Retinoblastoma and Simulating Lesions
Ralph C. Eagle Jr.
Retinoblastoma is the most common intraocular tumor of childhood. In Asia, Africa, and South America, where uveal melanoma is relatively rare, retinoblastoma probably is the most common primary intraocular tumor. In Europe and the United States, it is second in overall prevalence, reflecting the propensity of lightly pigmented adult Europeans to develop uveal melanoma. The incidence of retinoblastoma has been estimated to be 1 in 15,000 to 1 in 34,000 births. Retinoblastoma is cosmopolitan and affects all races. An increased incidence of sporadic retinoblastoma in some socioeconomic groups and locales suggest that environmental factors such as poor nutrition may play a role in its pathogenesis. Both sexes are affected equally, and the tumor has no predilection for the right or left eye. The average age at diagnosis is 18 months, and retinoblastoma is quite uncommon after 4 years of age. Rare cases do occur in older children and have been reported in adults as well. Of 400 consecutive patients with retinoblastoma evaluated on the Ocular Oncology Service at the Wills Eye Hospital, 8.5% were older than 5 years at the time of initial diagnosis. All had unilateral sporadic retinoblastoma.
Retinoblastoma has many unusual and interesting clinical characteristics. Foremost, it is a hereditary cancer: 5% to 10% of the tumors are inherited. In affected families, retinoblastoma is transmitted as an autosomal dominant trait, and the tumor frequently affects both eyes. Bilaterality is a clinical marker for potentially transmissible disease. About one third of all retinoblastomas are bilateral; the additional bilateral cases are caused by new germline mutations. The great majority of retinoblastomas arise sporadically in patients with a negative family history. About 75% of the sporadic tumors are caused by somatic mutations and are not transmissible, while the remaining 25% arise from new germline mutations and can be passed on to offspring. Sporadic tumors caused by somatic mutations are always unilateral. Unfortunately, unilateral involvement does not exclude heritable disease: about 10% to 15% of infants with unilateral sporadic tumors have germinal mutations. The retinoblastoma gene is discussed below.
Clinical Presentation
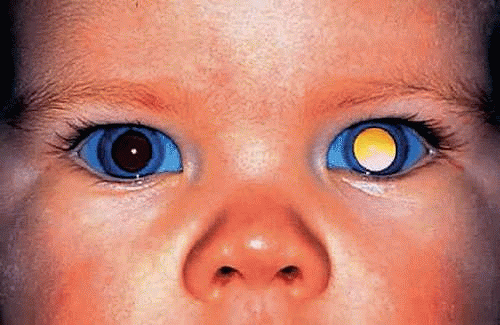
The clinical presentation of retinoblastoma depends on the size, location, growth pattern, and stage of the lesion when the parents consult a physician and the availability and sophistication of medical care in the community. In the United States and Europe, about 90% of patients present with leukocoria, an abnormal, typically white pupillary reflex (Figs. 1,2,3,4). Leukocoria is the classic clinical manifestation of retinoblastoma. In 1767, Hayes fancifully likened this appearance to the illuminated tapetal light reflex of the cat (amaurotic cat’s-eye reflex). The white pupillary reflex is caused by tumor in the vitreous cavity (endophytic tumors) or total retinal detachment (exophytic tumors).
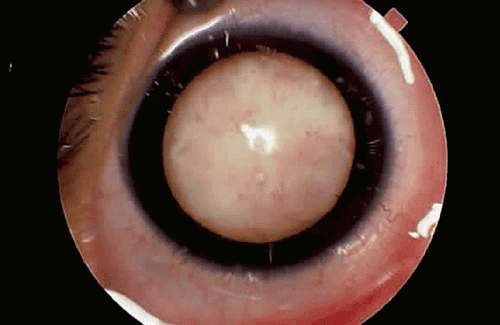 Figure 3. Leukocoria, endophytic retinoblastoma. Tumor growing in the vitreous cavity produces white pupillary reflex. (Photo courtesy of Jerry A. Shields, M.D., Wills Eye Hospital.) |
Strabismus or ocular misalignment occurs in about one third of cases and may herald a relatively small tumor that involves the fovea. For this reason, careful ophthalmoscopy should be performed on all children with strabismus to exclude retinoblastoma or some other significant retinal pathology. About half of the eyes enucleated for retinoblastoma have iris neovascularization, which may produce neovascular glaucoma, iris heterochromia, and even secondary buphthalmos. Pupillary block glaucoma caused by anterior displacement of the lens–iris diaphragm may be found in eyes with exophytic tumors and total bullous retinal detachments.
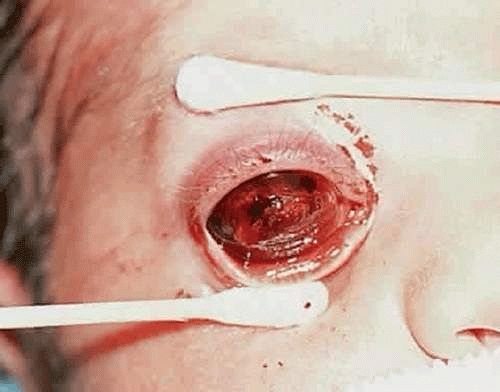
Endophytic retinoblastoma and the rare diffuse infiltrating variant of the disease often seed the anterior chamber and can form a pseudohypopyon of tumor cells that may be initially misdiagnosed as inflammation. In some cases, the tumor in the anterior chamber forms clumps or seeds. In other instances, there may be infiltration of the iris stroma. An orbital cellulitislike picture is another type of pseudoinflammatory presentation (Fig. 5). In such cases, histopathologic examination usually reveals advanced neovascular glaucoma that has caused extensive infarction of both the tumor and other intraocular structures.
