Purpose
To evaluate the efficacy of methotrexate (MTX) and the effect of its withdrawal on relapse rate of uveitis associated with juvenile idiopathic arthritis (JIA).
Design
Retrospective case series.
Methods
Data of 22 pediatric JIA patients who were being treated with MTX for active uveitis were studied retrospectively. Relapse rate after the withdrawal of MTX was established. Anterior chamber (AC) inflammation, topical steroid use during the first year of MTX treatment, and associations of relapses after the withdrawal were evaluated statistically. Duration of MTX treatment and its withdrawal was determined individually in collaboration with a rheumatologist with an intention to continue the treatment for at least 1 year and to withdraw in case of inactivity of uveitis and arthritis. Inactivity of uveitis was defined as the presence of ≤0.5+ cells in the AC.
Results
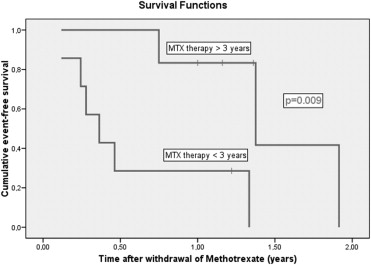
Eighteen patients (18/22; 82%) showed improvement of their uveitis with a significant decrease in activity of AC inflammation after a minimal period of 3 months of MTX treatment. A topical steroid–sparing effect was observed when MTX was administered for a period of 3 to 9 months. MTX was discontinued because of inactive uveitis in 13 patients. In 9 patients (8/13; 69%) a relapse of uveitis was observed after a mean time of 7.5 months (± SD 7.3). Six patients (6/13; 46%) had a relapse within the first year after the withdrawal. Relapse-free survival after withdrawal of MTX was significantly longer in patients who had been treated with MTX for more than 3 years ( P = .009), children who were older than 8 years at the moment of withdrawal ( P = .003), and patients who had an inactivity of uveitis of longer than 2 years before withdrawal of MTX ( P = .033). Longer inactivity under MTX therapy was independently protective for relapses after the withdrawal (hazard ratio = 0.07; 95% confidence interval 0.01-0.86; P = .038), which means that 1-year increase of duration of inactive uveitis before the withdrawal of MTX results in a decrease of hazard for new relapse of 93%.
Conclusions
A high number of patients with inactive uveitis relapse quickly after the withdrawal of MTX. Our results suggest that a longer period of inactivity prior to withdrawal and a longer treatment period with MTX reduce the chance of relapse after withdrawal.
Chronic anterior uveitis is known as a severe extra-articular manifestation of juvenile idiopathic arthritis (JIA) and it may occur in up to one third of patients with JIA. Chronic anterior uveitis can have a complicated course and it can be potentially deleterious to vision.
The aim of treatment of children with JIA-associated uveitis should be control of the inflammation and prevention of the development of sight-threatening complications with an acceptable level of side effects. Topical corticosteroids are the first step of treatment, but local therapy is frequently insufficient and chronic use of topical corticosteroid can enhance the development of cataract and glaucoma. Therefore, systemic immunosuppressive medication is warranted in many cases. Methotrexate (MTX) is an effective treatment option for arthritis in patients with JIA. Several retrospective studies have shown the effectiveness and safety of MTX in the management of uveitis in the past decennia, and MTX has been recognized as an effective agent in management of JIA-associated uveitis. Although treatment with MTX is generally used for JIA uveitis, there are still some unanswered questions about the optimal duration of MTX therapy and about the risk of relapse of uveitis after discontinuation of MTX therapy. In this study we attempt to clarify these issues.
Patients and Methods
Medical data of MTX-treated patients with JIA-associated uveitis attending a tertiary center for (pediatric) uveitis between 1989 and 2009 were studied retrospectively. JIA was diagnosed and classified by a pediatric rheumatologist in agreement with the International League of Associations for Rheumatology criteria. Diagnosis of uveitis was made by an ophthalmologist specialized in pediatric uveitis. The diagnostic criteria for uveitis were those defined by the International Uveitis Study Group.
The JIA-associated uveitis had to manifest before the age of 16 years, and we included all the patients who were treated with MTX for active JIA-associated uveitis (eventually in combination with active arthritis) before the age of 20. The inclusion cut point of 20 years was chosen to prevent losing data of patients who had been treated with MTX late in adolescence. The patients in whom arthritis (and not uveitis) was an indication to start the MTX treatment were not included. All patients in whom MTX was withdrawn had a minimum follow-up of 1 year after the withdrawal; no patients were excluded because of shorter follow-up.
MTX was prescribed with collaboration of an ophthalmologist and a pediatric rheumatologist. MTX was administered orally or subcutaneously at a dosage of 10 to 15 mg/m 2 /week. Duration of MTX treatment was determined individually in collaboration with a rheumatologist with an intention to treat the patients for at least 1 year and to withdraw MTX in case of inactivity of uveitis and arthritis. Since there are no standard guidelines for MTX withdrawal available, the drug was usually withdrawn after a variable period of inactivity of uveitis without dose reduction.
The following data were recorded for each patient: age, gender, date of onset of uveitis and arthritis, laterality, subtype of JIA, antinuclear antibody (ANA) serologic status, date and indication of MTX start, date and reason for MTX withdrawal, side effects, treatment with other local or systemic anti-inflammatory therapy, and relapses after the withdrawal. Further, grade of anterior chamber cells and presence of complications were noted. The database was built on patient level with notification of the highest grade of inflammation and dosage of topical steroids in the affected eye. Complications were scored if they were present in any of the affected fellow eyes. Activity of anterior chamber (AC) inflammation (cells) on standard slit-lamp examination was evaluated according to the recommendations of the SUN working group. Additionally, topical corticosteroid requirement was quantified by the number of drops used per day. Activity of AC inflammation and corticosteroid requirement was scored at fixed time points: moment of starting MTX therapy and at 3, 6, 9, and 12 months of MTX use. Activity of uveitis was defined as the presence of ≥1+ cells in the AC. Improvement on MTX was considered as at least a 2-step decrease of the cell grade in the AC. Inactivity of uveitis was defined as the presence of ≤0.5+ cells in the AC. Relapse of uveitis after withdrawal of MTX was scored in case of presence of 1+ or more cells in the AC in patients with inactive uveitis at the moment of the withdrawal. There was no difference in follow-up schedules pre- and post-MTX withdrawal (approximately every 6 weeks).
Statistical analysis was performed using SPSS version 15.0.1 (SPSS Inc, Chicago, Illinois, USA). Fisher exact test was used for univariate analysis of categorical variables. McNemar test was used to analyze linked dichotomous variables. Wilcoxon test for paired samples was used to analyze means of abnormally distributed linked samples; for independent samples with abnormal distribution Mann-Whitney U test was applied. For correlations between normally distributed continuous variables, Pearson correlation coefficient (r) was computed. Kaplan-Meier survival analysis with a log rank test was used to analyze survival curves and to compare groups. Cox proportional hazard regression was applied for multivariate analysis of variables significant in univariate analysis and adjusted for gender as a potential confounder. P values of less than .05 were considered statistically significant. All significances are 2-tailed. In the presentation of the results we used mean if the data were normally distributed and median if they were not.
Results
Twenty-two patients with active uveitis at initiation of MTX were included in the study. General baseline characteristics of the study population are listed in Table 1 .
| Characteristics | Number of Patients, n (%) | |
|---|---|---|
| Initiation of MTX N = 22 | Withdrawal of MTX N = 13 | |
| Active uveitis | 22 (100) | NA a |
| Gender | ||
| Female n (%) | 15 (68) | 9 (69) |
| Age in years | ||
| At onset of arthritis, median (range) b | 3.3 (1.5-11.3) | 3.2 (1.7-11.3) |
| At onset of uveïtis, median (range) | 4.0 (1.9-9.5) | 4.1 (1.9-7.4) |
| At start MTX (mean ± SD) c | 8.0 ± 4.0 | 6.0 ± 3.0 |
| Duration of uveitis in years | ||
| At start MTX, median (range) | 1.8 (0.1-10.9) | 0.6 (0.1-7.0) |
| Type of JIA | ||
| Oligoarticular n (%) | 20 (91) | 12 (92) |
| Polyarticular n (%) | 1 (4) | 1 (8) |
| Psoriatic n (%) | 1 (4) | NA |
| ANA status | ||
| Positive n (%) | 20 (91) | 11 (85) |
| Laterality | ||
| Bilateral n (%) | 18 (81) | 12 (92) |
a All patients had inactive uveitis at the moment of withdrawal of MTX.
Clinical Characteristics at Initiation of MTX Therapy
Duration of uveitis before starting MTX therapy ranged from 0.1 to 10.9 years (median 1.8 years) ( Table 1 ). In 50% of patients (11/22) MTX was initiated because of active uveitis and in another 50% because of a combination of active uveitis and arthritis. In all but 1 patient MTX was initially administered orally. Activity of the AC inflammation at the start of MTX therapy ranged from 1+ to 3+ cells ( Table 2 ). Ocular complications present at start of MTX included posterior synechiae (15/22; 68%), band keratopathy (5/22; 23%), cystoid macular edema (4/22; 19%), papillitis (4/22; 19%), and glaucoma (2/22; 9%). Five patients (23%) underwent cataract surgery before the start of MTX.
| MTX At Start | MTX 3 Months | MTX 6 Months | MTX 9 Months | MTX 12 Months | |
|---|---|---|---|---|---|
| Anterior chamber activity | |||||
| Patients with active anterior uveitis, n (%) | 22 (100) | 10 (45) | 8 (36) | 8 (36) | 7 (32) |
| P value a | NA | <.001 b | <.001 b | <.001 b | <.001 b |
| Median of anterior chamber cell activity grade (range) c | 1 (1-3) | 0.5 (0-2) | 0.5 (0-3) | 0.5 (0-2) | 0.5 (0-2) |
| P value d | NA | .001 b | .037 b | .003 b | <.001 b |
| Topical steroids | |||||
| Patients on topical steroids, e n (%) | 20 (91) | 21 (95) | 19 (86) | 18 (82) | 18(82) |
| Dosage of topical steroids per day; median drops (range) | 3 (0-6) | 2 (0-4) | 2 (0-6) | 2 (0-6) | 3 (0-4) |
| P value d | NA | .011 b | .017 b | .039 b | .146 b |
a P values computed using McNemar test.
b Compared with the moment of start MTX.
c Anterior chamber activity is scored according to the recommendations of SUN working group.
d P values computed with Wilcoxon test.
e Prednisolone 1% was administered in all patients on topical steroids.
Uveitis Activity During MTX Therapy
Eighteen patients (18/22; 82%) showed improvement of their uveitis activity with at least a 2-step decrease of the AC cells within the first year ( Table 2 ). Of those; 15 patients achieved inactivity of uveitis within the first year of MTX therapy (median = 0.5 year), while 3 patients improved on MTX without reaching complete control of inflammation. A significant decrease in activity of uveitis in the whole group was seen at 3 months of MTX use ( Table 2 ).
Patients who improved on MTX treatment did not differ significantly from patients without improvement in age, gender, duration of uveitis, arthritis, ANA serologic status, MTX dosage, or other therapeutic strategies.
Systemic and Topical Medication During MTX Treatment
At the moment of initiation of MTX, 4 patients (4/22; 18%) were treated with systemic corticosteroids. In 3 of them (3/4; 75%) steroid treatment was withdrawn within 2 months of commencement of MTX and the other patient required continuation of prednisone during MTX treatment for achieving inactivity of uveitis.
At the initiation of MTX therapy, 20 patients (20/22; 91%) were treated with topical steroids (prednisolone 1%) with a median of 2 drops/day (range of 1-6 drops/day). The local corticosteroid therapy could be decreased in 13 of 22 patients (59%) with a significant topical steroid-sparing effect from 3 up to 9 months of start of MTX therapy. The statistical significance of steroid-sparing effect was lost at 12 months of MTX therapy ( Table 2 ).
None of the patients were using other immunosuppressive agents during the first year of MTX therapy. An anti–tumor necrosis factor (TNF)-α agent was added in 2 patients after 8 and 9 years of MTX therapy. Cyclosporine was added to MTX in 1 patient after 2 years of MTX therapy. Nine patients received periocular steroid injections (however, only 2 within the first year of MTX therapy).
Results After Withdrawal of MTX
General baseline characteristics of patients in whom MTX was withdrawn are presented in Table 1 . MTX was discontinued in 13 patients with inactive uveitis after 1.5 years mean duration of inactivity of uveitis (range 1 month to 3 years) and 3.1 years mean duration of the therapy. Follow-up after the withdrawal ranged from 1.0 to 11.1 years with a median of 1.7 years. Median age at withdrawal of MTX was 8.6 years (range 3.5-16.5 years).
In 9 of these patients (9/13; 69%) a new relapse of uveitis was observed after a mean time of 7.5 months (± SD 7.3), with the first relapse after 1 month of withdrawal of MTX. In all cases a new relapse took place within 2 years after the discontinuation of MTX, while 6 patients (6/13; 46%) relapsed within the first year after the withdrawal.
Relapse-free survival after the withdrawal of MTX was significantly longer in patients who had been treated with MTX longer than 3 years ( P = .009; Figure 1 ), children who were older than 8 years at the moment of the withdrawal ( P = .003; Figure 2 ), and patients who had an inactivity of longer than 2 years before the withdrawal of MTX ( P = .033; Figure 3 ).