Chapter 68 Reoperation for Sporadic Primary Hyperparathyroidism
Introduction
Primary hyperparathyroidism (primary HPT), the unregulated production of parathyroid hormone (PTH) that results in hypercalcemia, is common in the United States. The incidence is reported as 1 in 100,000 with a prevalence of 0.2% to 0.5% of the population and a female-to-male ratio of 3:1 (see Chapter 56, Primary Hyperparathyroidism: Pathophysiology, Surgical Indications, and Preoperative Workup).1,2 Depending on the series, between 74% and 92% of primary HPT cases are due to a solitary adenoma.3–8 Multiple adenomas have been reported in 3% to 12% of cases,5–10 and multigland hyperplasia has been reported in 2% to 23% of cases.3–8 Multigland hyperplasia may be sporadic or familial, occurring in patients with multiple endocrine neoplasia (MEN) syndromes2,11,12 and familial hyperparathyroidism (see Chapters 65, Surgical Management of Multiglandular Parathyroid Disease, and 67, Parathyroid Management in the MEN Syndromes). Parathyroid cancer can cause primary HPT in rare instances (1% to 2% of reported cases) (see Chapters 69, Parathyroid Carcinoma, and 70, Surgical Pathology of the Parathyroid Glands).3–8
The well-described sequelae of primary HPT such as nephrolithiasis and decreased bone density or fractures are often the presenting complaint of patients with primary HPT. However, many patients present with vague, nonspecific symptoms or are considered asymptomatic.13 Mortality from primary HPT has been reported in some studies as primarily the result of cardiovascular complications in patients with moderate to severe disease.14–16 This risk has been shown to decrease over time after operation, prompting some investigators to advocate for earlier diagnosis and surgical treatment.15,17,18
Prior to the advent of the intraoperative parathyroid hormone (IOPTH) assay, surgical therapy traditionally consisted of a four-gland, or bilateral neck, exploration (see Chapters 59, Standard Bilateral Parathyroid Exploration, and 63, Intraoperative PTH Monitoring during Parathyroid Surgery). All glands were identified; success of operation was defined as extirpation of abnormal parathyroid tissue and normocalcemia for greater than 6 months. Failures were considered either persistent hypercalcemia, with no resolution of hypercalcemia after operation, or recurrent hypercalcemia, with return of hypercalcemia 6 months or longer after operation. Success rates for a first-time operation for sporadic primary HPT with four-gland exploration have been reported to be as high as 98% to 99% with low complication rates of 3% or less.5,7
The IOPTH assay was first described in 1991 as a means to further decrease the operative failure rate,19 and it is now used commonly in parathyroid operations. The combination of the IOPTH assay and improved localization techniques has led many surgeons to prefer a directed operative strategy for well-localized, presumed solitary adenoma. In this approach, a unilateral neck exploration is used, with bilateral exploration reserved for cases in which abnormal parathyroid tissue is not found or an appropriate drop in IOPTH is not observed. Focused exploration success rates have been comparable to four-gland exploration (see Chapters 60, Minimally Invasive Single Gland Parathyroid Exploration, 61, Minimally Invasive Video-Assisted Parathyroidectomy, 62, Local Anesthesia for Thyroid and Parathyroid Surgery, and 63, Intraoperative PTH Monitoring during Parathyroid Surgery).20–23
Reoperations for persistent or recurrent hyperparathyroidism have been reported to cost more than twice as much as first-time operations24 with lower success rates and higher complication rates. Series that included patients with multigland disease describe success rates between 86% to 96%.25–30 Other series considering only those patients with presumed missed solitary adenoma have described success rates of 95% or better.31–33 This chapter provides a framework for a standard approach to patients with persistent or recurrent primary hyperparathyroidism resulting from a presumed single adenoma and reviews current localization and operative management.
Indications for Reoperation
The most important considerations in the management of the patient with persistent or recurrent primary HPT is first to determine if the patient truly has primary HPT and then to determine if the symptoms or manifestations are severe enough to warrant the potential increased morbidity of reoperative surgery. The biochemical evaluation of those patients presenting with persistent or recurrent primary HPT is the same as for initial primary HPT and includes assays for serum and urine calcium, PTH, and vitamin D. Hypercalcemia is defined as a serum calcium level above the normal range for the particular assay used. PTH assays have been through several iterations and are now in their third generation. Interpretation of PTH levels must include the individual laboratory reference values and consideration of the 25-OHD level to exclude secondary hyperparathyroidism caused by vitamin D deficiency.34 Urine should be measured for 24-hour calcium and creatinine levels in order to exclude familial hypocalciuric hypercalcemia (FHH). FHH is a familial disease that presents with asymptomatic hypercalcemia resulting from a mutation in a calcium receptor gene. These patients do not have the sequelae of primary HPT, as they have a normal PTH response but need a higher serum calcium level to suppress PTH secretion.35
The classic presenting signs and symptoms of primary HPT include kidney stones, manifestations of bone disease, gastrointestinal complaints, fatigue, and neuropsychiatric complaints.1,2 The higher risk of complications for reoperation for primary HPT has led many surgeons to advocate for reoperation primarily on those patients with objective signs of primary HPT, usually kidney stones or bone disease.1,25 In a number of reported series of reoperation, nephrolithiasis accounted for 45% to 55% of overall symptoms, whereas bone pain, fractures, or osteoporosis accounted for 21% to 53%.25,30,32,33 In cases of suspected but unproved nephrolithiasis, ultrasound is used to document stones, followed by computed tomography (CT) scan.34 In terms of bone disease, surgery is indicated for peri- or postmenopausal women and men 50 years of age and older with T scores of ≤− 2.5 and premenopausal women and men younger than 50 years of age with Z scores of ≤2.5.36
Abdominal pain from peptic ulcer disease (PUD) is frequent in MEN 1-related primary HPT resulting from concomitant gastrinoma37; primary HPT has been shown to be more virulent in these patients and parathyroidectomy is recommended to ameliorate both PUD and primary HPT symptoms.38 Abdominal pain in sporadic primary HPT is less objectively defined but has been reported to be due to PUD, constipation, and other complaints in 6% to 19% of cases in reoperative series.25,30,32,33 Fatigue, proximal muscle weakness, and lethargy are also reported commonly.25,30,32,33 Although the data are not yet strong enough to establish definitive indications for parathyroidectomy, associations between primary HPT and cardiovascular and neurocognitive dysfunction have been described.13
Our policy has been to offer reoperation to patients with biochemical evidence of recurrent or persistent primary HPT and documentation of nephrolithiasis or bone disease according to the criteria presented earlier. For asymptomatic patients or those with vague complaints, reoperation is offered only when the lesion is well localized on preoperative noninvasive imaging studies (Box 68-1, Issue 1).
Operative Planning
After establishing the diagnosis of persistent or recurrent hyperparathyroidism, the next step in considering a patient for reoperative parathyroid surgery is to review both the operative note and pathology from the prior operation(s). It is important to understand the location and extent of dissection at the first operation and the number of parathyroid glands identified or biopsied. Pathologic evaluation of the specimens is crucial. Although the operative note may describe the visualized parathyroid glands, histologic evaluation is important to differentiate among nonparathyroid tissue, normal parathyroid, and hypercellular tissue. In many cases, tissue that visually appeared to be a parathyroid gland is not found to contain any parathyroid tissue. It is particularly important to consider whether the patient has a missed or recurrent adenoma versus multigland hyperplasia, as the operative approach for these entities is different. Understanding the number of parathyroids identified and the locations in which the first surgeon looked (both orthotopic and ectopic) helps the reoperative surgeon interpret localization studies and plan the next operative approach (see Chapters 2, Applied Embryology of the Thyroid and Parathyroid Glands, and 58, Principles in Surgical Management of Primary Hyperparathyroidism).
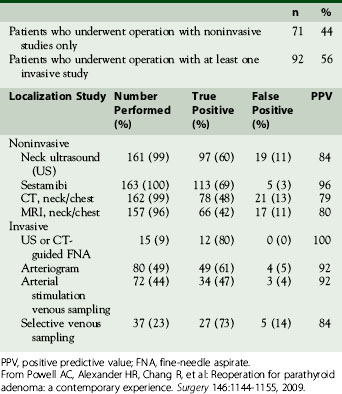
Once operative notes and pathology reports have been reviewed, localization studies should be performed and are the foundation of reoperative parathyroid surgery (see Chapter 57, Guide to Preoperative Parathyroid Localization Testing). A reoperation for primary HPT should be guided by positive localization studies. Going back in the scarred neck “blind” is a recipe for disaster and likely to result in a second failed operation. Localization studies are divided into the categories of noninvasive (ultrasound, nuclear medicine scanning, and cross-sectional imaging) and invasive approaches (arteriography and vascular sampling). Noninvasive studies are the first approach. Sestamibi and ultrasound are usually less expensive compared to the cross-sectional imaging of CT and magnetic resonance imaging (MRI); the cost of arteriography and vascular sampling are more variable but generally more expensive than other procedures.39 Noninvasive studies are now often able to provide the necessary information for a successful extirpation without the need for further studies. In our recent comparison of our reoperative experience between 1996-2008 and 1982-1995, fewer patients underwent invasive studies (56% versus 73%, respectively). We attributed this difference to improvements in ultrasound and especially nuclear medicine scanning and to the combination of the two.32,33 The results of all imaging studies in our most recent series are presented in Table 68-1.
At the National Institutes of Health (NIH), neck ultrasonography is performed with either a 10- or a 7-MHz near-field transducer. The neck is scanned from the submandibular glands down to below the medial aspects of the clavicles; the thyroid lobe(s), carotid arteries, and jugular veins are identified. Parathyroid adenomas present as small, hypoechoic, ovoid masses (see Chapter 13, Ultrasound of the Thyroid and Parathyroid Glands).40 Ultrasound is best for lesions located centrally in the neck but has lower sensitivity in the upper neck and in the anterior mediastinum below the clavicles.41 Ultrasound is operator dependent; it is important that ultrasonographers experienced with parathyroid imaging perform and interpret the images. Finally, ultrasound, like CT and MRI, has a high false-positive rate; often these techniques will identify more than one candidate lesion, often a lymph node.
Nuclear medicine scanning uses the technetium-99 m (Tc-99m) sestamibi (MIBI) isotope to image parathyroid tissue; a second isotope, commonly Tc-99m pertechnate, is often used to identify and subtract thyroid tissue from the final images (see Chapter 57, Guide to Preoperative Parathyroid Localization Testing). MIBI scanning was first introduced in the early 1990s for primary hyperparathyroidism after success with myocardial perfusion imaging.42,43 MIBI scanning without subtraction images was extended to reoperative cases with limited results initially.44 MIBI sensitivity in reoperation improved with the addition of subtraction images using a second isotope.45
The major limitations of MIBI imaging are the two-dimensional nature of the images and the imprecise localization. Other modifications and improvements have been made to MIBI scanning in efforts to overcome these limitations, including oblique images and single-photon emission computed tomography (SPECT). SPECT allows for three-dimensional imaging and better characterization of the size of the adenoma. In one study of MIBI with SPECT, the sensitivity for localizing a parathyroid lesion was similar in both first-time and reoperative cases (87% versus 92%, respectively).46 Our standard protocol for nuclear medicine scanning consists of the injection of Tc-99m pertechnate and Tc-99m sestamibi with early/delayed imaging, subtraction, oblique views, and SPECT.41 In several series from the NIH, the positive predictive value of MIBI has been shown to be 94% to 96%,32,33,39 which is the highest of all noninvasive modalities. The combination of ultrasound and MIBI has further increased the likelihood of success at operation without the need for invasive studies. When ultrasound and MIBI were analyzed together in our recent series, success at operation was 100% when the ultrasound and MIBI were both positive, even if they were discordant.33
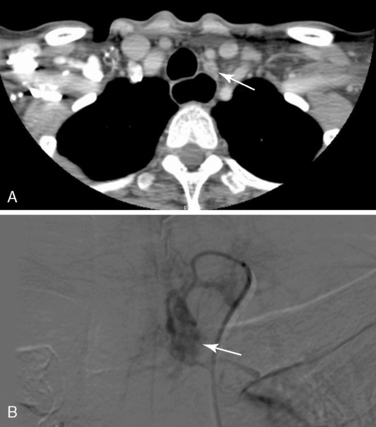
The utility of CT and MRI has been studied in many series of reoperative parathyroid patients. We have shown that the positive predictive value (PPV) is approximately 80% for both modalities.32,33 Cross-sectional imaging adds three-dimensional data and can help better localize a lesion in relationship to other structures. CT in particular can be helpful in distinguishing parathyroid lesions from the soft tissues of the neck, especially those in the tracheoesophageal groove and mediastinum41 (Figure 68-1). We predominantly use CT or MRI to evaluate equivocal ultrasound or MIBI results before proceeding to invasive localization and to more specifically localize MIBI findings in the chest.
Recently, four-dimensional CT (4D-CT) has begun to be used in both the first-time and reoperative settings for primary HPT. Four-dimensional CT provides the usual three-dimensional cross-sectional imaging and adds perfusion data over time to provide the fourth dimension. The additional perfusion data are able to differentiate between normal and hyperfunctioning parathyroid tissue by demonstrating the rapid uptake and washout characteristics of hyperfunctioning tissue. Thus, 4D-CT is able to provide both anatomic and functional data.47 In a study of 75 patients with primary HPT at MD Anderson Cancer Center, 4D-CT was found to have significantly better sensitivity than MIBI and ultrasound in localizing an adenoma. This study only included 12 patients with previous neck surgery, and only one of these patients had a previous parathyroidectomy.47 The same group then studied 45 patients with primary HPT who had undergone previous neck exploration; 23 of these patients had previous operations for primary HPT, whereas the other patients had other reasons for neck exploration (predominantly thyroid operations). In both categories of patients, 4D-CT was found to be equivalent or superior to MIBI and superior to ultrasound. In the group of patients with previous operations for primary HPT, the sensitivity of 4D-CT as compared to MIBI was 93% versus 55%.48 Additional studies need to be performed to provide more data on 4D-CT, but many centers are now starting to use 4D-CT as part of their reoperative localization.
Overall, localization by noninvasive imaging is considered sufficient when two or more studies provide concordant information without major discordant data. If the studies are negative or provide discordant information, we proceed to invasive localization. For indeterminate but suspicious lesions in the neck, CT or ultrasound-guided fine-needle aspirate (FNA) is very specific and, if positive, renders further invasive localization procedures unnecessary. The specific case of a positive ultrasound finding in the neck with a negative MIBI scan is an excellent situation to use FNA to confirm the ultrasound. Cytologic analysis and PTH assays have both been used to analyze the aspirate material. Although some groups have reported success with cytology, this may be due to a histologic analysis of fragments of tissue found in samples using large needles (16 gauge) instead of a smear.49 Cytology can be limited by inadequate sample material, the possible inclusion of thyroid tissue in aspirates for lesions that are intra- or juxtathyroidal,50 and the inability to distinguish between adenoma and hypercellular parathyroid tissue.49 For many of these reasons, other groups have not found cytology useful in localization.51
PTH assay of the aspirate appears to be more robust and reproducible. Our protocol is to use a 19- to 23-gauge needle attached to a syringe and make two to four passes into the lesion. The aspirate is then diluted to a volume of 1 cc and analyzed for PTH; a result is considered positive if the aspirate PTH is twice that or more of serum PTH.50 We have found the positive predictive value of FNA using PTH aspirate to be 100%.32,33 Rapid PTH assay can be used with the aspirates with good results.52 Small hematomas and transient bronchospasm have been seen as complications of the procedure but are self-limiting and easily treated.50 Despite the high utility of FNA and the low complication risk, some patients do not have lesions amenable to FNA, or the aspirate is negative, and further localization must be undertaken.
Of the invasive vascular procedures, digital subtraction angiography (DSA) is our preferred next study. Some centers use conventional angiography (CA) instead of DSA, but our data have shown DSA to be equivalent or superior to CA in all locations and superior in the mediastinum.53 Selective catheterization of bilateral internal thoracic, inferior thyroid, and common carotid arteries is performed according to our published protocol54; the superior thyroid arteries are studied only if the others are negative. A blush identifies an adenoma53,54 (see Figure 68-1). Immediately following DSA, arterial stimulation venous sampling is performed, in which samples are drawn from the superior vena cava after selective hypocalcemic (sodium citrate) arterial stimulation injection and assayed for PTH. Sampling is considered positive if the PTH level after stimulation is 1.4 times or more above the baseline PTH value prior to stimulation.33 Although complications such as stroke and embolism are possible, no complications have been reported in our series.54 At the NIH, DSA and arterial stimulation venous sampling were comparable with a 92% positive predictive value for both, although the localization provided by the arterial stimulation venous sampling was not as precise.33
If localization is still negative or equivocal after DSA and arterial stimulation venous sampling, selective venous sampling (SVS) is performed. All patent inferior, middle, and superior thyroid veins, thymic veins, and the vertebral veins are sampled via a femoral vein and assayed for PTH. Sampling is considered positive if there is a step-up of at least two times more than a baseline sample obtained from the iliac vein.55 The data provided from DSA are important to review prior to SVS, as they provide anatomic detail. These data are especially important in the reoperative setting with potentially anomalous venous drainage from past surgery. Although the 84% positive predictive value of SVS has been shown to be lower than that of DSA and arterial stimulation venous sampling,32,33 it is an important tool in patients for whom all other localization studies have been negative.55,56 In our most recent study, 14 patients were explored based primarily on SVS data with the adenoma found in 11 patients (79%).33
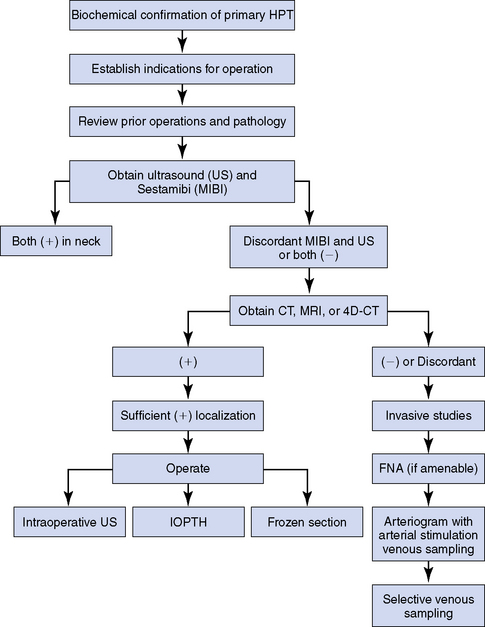
Our approach to the evaluation of the patient with persistent or recurrent primary HPT is summarized in Figure 68-2. Once sufficient localization has been obtained as described, the patient is explored (Box 68-1, Issue 2).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree