Retina and Vitreous
Elise Héon
Nasrin Najm-Tehrani
Brenda Gallie
Wai-Ching Lam
Peter Kertes
Robert Devenyi
Carol Westall
Thomas W. Wilson
Alex V
The retina develops from the neuroectodermal cells of the optic cup around 4 weeks of gestation. Retinal development occurs in parallel with the development of other organ systems and therefore, explains why retinal diseases of childhood are often associated with disorders of the ear, kidney, or central nervous system. Primary care providers should be informed of the possibility of an underlying syndrome in all children with retinal disease. The retina matures from the center to the periphery.
The photoreceptors, essential to the processing of light, start to develop between 4 and 12 weeks of gestational age. The macula becomes recognizable between 24 and 26 weeks and the fovea will continue to mature after birth until 4 years of age. The retina is a fragile multilayered tissue where light is processed. The major step in the processing of light is called phototransduction, which takes place in the photoreceptors. The integrity of a photoreceptor requires a healthy surrounding, including the retinal pigmented epithelium (RPE) and inner sensory retina. Anomalies at any of these levels may lead to photoreceptor dysfunction and visual loss. This is usually the basis of the complex field of retinal dystrophies. Likewise, abnormalities of the choroid, the vasculature of which nourishes the outer retina, may also lead to retinal degener- ation. For most retinal and choroidal dystrophies, there is no treatment available, but this will change with the great influx of genetic knowledge about these conditions and the rapid progress of retinal gene and pharmacologic therapy.
Retinopathy of prematurity (ROP) is a potentially blinding disease affecting the retinal vessels in premature babies. It is characterized by arrest of the normal process of retinal vessel growth with resultant arteriovenous shunts and development of abnormal new vessels that may eventually lead to tractional retinal detachment and blindness. The disease is detected through a screening program for premature babies who are at risk and involves regular examination of the retina. The guidelines for inclusion in screening programs vary slightly between countries depending on local variations in incidence of ROP and neonatal care, but the infants at highest risk are those of low gestational age (<32 weeks) and low birth weight (<1,500 g). In these infants screening for ROP should start at 4 to 6 weeks after birth and continue at regular intervals until retinal vascularization has reached the temporal periphery (zone 3). The use of wide angle digital imaging has greatly improved our ability to observe and record the course of retinal maturation and document progression of disease, which allows for comparison between patient examinations and better communication between ophthalmologists and other health care providers. Fortunately, in the majority of cases ROP regresses spontaneously; however, in a small percentage the disease will progress with development of severe stages of ROP, with major visual consequences.
Retinoblastoma is the most common malignant tumor of childhood, but it is quite rare at an incidence of 1 in 15,000 live births. Retinoblastoma can be hereditary or nonhereditary. If left untreated, it can be fatal. For retinoblastoma and ROP, one must remember that the earliest diagnosis will produce the most favorable outcome.
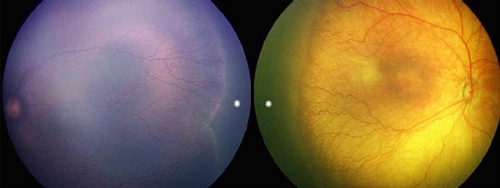
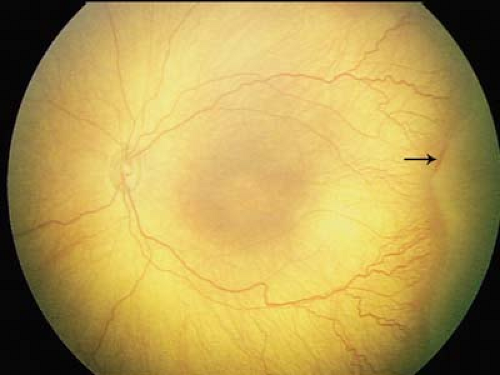
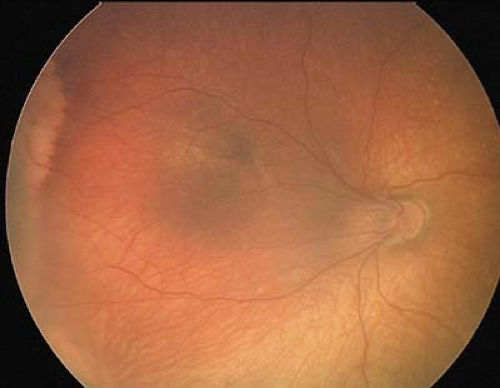
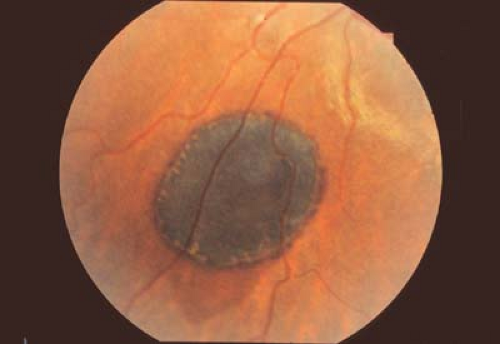 Figure 8.2 Congenital Hypertrophy of the Retinal Pigmented Epithelium (CHRPE) Congenital hypertrophy of the pigment epithelium may be isolated or associated with systemic disorders such as neurofibromatosis (see Chapter 23: Phakomatoses) or familial adenomatous polyposis (FAP). When FAP is associated with benign soft tissue and bony tumors, the condition is called Gardner syndrome. When brain tumors are present, the patient is said to have Turcot syndrome. FAP is associated with a very high risk for colonic cancer. Patients with more than two CHRPE lesions in one eye, bilateral CHRPE, or a family history of colon cancer should be screened with periodic colonoscopy starting in childhood. CHRPE lesions are usually asymptomatic unless the macula is involved. Lesions can be of various size and shape. They may have a hypopigmented ring (shown here) or a “tail.” |
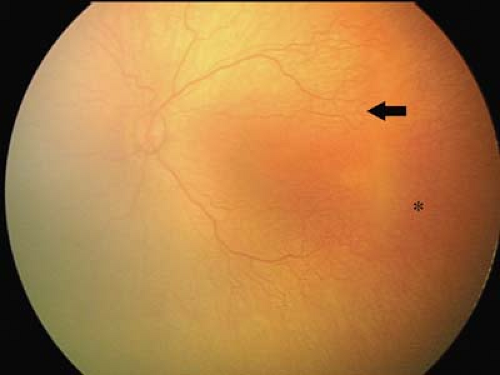
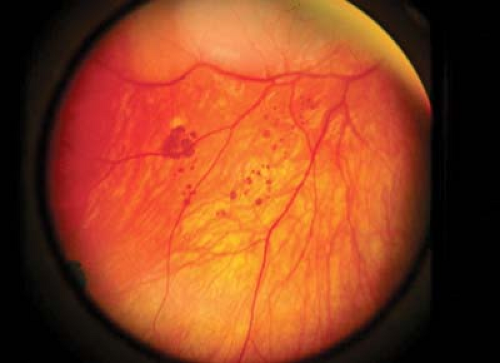 Figure 8.3 Bear Tracks These congenital hyperpigmented areas of the retinal pigmented epithelium are clustered in a pattern that has similarity to the footprints of a bear. They tend to cluster in one quadrant, usually unilaterally, and increase in number and area covered from posterior to anterior. Unlike congenital hypertrophy of the pigment epithelium (Fig. 8.2), they are not associated with systemic disease and usually do not have hypopigmented edges or tails. There may be one or more clusters, each typically with a larger lesion surrounded by one or more smaller lesions. |
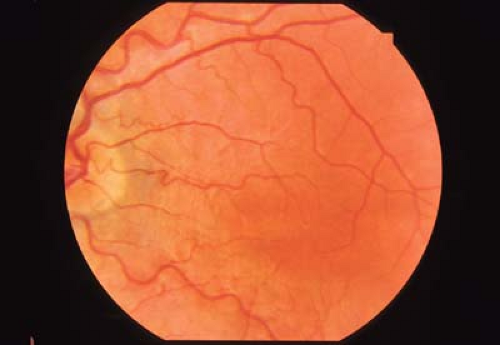
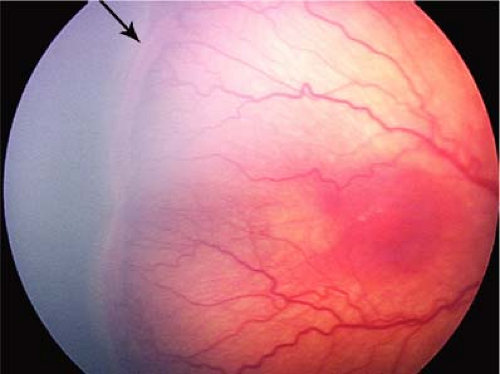
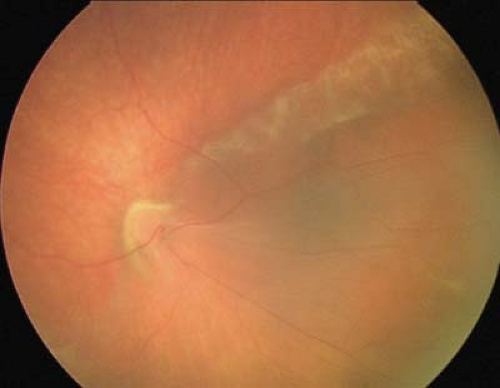
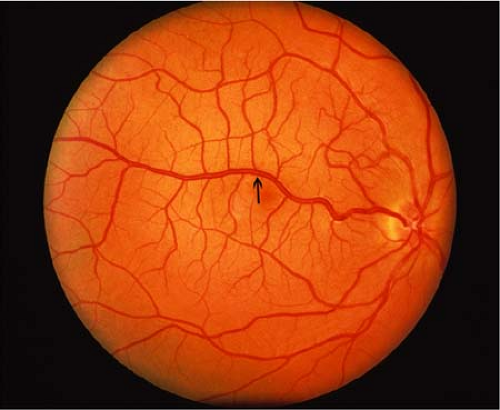 Figure 8.4 Congenital Retinal Macrovessel This rare benign vascular anomaly (arrow) is not associated with leakage or systemic disease (as might be seen in the capillary hemangioma of von Hippel-Lindau disease, Chapter 23: Phakomatoses, Fig. 23.12). Even though these vessels may appear in areas of retina not usually vascularized in this fashion, as shown here, unlike the anomalous vessels of macular hypoplasia (Fig. 8.1), congenital macrovessels are typically associated with normal retinal development and function. |
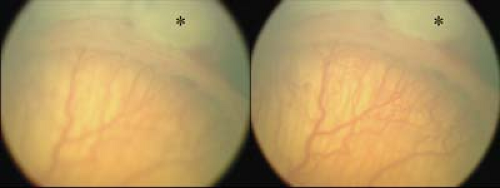
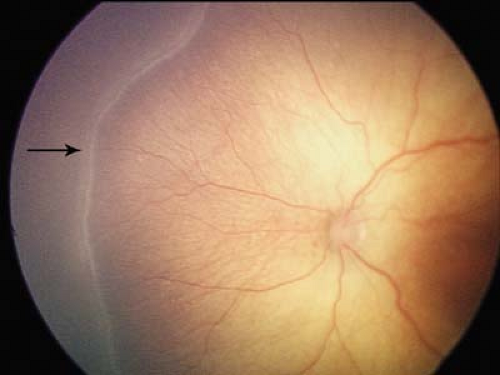 Figure 8.7 Stage 2 Retinopathy of Prematurity (ROP) With progression of ROP the demarcation line will become elevated, gain volume, and form a ridge (arrow). The distinction between stage 1 and stage 2 is much better appreciated in a three-dimensional view of the retina, using scleral depression to look at the ridge in profile (i.e., tangentially). It may be more difficult to distinguish between stage 1 (Fig. 8.6) and 2 on two-dimensional imaging. Note the associated vascular changes including peripheral vessel dilation, tortuosity, and hyperacute branching. Also note the few peripapillary intraretinal hemorrhages as sometimes seen in active disease following examination. |
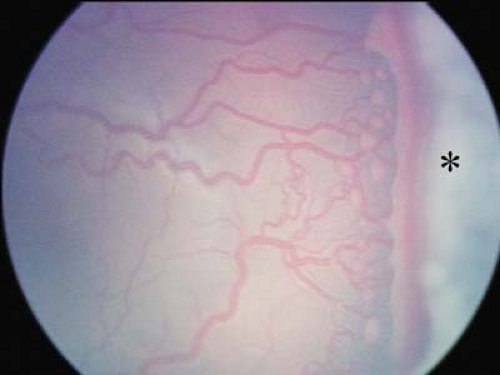
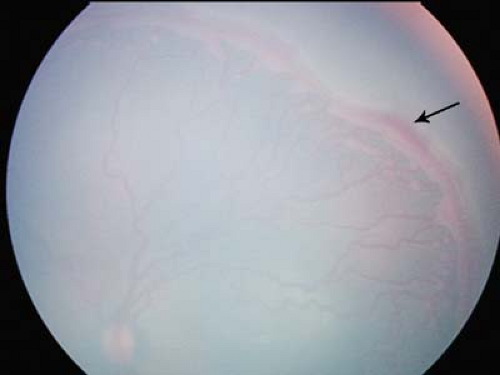 Figure 8.10 Stage 3 Retinopathy of Prematurity (ROP)—Severe This image shows severe stage 3 ROP as characterized by a broad neovascularized extraretinal ridge with a ragged posterior border with clearly visible popcorns (Fig. 8.12). There is hemorrhage along the anterior border of the ridge (arrow). The stage 3 ROP extended for 360 degrees (12 clock hours) in this baby. The retinal vessels in the posterior pole around the optic disc are clearly dilated and tortuous. |
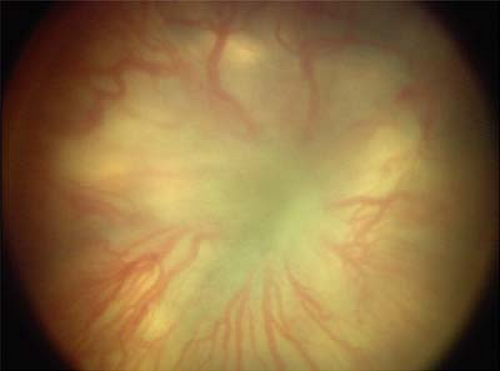
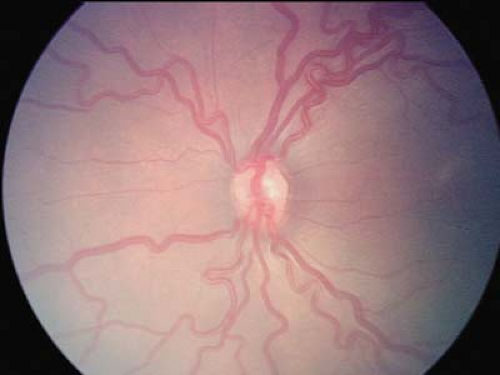 Figure 8.16 Retinopathy of Prematurity (ROP)—Plus Disease
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|