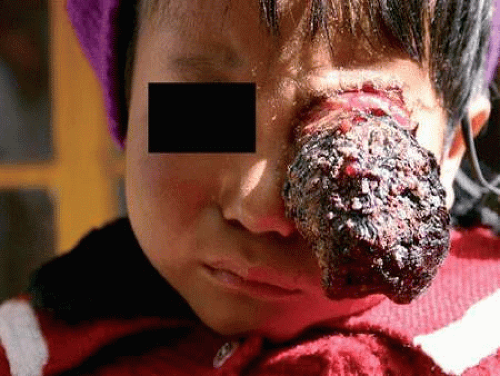
In underdeveloped countries, patients may present in the late stages of the disease after the tumor has extended extraocularly, causing exophthalmos and an orbital mass (Fig. 6).
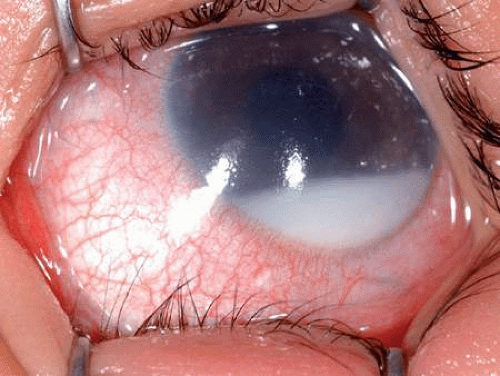
Rare cases of clinically manifest congenital retinoblastoma have been reported. Such cases of congenital retinoblastoma may present with a massive hyphema and an enlarged ectatic cornea that spontaneously perforates (Fig. 7). It has been estimated that half of retinoblastomas actually may be present at birth but are inapparent clinically.
Natural History
Retinoblastoma is a highly malignant neoplasm that grows relentlessly and almost always has a fatal outcome if untreated. The tumor arises from retinal cells and initially is confined to the retina. Clinically, there appears to be a direct correlation between the age of the patient and retinal topography that follows a central to peripheral distribution. Tumors in the macular region present earliest, while tumors in the more peripheral retina develop later. In Abramson’s series of 565 eyes with bilateral retinoblastoma, all macular tumors and most retinoblastomas of the posterior pole were detected before age 24 months.
As a retinoblastoma grows, the tumor invades the vitreous cavity and/or the subretinal space. Endophytic retinoblastomas that arise from the inner layers of the retina seed the inner surface of the retina, the vitreous cavity, and eventually the anterior chamber, where the tumor cells may infiltrate the iris stroma or trabecular meshwork or form a pseudohypopyon in the inferior angle (Fig. 8). Exophytic tumors that arise from the outer retinal layers typically cause a serous retinal detachment. The retinal detachment is typically high and bullous in configuration, and the retina often is plastered against the posterior surface of the lens (Fig. 9). Such eyes frequently have secondary closed-angle glaucoma due to pupillary block. The subretinal fluid often contains seeds of tumor, and a sheet of tumor cells may blanket the inner surface of the retinal pigment epithelium (RPE). Retinoblastoma cells often invade the space between the RPE and Bruch’s membrane, causing focal RPE detachments. Bruch’s membrane initially serves as a mechanical barrier to invasion. Eventually, however, the retinoblastoma cells breach Bruch’s membrane and invade the choroidal stroma (Fig. 10). The uveal tract’s rich blood supply nurtures the proliferating tumor cells and serve as a major route for distant hematogenous dissemination. Tumor necrosis and a perivascular growth pattern occur infrequently when the tumor grows in richly vascularized uveal tissue but can occur when uveal invasion is massive. Although multivariant analysis suggests that the presence of extrascleral extension is a much better indicator of poor prognosis, massive uveal invasion appears to carry a risk for distant metastasis. In one series, the mortality rate was about 25% with minimal choroidal invasion; this rose to 65% if the invasion was massive. Involvement of the anterior chamber, iris stromal, and trabecular meshwork also are thought to affect prognosis adversely, but the actual magnitude of this effect is uncertain. A prospective multicenter clinical trial sponsored by the Children’s Oncology Group currently is evaluating the effect of histopathologic prognostic factors such as uveal, anterior segment, and optic nerve invasion on prognosis.
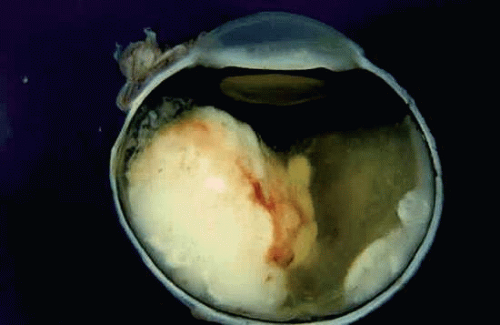 Figure 8. Retinoblastoma, endophytic growth pattern. A white encephaloid mass arises from the inner retina and invades the vitreous cavity. Most of the retina remains attached. |
Metastatic disease usually is evident within 1 or 2 years after therapy. Late metastasis is so rare in retinoblastoma that it should raise the possibility of a second independent primary tumor.
After a retinoblastoma has filled the globe and destroyed its contents, it extends extraocularly. In the anterior segment, the tumor extends extraocularly through the aqueous outflow pathways, preexisting emissarial canals, or perforations in the cornea. Posterior segment tumors that have invaded the choroid can extend extraocularly along emissarial canals or can invade the orbit by directly infiltrating and destroying the sclera. Patients from underdeveloped countries often present with orbital masses of tumor tissue, which contain ocular remnants, mainly sclera. Secondary buphthalmos and staphylomas caused by secondary glaucoma can facilitate extrascleral extension. Secondary closed-angle glaucoma caused by pupillary block or iris neovascularization is relatively common in eyes with retinoblastoma. Fifty percent of eyes enucleated for retinoblastoma have iris neovascularization. Angiogenic factors such as vascular endothelial growth factor (VEGF) produced by the tumor cells or the ischemic retina are the probable cause of the iris neovascularization.
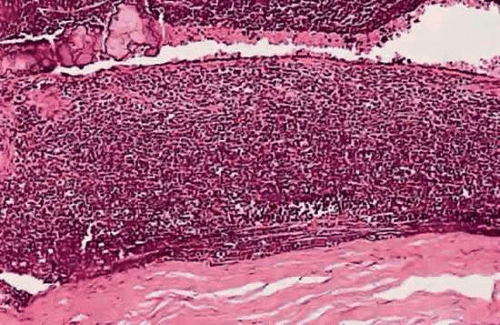
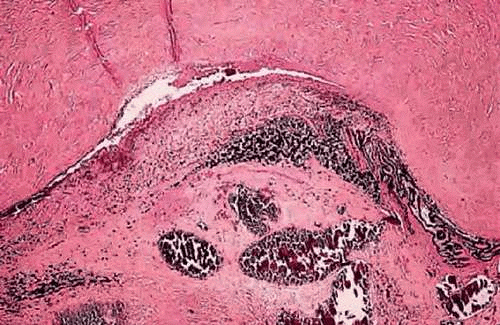
The optic nerve is the most common avenue for extraocular extension by retinoblastoma. Retinoblastoma has a marked proclivity to invade the optic nerve (Fig. 11). Some degree of optic nerve invasion was present in 29.5% of 814 retinoblastomas reported by Magramm and coworkers. In this respect, the behavior of retinoblastoma contrasts sharply with that of choroidal melanoma, which invades the optic nerve only 5% of the time. The tumor may travel to the brain along the parenchyma of the optic nerve, via the subarachnoid space, or both. Rare tumors can travel around the chiasm and reach the posterior aspect of the fellow eye. Retinoblastoma cells also can be disseminated widely by the cerebrospinal fluid in the subarachnoid space.
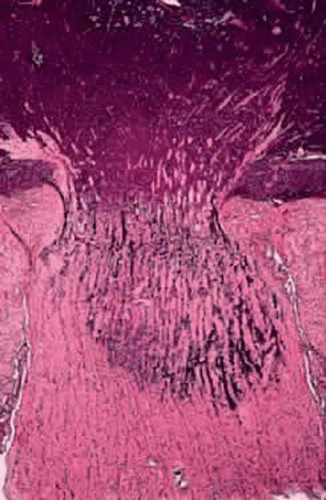 Figure 11. Retinoblastoma, grade III optic nerve invasion. The retrolaminar parenchyma of the optic nerve contains a basophilic infiltrate of retinoblastoma cells. (H&E, 10×) |
Optic nerve invasion is an extremely important prognostic factor in retinoblastoma. Mortality rates correlate directly with the depth of optic nerve invasion. In Magramm’s series, the mortality rate was 10% if there was superficial invasion of the nerve head only (grade I) and 29% for involvement up to and including the lamina cribrosa (grade II). It rose to 42% when there was retrolaminar invasion (grade III) and 78% when the tumor extended to the surgical margin (grade IV). It is therefore obvious that the surgeon should try to obtain as a long a segment of optic nerve as possible when enucleating an eye that is known or suspected to contain a retinoblastoma. Furthermore, it is prudent that enucleation should not be performed by an inexperienced surgeon, if at all possible. Incomplete removal of tumor in the nerve, or even worse, soiling of orbital tissue with tumor caused by inadvertent “button-holing” of the sclera, can have disastrous, fatal consequences.
The pathologist must pay particularly careful attention to the optic nerve during his or her evaluation. During gross dissection, it is important to to avoid contaminating this important surgical margin with tumor. The optic nerve margin should be marked with indelible pencil or India ink before sectioning, and the nerve should always be removed before the globe is opened and submitted in a separate cassette. This is particularly important if fresh tumor is harvested in the operating room. In such cases, it is imperative that the nerve be submitted separately. Blood staining, obliquity of sectioning, and slight crushing serve to identify the true surgical margin during specimen grossing.
In addition to evaluating the transverse section of the surgical margin, it is also extremely important to make sure that longitudinal sections through the center of the optic nerve head are prepared from the main P-O block. If possible, several levels should be obtained. Examination of longitudinal sections is important because invasion of the optic nerve posterior to the lamina cribrosa is tantamount to to extraocular extension of the tumor and is an indication for adjuvant chemotherapy in many centers.
Retinoblastoma typically metastasizes hematogenously to lungs, bones, brain, and other organs after the tumor cells gain access to the blood vessels in the uvea or orbital tissues. Cervical and preauricular adenopathy can develop when tumors with extensive anterior segment involvement gain access to lymphatics in the conjunctival stroma. Lymphatics are not present in the orbit.
Retinoblastoma occasionally undergoes spontaneous regression. In the past, the incidence of spontaneous regression was estimated to be 1%, which is greater than any other malignant tumor. However, it currently is believed that many lesions that once were classified as spontaneously regressed retinoblastomas actually are benign, generally nonprogressive retinal tumors caused by mutations in the retinoblastoma gene called retinomas or retinocytomas. There is some controversy in the literature about the best terminology for these lesions. In 1982, Gallie and coworkers suggested the term retinoma in a clinical study without confirmatory histopathology. A histopathologic study published in 1983 by Margo and Zimmerman designated identical lesions as retinocytoma, based on terminology of neural tumors of the pineal gland. The term retinocytoma is favored by many pathologists and is used in major textbooks of ophthalmic pathology, including the Armed Forces Institute of Pathology fascicle on Tumors of the Eye and Ocular Adnexa. Gallie objects to retinocytoma, however, because that term previously was applied to a rosette-rich variant of retinoblastoma. Others consider the term retinoma to be too nonspecific. In this chapter, an attempt will be made to avoid this controversy by using the term retinoma/retinocytoma. The term spontaneously arrested retinoblastoma, favored by some, is more appropriate than spontaneously regressed retinoblastoma. Based on the results of molecular genetic studies, Dimaris recently has redefined retinoma as a precancerous lesion characterized by the loss of function of both copies of the RB1 gene but lacking the additional genomic changes characteristic of retinoblastoma.
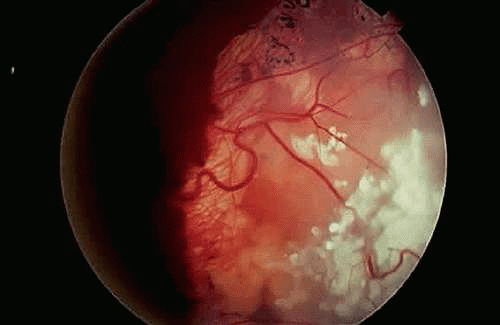
Initially, retinomas or retinocytomas were thought to represent spontaneously regressed retinoblastomas on clinical grounds because they resemble retinoblastomas that have regressed after radiation therapy (Fig. 12). They have a translucent “fish-flesh” appearance, contain abundant calcification that has been likened to cottage cheese, and are surrounded by a ring of RPE depigmentation. Retinomas/retinocytomas generally are small tumors that are found in eyes that retain useful vision. They may be found incidentally or are discovered in a parent or sibling when the detection of retinoblastoma in a child prompts examination of other family members. Rare cases of retinocytoma have been observed to undergo malignant transformation into retinoblastoma. Histopathologic examination in one such case confirmed the presence of benign cytology and photoreceptor differentiation in the original basal part of the tumor (Fig. 13). Retinocytomas are relatively resistant to radiation, as are other benign tumors. Therefore, it is not surprising that foci of photoreceptor differentiation are found more often in eyes that are enucleated after external-beam radiotherapy or chemotherapy.
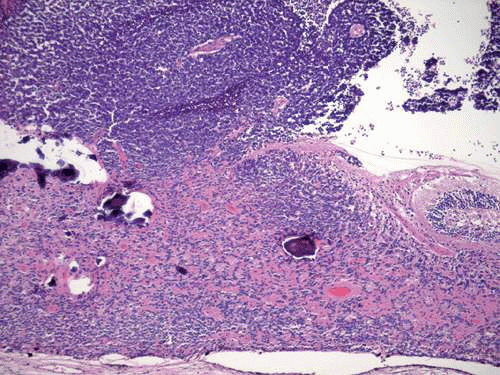 Figure 13. Histopathology of retinocytoma seen in Figure 12. Eye was enucleated after the lesion underwent malignant transformation. Composed of photoreceptor differentiation, the basal part of tumor is relatively eosinophilic compared with the overlying area of malignant transformation (above), which appears blue and contains vessels cuffed by retinoblastoma cells. (H&E, 25×) |
Bona fide cases of spontaneous regression of retinoblastoma do occur but are rare. They typically are found in the setting of a blind, phthisical eye. It is thought that both the tumor regression and phthisis bulbi are caused by extensive ischemic necrosis that affects both the tumor and normal intraocular structure in an eye with neovascular glaucoma and markedly elevated intraocular pressure (Fig. 14). Infants with extensive ischemic necrosis of both tumor and eye may present with inflammatory signs and may be misdiagnosed initially as having orbital cellulitis.
Gross Pathology
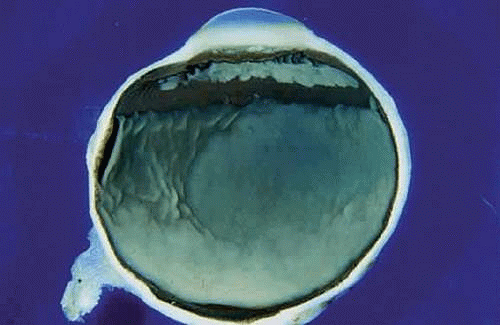
The gross appearance of retinoblastoma is somewhat variable, reflecting the stage of the disease at enucleation. Because the retina can be considered to be a peripheral colony of brain cells, it is not surprising that the tumor characteristically has a white encephaloid, or brainlike, appearance grossly. The whitish tumor thickens, replaces and destroys part or all of the retina, and may totally fill the vitreous cavity. The mass typically contains lighter foci of necrotic tumor or calcification. Gritty crystals of calcium apatite or lemon-yellow pigmentation may be encountered in some advanced lesions.
Totally necrotic retinoblastomas that have undergone acute infarction occasionally are encountered. Such tumors typically have a blood-tinged, orange, or soupy and grayish necrotic appearance grossly. Infarction caused by severe neovascular glaucoma also affects other intraocular structures, including the iris, ciliary body, and retina, causing pigment dispersion and inflammatory signs and symptoms. Affected infants may be initially misdiagnosed as having orbital cellulitis.
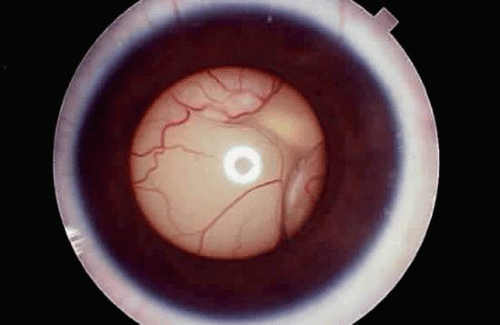
Several tumor growth patterns are recognized macroscopically. Exophytic retinoblastomas arise from the outer retinal layers, proliferate in the subretinal space, and cause secondary retinal detachment (Fig. 7). The detached retina is typically highly elevated, and its vessels are visible behind the lens on clinical examination. Eyes with exophytic retinoblastomas often develop secondary pupillary block glaucoma. Clinically, exophytic retinoblastomas can be confused with simulating lesions that cause retinal detachment, such as Coats disease.
Endophytic retinoblastomas grow into the vitreous cavity, obscuring the retina, which remains attached (Fig. 6). Tumor fills the vitreous cavity by direct extension and seeding. Retinal vessels are not evident in the white pupil on slit-lamp examination. A pseudohypopyon of tumor cells can develop if seeding involves the anterior chamber. Hence, endophytic tumors can be confused with primary inflammatory disorders such as toxocariasis, mycotic endophthalmitis, or granulomatous uveitis. As a rule, purely endophytic or exophytic retinoblastomas are uncommon. Most tumors exhibit foci of both growth patterns.
A diffuse infiltrative growth pattern occurs in <2% of cases. Such diffuse infiltrative tumors are often overlooked or misdiagnosed because they diffusely thicken the retina and do not form a discrete tumefaction (Fig. 15). Diffuse infiltrative retinoblastoma occurs in older children, whose mean age at diagnosis is about 6 years. Such tumors are invariably unilateral and sporadic. Like endophytic retinoblastoma, diffuse infiltrative retinoblastoma typically presents with anterior segment seeding and pseudohypopyon; it is often misdiagnosed as a primary inflammatory disorder or juvenile xanthogranuloma. Some have suggested that this variant of retinoblastoma may have a relatively good prognosis despite delays in diagnosis, but this issue remains controversial.
Retinoblastoma is a friable, poorly cohesive neoplasm that can readily disperse or seed widely. Tumor seeds deposited on the inner or outer surfaces of the retina frequently give rise to secondary tumors. The latter may be impossible to differentiate grossly or histologically from multifocal primary lesions. This is unfortunate, because true multifocal involvement of the retina signifies hereditary, potentially transmissible disease.
Histopathology
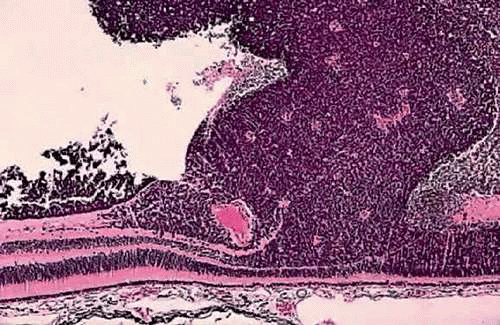
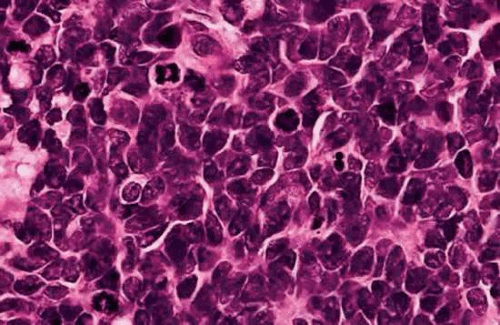
Retinoblastoma has a characteristic appearance and staining pattern in routine sections stained with hematoxylin and eosin (H&E) that instantly suggests the diagnosis under low magnification. A basophilic mass arises from and destroys the retina and fills part or all of the vitreous cavity (Fig. 16). The basophilic tumor usually contains eosinophilic and reddish-purple foci (Fig. 17).
The basophilic areas are composed of viable retinoblastoma cells, which appear blue because they are poorly differentiated neuroblastic cells that have scanty cytoplasm and intensely basophilic nuclei (Fig. 18). Many mitoses and fragments of apoptotic nuclear debris usually are present. Retinoblastoma grows rapidly and has a marked propensity to outgrow its blood supply and undergo spontaneous coagulative necrosis. This usually occurs when the proliferating cells have extended about 90 to 110 μ away from a blood vessel (Fig. 17). The necrotic tumor cells lose their basophilic nuclear DNA and become pink or eosinophilic. The residual viable cells typically form cuffs or sleeves around vessels, imparting a multilobulated or papillary appearance to some tumors. These perivascular cuffs were called pseudorosettes by some in the past. Foci of dystrophic calcification develop in the necrotic parts of the tumor in many cases. Histopathologically, the calcific foci appear reddish-purple in H&E sections, and the presence of calcium can be confirmed by the von Kossa or alizarin red stains. Electron microscopy suggests that calcification probably begins in the mitochondria of necrotic cells. Clinically, the demonstration of calcification by ultrasonography or computed tomography can help to differentiate retinoblastoma from other simulating lesions.
Intensely basophilic deposits of DNA released from necrotic tumor cells are another characteristic histopathologic feature of retinoblastoma. DNA deposition generally is found in eyes with extensively necrotic tumors. Typically, the DNA deposits surround retinal or iris vessels or are found in the trabecular meshwork or in basement membranes such as the lens capsule or the internal limiting membrane of the retina.
Rosettes and Photoreceptor Differentiation
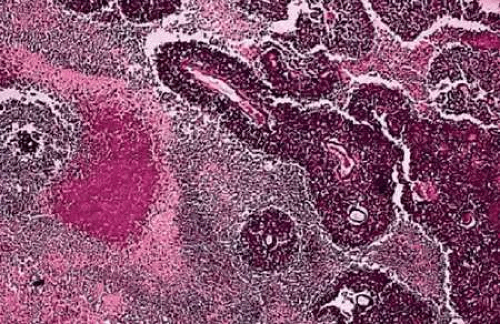
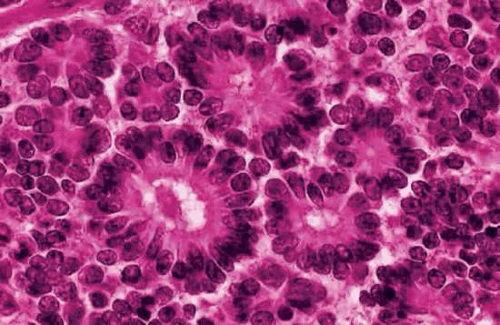
The most characteristic histopathologic feature of retinoblastoma is the rosette described by Simon Flexner in 1881 and Hugo Wintersteiner in 1897 (Fig. 19). Flexner-Wintersteiner rosettes represent an early attempt at retinal differentiation. Histologically, these rosettes are composed of a ring of cuboidal cells surrounding a central lumen. The lumen, which corresponds to the subretinal space, contains hyaluronidase-resistant acid mucopolysaccharide (AMP) similar to photoreceptor matrix AMP. The cells surrounding the lumen are joined near their apices by intercellular connections (zonulae adherentes), analogous to the external limiting membrane of the retina. Cilia, which exhibit the 9 + 0 pattern of microtubular doublets found in the central nervous system, project into the lumen. Cilia are hypothesized to be the precursor of photoreceptor outer segments.
The presence of many Flexner-Wintersteiner rosettes in a tumor may have prognostic significance. In one series, patients who had moderately well-differentiated tumors that contained abundant Flexner-Wintersteiner rosettes had about a sixfold better prognosis than those whose tumors lacked rosettes. Flexner-Wintersteiner rosettes are found more frequently in small, relatively early tumors and may be totally absent in large tumors, which usually are poorly differentiated. Flexner-Wintersteiner rosettes rarely are found in foci of metastatic retinoblastoma.
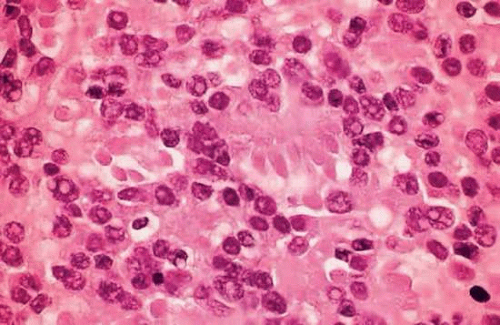
Another less-differentiated type of rosette that occurs occasionally in retinoblastoma was described by James Homer Wright (Fig. 20). Wright rosettes are indicative of neuroblastic differentiation. They lack a central lumen, and their constituent cells encompass a central tangle of neural filaments. Wright rosettes are relatively nonspecific. They also occur in neuroblastoma and are a characteristic feature of cerebellar medulloblastoma.
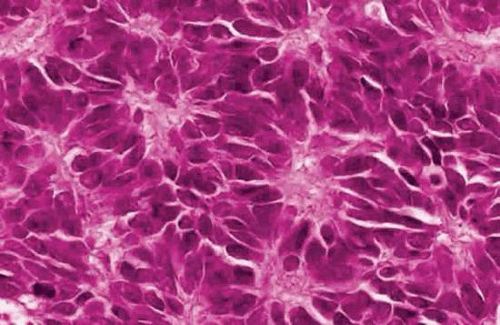 Figure 20. Wright rosettes. An irregular ring of nuclei encompasses a central tangle of neural processes. No lumen is present. (H&E, 250×) |
Fleurettes, described by Ts’o, Zimmerman, and Fine in 1970, are aggregates of neoplastic photoreceptors that represent a greater degree of retinal differentiation (Fig. 21). Found in approximately 6% of cases, fleurettes typically occur in a viable area of tumor that appears less cellular and relatively more eosinophilic than adjacent undifferentiated retinoblastoma during low-magnification microscopy of sections stained with H&E. The term fleurette denotes a bouquetlike arrangement of cytologically benign cells joined by a series of zonulae adherentes that may form a short segment of neoplastic external limiting membrane. Bulbous eosinophilic processes that represent abortive photoreceptor inner segments form the “flowers” of the bouquet. Electron microscopy occasionally discloses stacks of cellular membranes representing early outer segment differentiation. The demonstration and characterization of photoreceptor differentiation firmly established that retinoblastoma is not a glioma of the retina and affirmed that the adoption of the name retinoblastoma by the American Ophthalmological Society in 1926 at Frederick Verhoeff’s suggestion was indeed appropriate.
Foci of photoreceptor differentiation are found more frequently (40% of cases) in eyes that are enucleated after radiotherapy or chemoreduction. Presumably, the chemo- or radiotherapy preferentially kills the malignant part of the tumor and discloses the benign foci, which are relatively radio- or chemoresistant. Retinal tumors composed entirely of fleurettes are thought to be a benign variant of retinoblastoma that is incapable of metastasis. Such tumors are called retinomas or retinocytomas (see above). Compared with retinoblastoma, the cells comprising retinocytoma are quite bland, and mitotic activity is uncommon. In addition, calcification occurs in viable parts of retinoma/retinocytoma. Dimaras et al found that 15% of eyes enucleated for retinoblastoma contained foci of photoreceptor differentiation. They speculated that the incidence would be even higher if the tumors had been thoroughly sampled by serial sectioning. Furthermore, they postulate that these foci represent residual precursor retinomas/retinocytomas that have undergone malignant transformation during a stepwise progression to cancer and subsequently have been overgrown by the resultant retinoblastoma (see below).
Cell of Origin
Retinoblastoma is thought to be derived histogenetically from primitive retinal cells that are capable of differentiation into both neuronal and glial elements. Immunocytochemical studies of retinoblastomas have demonstrated cells that are immunoreactive for either photoreceptors or the giant glial cells of Mueller. Although the actual cell of origin is uncertain, some believe that retinoblastoma could be derived from photoreceptors.
Molecular Genetics and the Retinoblastoma Oncogene
Retinoblastoma is a heritable cancer. Between 5% and 10% of cases are inherited as what appears to be a classic mendelian autosomal dominant trait with incomplete penetrance and expressivity. In other words, a carrier of familial retinoblastoma transmits the tumor to one half of his or her offspring. Molecular genetic studies have shown that the gene responsible for retinoblastoma actually is recessive on the molecular level. In fact, the retinoblastoma gene is the paradigmatic human recessive oncogene or tumor suppressor gene.
This retinoblastoma or RB1 gene is located in the 14 band of the Q or long arm of chromosome 13 (13q14). The RB1 gene is 180,388 base pairs in length, and its protein product pRB comprises 928 amino acids. pRB is abundant in the nucleus, where it is involved in control of the cell cycle. During the G1 or resting phase of the cell cycle, pRB is bound to transcription factors such as E2F. Phosphorylation of pRB causes release of E2F. Uncomplexed E2F, in turn, activates a variety of other genes and transcription factors that are important in the initiation of DNA synthesis (S phase). Absence of pRB causes continual cell division and lack of terminal differentiation. The RB protein is phosphorylated by cyclin D2 and its cyclin-dependent kinase (cdk) partner. Certain oncoviruses cause tumors by synthesizing proteins (e.g., adenoviral protein E1A and SV40 large T protein) that bind to pRB and inactivate it.
Classically, the Rb1 gene is thought to cause cancer when its protein product is absent or dysfunctional. Healthy persons have two normal or wild-type Rb1 genes. Both alleles of the Rb1 gene are absent or inactivated in the cells of a retinoblastoma. Carriers of familial retinoblastoma are heterozygous for the Rb1 gene. Although the pRB produced by a heterozygote’s single functional gene is sufficient to inhibit tumorigenesis, heterozygotes are at substantial risk to develop retinoblastoma.
The genotype of a child with familial retinoblastoma includes one functional copy of the Rb1 gene. The other copy is absent or is mutated and codes for dysfunctional RB protein. Retinoblastoma develops when the solitary remaining wild-type gene is lost or inactivated, by chance, in a cell within the developing retina. Cytogenetic mechanisms responsible for gene inactivation (and resultant homozygosity for the recessive allele) include chromosomal loss or deletion, somatic recombination, and point mutation. The spontaneous mutation rate of the normal wild-type gene RB is 1 in 10 million or greater. It is estimated that 100 million mitoses occur during the growth and development of each retina. Hence, it is highly probable that the second functional copy of the Rb1 gene will be lost or inactivated in at least one retinal cell in an individual whose genotype is heterozygous. Further, it is equally probable that gene inactivation and tumorigenesis will occur in both eyes.
An analogous situation occurs in patients who have 13Q- or chromosomal deletion retinoblastoma, which is associated with a karyotypically apparent deletion that includes the Q14 band of chromosome 13. In an analogous fashion, retinoblastoma develops after the chance loss or inactivation of the solitary suppressor gene in a retinal cell. Patients with 13Q- syndrome have severe mental retardation and other congenital abnormalities that may include microcephaly, hypertelorism, ptosis, micrognathia, deformed low-set ears, a wide nasal bridge, cardiac anomalies, anal atresia, microphthalmia, colobomas, and cataracts.
Immune surveillance appears to arrest some tumors despite the appropriate additional mutations. This probably is responsible, in part, for the incomplete penetrance of the Rb1 gene, which is estimated to be approximately 80% (in other words, there is an 80% chance that one tumor will develop in one eye). A few families with low-penetrance retinoblastoma have been reported. They have reduced levels of wild-type RB protein or mutant RB protein that retains partial activity.
Most retinoblastomas are sporadic tumors that occur in patients who have a negative family history and a normal karyotype. Three fourths of the sporadic tumors are caused by somatic mutations. Always unilateral and solitary, such somatic sporadic retinoblastomas presumably are caused by the chance inactivation of both Rb1 genes in a single cell in the developing retina (somatic mutation). The remaining 25% of sporadic retinoblastomas are caused by germinal mutations. Such sporadic germinal tumors represent new heritable cases. In most instances, the primary mutation or gene inactivation is thought to occur early in embryogenesis in the fusing gametes or in the fertilized ovum and thus is present in all the patient’s cells, including gametes. Hence, persons who have this variety of sporadic retinoblastoma can transmit the tumor to their offspring. Like familial tumors, retinoblastomas caused by sporadic germinal mutations typically are bilateral and multifocal and occur at an earlier age.
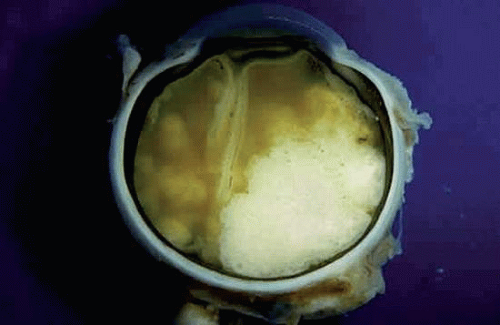
About one third of patients with retinoblastoma have bilateral tumors (Fig. 2). The sporadic occurrence of bilateral tumors indicates that the affected patient has a germline mutation and is capable of transmitting the disease to one half of his or her offspring. Unfortunately, the opposite is not true. As a result of incomplete penetrance and expressivity, 10% to 15% of unilateral, sporadic retinoblastomas are caused by potentially transmissible germline mutations. Statistics used for genetic counseling (Table 1) reflect both the effect of gene penetrance and the proportion of familial, chromosomal deletion and sporadic somatic and germinal retinoblastomas in the population.
Table 1 Genetic Counseling: Risk That Subsequent Child Will Have Retinoblastoma | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
Molecular genetics readily explains several hitherto puzzling clinical features of retinoblastoma. For example, retinoblastoma is predominantly a tumor of early childhood. Although rare adult cases have been reported, the mean age at diagnosis is 18 months and the tumor is extremely rare after 4 years of age. Cytogenetic misadventures that cause gene inactivation generally occur during cellular division. Most mitotic activity in the retina actually ceases before birth, making neoplastic transformation in older persons highly unlikely. Congenital retinoblastoma has been observed in both premature and term infants.
Retinoblastomas caused by germinal mutations typically develop at an earlier age than retinoblastomas caused by sporadic somatic mutations (mean age 12 months vs. 23 months), presumably because only a single Rb1 gene allele, rather than two, must be inactivated. Knudson graphically compared the ages of patients who had unilateral and bilateral tumors with the logarithm of the proportion in each group as yet undiagnosed. His results led him to postulate that two separate events or “hits” are necessary for the development of sporadic retinoblastomas (“two-hit hypothesis”). The curve for bilateral, hereditary tumors is a simple exponential relation, consistent with a single gene inactivation or “hit” superimposed on the inherited genotypic defect.
Patients who are heterozygous carriers of familial retinoblastoma are predisposed to develop other malignant tumors. A survivor of bilateral retinoblastoma has a 20% to 50% chance of developing a second tumor within 20 years (Armed Forces Institute of Pathology series: 26% within 30 years). These secondary nonocular tumors include osteogenic and other soft tissue sarcomas, carcinomas of the upper respiratory passages, malignant melanomas, and carcinomas of the skin. Most second tumors occur within the field of irradiation many years after external-beam radiotherapy for intraocular retinoblastoma. However, a 500-fold increase in the incidence of osteogenic sarcoma in the nonirradiated femur has been reported.
Some of the most interesting secondary nonocular neoplasms that develop in heterozygous carriers of the Rb1 gene are tumors of the pineal gland or parasellar region that resemble ectopic retinoblastomas. This association between pinealoma (also termed pineoblastoma) and bilateral hereditary retinoblastoma has been termed trilateral retinoblastoma. The pineal gland, which serves as a “third eye” in some primitive reptiles, shares antigenic determinants with the retina and exhibits transient photoreceptor differentiation in the neonatal rat. Photoreceptor differentiation has been identified in human pineal tumors.
The Rb1 gene has been implicated as a contributing factor in a variety of other systemic malignancies, including breast, lung, and bladder cancer. This is not surprising, considering the Rb1 gene’s fundamental role in control of the cell cycle.
Recent molecular genetic studies suggest that the initial concepts outlined previously concerning the Rb1 tumor suppressor gene and its role in the pathogenesis of retinoblastoma are an oversimplification. Dimaras recently has shown that both copies of the Rb1 gene are inactivated and Rb protein is absent in retinoma/retinocytoma, which is now considered to be a benign precursor lesion of retinoblastoma. Additional mutations are necessary for malignant transformation of retinoma into retinoblastoma. The retinoblastoma gene plays an important role in the regulation of cellular division in all cells in the body, yet only retinoblastoma, a relatively rare neoplasm that affects a small population of highly differentiated cells, results from the inactivation of both copies of the RB1 gene. The highly retinal-specific predisposition imposed by mutations in the RB1 gene is hypothesized to result from the specific pattern of expression of other genes in the unidentified cell of origin in the developing human retina rather than RB1.
Classification and Treatment of Retinblastoma
There are several staging systems for retinoblastoma. The Reese-Ellsworth classification for intraocular tumors has prognostic significance for retention of an eye, maintenance of sight, and the control of local disease (Table 2). Unfortunately, the Reese-Ellsworth classification recently has become less useful because it is based on the response to external-beam radiotherapy, a treatment modality that has fallen out of favor and is now used infrequently. The St. Jude Children’s Research Hospital Clinical Staging System (Table 3) attempts to relate the extent of the disease within and outside the eye to prognosis for sight as well as for freedom from systemic disease. This system is based on histologic examination and therefore requires enucleation.
Table 2. Reese-Ellsworth Classification | |
|---|---|
|
Table 3. St. Jude Children’s Research Hospital Clinical Staging System | |
|---|---|
|
A new International Classification of Retinoblastoma (ICRB) was drafted at the International Society of Retinoblastoma and Genetic Eye Diseases meeting in Paris, France, in 2003. The international classification is a relatively simple, practical classification based on clinical findings (Table 4). It was designed primarily to evaluate the potential response to modern therapy, including chemoreduction. The international classification includes five groups: A through E. Group A comprises small tumors <3 mm in diameter that are located outside the macula. Group B tumors are >3 mm in diameter but lack seeding. Other small tumors are included in group B if they involve the macula or have associated clear subretinal fluid. A tumor with localized subretinal or vitreous seeds is placed in group C. If the seeding is diffuse, the tumor is placed in group D. Group E eyes contain large tumors that occupy >50% of the globe. Eyes are also placed in group E if they have opaque media, neovascular glaucoma, or high-risk features such as postlaminar optic nerve invasion; significant choroidal invasion; and invasion of the anterior chamber, sclera, or orbit.
Table 4. International (ABC) Classification of Retinoblastoma | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
A detailed discussion of the treatment of retinoblastoma is beyond the scope of this chapter. A number of reports dealing with modern treatment modalities are referenced below. In short, there have been major changes in the treatment of retinoblastoma in developed countries during the last decade. A concerted effort has been made to avoid the use of external-beam radiotherapy, which leads to disfiguring facial deformities and predisposes retinoblastoma gene carriers to secondary malignant neoplasms in the field of radiation such as soft tissue sarcomas. Instead, many infants with retinoblastoma, particularly those with germline mutations and bilateral tumors, are being treated with chemotherapy. In many instances, chemotherapy is used to shrink tumors so that they are amenable to additional treatment or “consolidation” with local therapeutic agents such as cryotherapy, infrared laser transpupillary thermotherapy, or radioactive plaque brachyradiotherapy. Called chemoreduction, this form of chemotherapy is used most commonly in the treatment of bilateral retinoblastoma. About 75% of eyes with unilateral retinoblastoma are still enucleated. Initial studies from the Oncology Service at Wills Eye show that chemoreduction is successful in treating most patients with international classification class A, B, and C retinoblastoma, with success defined as avoidance of enucleation and/or external-beam radiotherapy. The success rate was 100% in class A, 93% in class B, and 90% in class C with currently 13 years experience. The effectiveness drops to about 50% in class D tumors with diffuse seeding. Although chemoreduction should prevent the development of secondary malignant neoplasms in the field of radiation by avoiding external-beam radiotherapy, it is possible that secondary tumors still might develop in some genetically predisposed patients as a response to chemotherapy. Particularly, there is concern that the chemotherapeutic agent etoposide might cause secondary acute myelogenous leukemia. Systemic chemotherapy does appear to prevent distant metastases, and preliminary results also indicate that it may lead to a lower incidence of “trilateral retinoblastoma,” which is almost invariably fatal.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree