of the retina. |
Retinal hemorrhages were first described in patients with a blood disease by Liebreich in 1863.8 Since this time, retinal hemorrhages have been described in various blood dyscrasias including but not limited to: acute myeloid leukemia,9 chronic myelogenous leukemia,5 chronic lymphocytic leukemia,10 idiopathic thrombocytopenic purpura,11 aplastic anemia,12 pernicious anemia,13 and autoimmune hemolytic anemia.14 Factors that contribute to the development of retinal hemorrhages in the setting of blood dyscrasias include anemia, thrombocytopenia, elevated white blood cell count, and blood or serum hyperviscosity.
In a study of 67 patients with anemia and/or thrombocytopenia, Rubenstein et al15 found that retinal hemorrhage is much more likely to occur when anemia is accompanied by thrombocytopenia than when either is present alone. In a study of 152 patients with various blood diseases, Holt and Gordon-Smith8 reported that retinal hemorrhages in leukemic patients were observed most frequently when severe anemia, severe thrombocytopenia, or a high percentage of circulating blast cells were also present. In a prospective study of 117 patients with acute leukemia, Guyer et al16 reported that thrombocytopenia is the most important factor in the pathogenesis of intraretinal hemorrhages. Consistent with this observation, Reddy and Jackson17 —in a study of 127 patients with acute leukemia—found that intraretinal hemorrhages were significantly associated with high white count and low platelet count. Additionally, in a study of 41 patients with myelodysplastic syndrome (a group of preleukemic disorders) retinal hemorrhage significantly depended on platelet count.18 Finally, in a study of 226 patients, retinal hemorrhages were seen in all patients with both severe anemia and severe thrombocytopenia.1 On the contrary, Jackson et al19 found no association between intraretinal hemorrhages and the hemoglobin level or platelet count in 63 newly diagnosed acute leukemia patients. The same authors reported a higher median white blood cell count in patients with intraretinal hemorrhages than in those without intraretinal hemorrhages and concluded that a high white blood cell count may be at least as important as anemia and thrombocytopenia in the pathogenesis of retinopathy in acute leukemia. However, Keresh et al20 noted no correlation with presence of retinopathy and hematocrit and white blood cell counts in patients with myeloid leukemia.
Reddy and Jackson17 observed white-centered hemorrhages in 31% of the 127 patients studied with acute leukemia. In this study, higher white cell counts but not presence of anemia or thrombocytopenia were associated with white-centered hemorrhages.17 In a study of 122 patients with AML, 40 had retinal hemorrhages; dot-shaped and flame-shaped were the commonest types of retinal hemorrhage in this study.21 Interestingly, it has been reported that retinal hemorrhage in the setting of leukemia did not correlate with patient prognosis.8
In a 4-year prospective study of 82 patients with acute leukemia, Jackson et al22 found that patients with a macular hemorrhage were at significantly greater risk for developing intracranial hemorrhage (particularly in the promyelocytic [M3] subtype of acute myeloid leukemia [AML]) within the first 30 days following diagnosis compared with patients without a macular hemorrhage. Based on these findings, the authors recommended that patients with a macular hemorrhage be monitored intensively for the development of intracranial hemorrhage and receive priority in the allocation of platelets when platelets are in short supply. Richards et al23 also reported a high incidence of intraocular hemorrhage in patients with acute promyelocytic leukemia (M3 subtype of AML) and found no consistent detectable abnormalities in the hematologic parameters or coagulation studies predictive of ocular hemorrhage.
In a pathologic study of the eyes of 76 patients who died of leukemia and allied disorders, Allen and Straatsma24 found that retinal hemorrhage was the most frequent and serious ocular complication and that the most significant hemorrhages occurred in the acute forms of leukemia. Other large clinical studies have also listed retinal hemorrhage among the commonest retinal lesions in leukemic patients.1,17,21,25
Microaneurysms
Microaneurysms are small outpouchings in the retinal capillary wall that appear as tiny red dots in the retina on ophthalmoscopic examination. They are classically seen in the posterior fundus in diabetic retinopathy. Microaneurysms have also been reported in leukemia and plasma cell dyscrasias.26,27,28,29 Interestingly, the microaneurysms in blood dyscrasias tend to be located in the peripheral retina, in contrast to the location in the posterior retina in diabetic retinopathy. In a pathologic study using trypsin digestion of flat mounts of the retina, Duke et al27 emphasized the relative preservation of pericytes in patients with chronic leukemia as compared with the marked loss of pericytes in diabetic retinopathy. Figure 18-7 shows the typical microaneurysms in the retinal periphery in a flat mount preparation with trypsin digestion in a patient with a blood dyscrasia (multiple myeloma [MM]). Common pathologic features between the microaneurysms of diabetes and blood dyscrasias include the globular shape of the lesion, the location predominantly on the venous side of the capillary, and the presence of intramural and intraluminal periodic acid–Schiff (PAS)-positive deposits.27 Factors that may play a role in the formation of microaneurysms in the setting of blood dyscrasias include increased venous pressure (i.e., central retinal vein occlusion [CRVO]), increased blood viscosity with a secondary increase in venous pressure (i.e., hyperviscosity syndrome associated with plasma cell dyscrasias), and anoxia (i.e., severe anemia). Of note, in a study of 127 patients with acute leukemia, Reddy and Jackson17 did not observe microaneurysms in any of their patients.
Hard Exudates
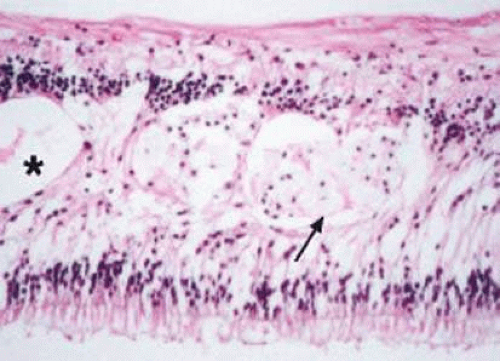
Hard exudates are refractile, yellowish deposits of proteins and lipids that are derived from incompetent or leaky retinal capillaries. Hyperpermeability of the retinal capillaries may be seen in hyperviscosity syndromes, retinal venous congestion, and CRVO secondary to plasma cell dyscrasias.30,31,32 The hard exudates that result from this capillary hyperpermeability are localized predominantly to the outer plexiform layer of the retina (Fig. 18-8).
Retinal Edema
In addition to the formation of hard exudates, hyperpermeable or leaky retinal capillaries can lead to focal or generalized retinal edema. Mild opacification or graying of the normally transparent retina is noted in areas of retinal edema on ophthalmoscopic examination. Cystic degeneration of the retina may result from long-standing retinal edema. Retinal edema localized to the macular region may manifest as cystoid macular edema (CME). Histologically in CME, the cysts often contain eosinophilic proteinaceous material and are located in the outer plexiform and, to a lesser extent, in the inner nuclear layers of the retina (Fig. 18-9). Occasionally, aggregates of lipid-laden macrophages are observed in the regions of the cystic degeneration (Fig. 18-9). As with hard exudates, retinal edema may be observed in hyperviscosity syndromes induced by the blood dyscrasias or in secondary retinal vein occlusions. The CME observed in blood dyscrasias may respond to specific treatment for CME (i.e., topical or systemic acetazolamide, anti-VEGF [vascular endothelial growth factor] therapy)33 and may resolve completely with treatment of the underlying disease process (i.e., after bone marrow transplantation in chronic myeloid leukemia [CML]).34
Cotton-Wool Spots (CWSs)
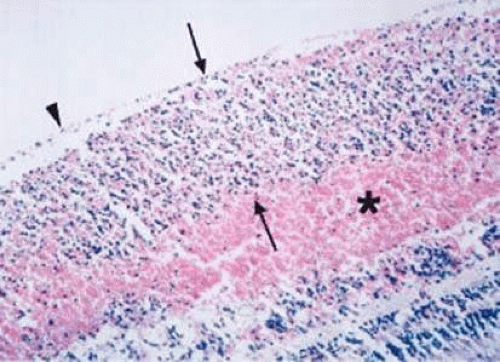
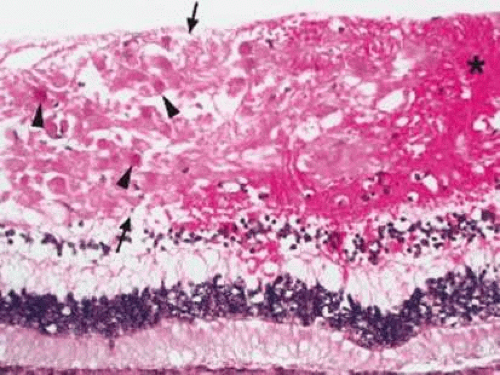
CWSs, also known as soft exudates, are microinfarctions in the nerve fiber layer. The normal flow of axoplasm is blocked in response to focal retinal ischemia caused by occlusion of a precapillary arteriole.35 Clinically, CWSs are superficial whitish retinal lesions with irregular feathery borders that may be associated with small retinal hemorrhages. With resolution, the CWSs will fade and an area of retinal depression may develop secondary to inner retinal ischemic atrophy. Histologically, CWSs are characterized by fusiform thickening of the nerve fiber layer with globular cytoid bodies (Fig. 18-10)—swollen ganglion cell axons with degenerated cellular organelles (i.e., mitochondria, neurofilaments, endoplasmic reticulum). CWSs are often encountered in the retinopathy associated with blood dyscrasias. In one report, the presence of a single CWS in each eye of a patient in the absence of diabetes and systemic hypertension led to the diagnosis of MM.36 In a prospective study of 54 newly diagnosed patients with acute leukemia, Abu El-Asrar et al37 reported that patients with CWSs had significantly lower mean and median survival times than those patients without CWSs. Thus in this study, the presence of CWSs was a poor prognostic sign for survival in acute leukemia.
The presence of severe anemia may be a risk factor for the development of CWSs in blood dyscrasias. Holt and Gordon-Smith8 observed CWSs in 14 patients with severe anemia with a mean hemoglobin concentration of 5.6 g/100 mL. Additionally, in a study of 18 patients with aplastic anemia, CWSs were observed in 38% of patients.12 In contrast, Guyer et al16 found no statistically significant association between the presence of CWSs and hematologic parameters (including the hematocrit) in their prospective series. CWSs have been observed in the following: 11/122 patients with AML,21 18/288 patients with leukemia,38 15/127 with acute leukemia, and in 21/53 patients with myeloid leukemia.20 CWSs may result from occlusion of the precapillary arterioles by leukemic cells or by platelet-fibrin thrombi.
Retinal Vascular Changes
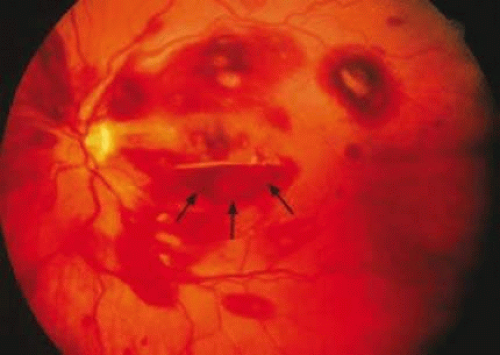
Retinal venous dilatation and tortuosity similar to, and in some cases indistinguishable from, the ophthalmoscopic findings in CRVO may be observed in blood dyscrasias, particularly the plasma cell dyscrasias (i.e., Waldenström macroglobulinemia (WM), monoclonal gammopathies, and less commonly MM].26,30,31,32 With venous distention, arteriovenous nicking may become more apparent and give rise to the “sausage link” appearance of the retinal veins. The classical picture of venous stasis retinopathy associated with hyperviscosity is seen in WM (Fig. 18-11). In a prospective study of 120 patients with newly diagnosed leukemia of all cell types, Schachat et al39 diagnosed a CRVO in 5 patients—all of whom had myeloid leukemia with extremely high white blood cell counts or platelet counts—and attributed the venous occlusion to blood hyperviscosity. The pathologic findings in venous stasis retinopathy associated with blood dyscrasias may be indistinguishable from those found in CRVO (Fig. 18-12).
Perivascular sheathing may be observed in the ocular fundus in patients with blood dyscrasias. The apparent opacification or loss of transparency of the retinal blood vessel wall may be due to infiltration by benign inflammatory cells in response to immune complex or other protein (e.g., amyloid, immunoglobulin heavy or light chains) deposition, direct infiltration by leukemic cells, or fibrosis secondary to hypertensive retinal vascular disease. Perivascular sheathing and a clinical picture of retinal vasculitis (with leakage from retinal vessels on fluorescein angiography) have been prominent ocular findings in patients presenting with certain blood dyscrasias, including human T-lymphotropic virus 1 (HTLV-1)-associated adult T-cell leukemia/lymphoma,40,41 cryoglobulinemia,42 hairy cell leukemia43 and, rarely, MM.44 Kim et al45 described a patient with relapsing acute lymphoblastic leukemia who presented with a retinal vasculopathy resembling frosted branch angiitis and an infiltrative optic neuropathy, which resolved with local radiation and intrathecal chemotherapy.
Pallor of the Ocular Fundus
The normal color of the ocular fundus is derived from the retinal pigment epithelium (RPE), the choroidal melanocytes, and the blood in the retinal and choroidal vasculature. The retina is normally transparent. In patients with severe anemia, the fundus may appear pale and the retinal vessels may be less red than normal.
Optic Disc Edema
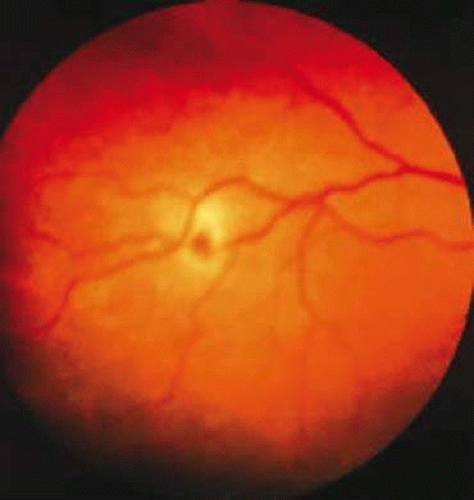
In optic disc edema, nerve fiber swelling in the optic disc and peripapillary retina is present and often associated with flame-shaped hemorrhages, whitish punctate lesions (secondary to obstructed axoplasmic flow), and peripapillary CWSs. On ophthalmoscopy, the optic disc and peripapillary retina appear elevated and the normally well-defined margins of the optic disc are blurred. Optic disc edema is usually bilateral and may be due to elevated intracranial pressure (i.e., papilledema) (Fig. 18-13) secondary to hemorrhage or a mass effect associated with the blood dyscrasia (i.e., meningeal infiltration, granulocytic sarcoma). The disc edema may also be secondary to serum (blood) hyperviscosity and may resemble that seen in CRVO. Less commonly, direct infiltration of the optic nerve or optic disc may cause marked optic disc edema.
Rosenthal46 emphasized that optic nerve infiltration occurs predominantly in children with acute lymphocytic leukemia (ALL) and must be differentiated clinically from papilledema. Patients with leukemic infiltration of the prelaminar optic nerve typically have marked swelling of the optic disc with a fluffy superficial infiltrate and variable hemorrhage (Fig. 18-14). The visual acuity may be minimally or severely affected. With retrolaminar optic nerve infiltration, moderate to marked disc elevation and edema with variable hemorrhage may be present and marked vision loss is usually observed. Optic nerve infiltration may occur despite prophylactic brain irradiation in leukemia because of the shields used to protect the eyes.47 In general, optic nerve infiltration by leukemia responds well to radiotherapy (with or without intrathecal chemotherapy). Brown et al48 reported the case of a patient with acute promyelocytic leukemia and optic disc infiltration who showed complete resolution with oral all trans-retinoic acid alone.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree