Recurrent Respiratory Papillomatosis
Craig S. Derkay
Cristina M. Baldassari
In discussing recurrent respiratory papillomatosis (RRP) of the aerodigestive tract, it is helpful to review our current knowledge regarding the etiology of the disease, including some basic information on human papillomavirus (HPV), the histopathology of respiratory papillomas, as well as the epidemiology of the disease and risk factors for its transmission. This chapter also addresses the common clinical features seen in children with RRP, including the pertinent findings on history, physical examination, endoscopy, and imaging studies. Surgical treatments, including the use of laser and nonlaser technologies, also are addressed. Additionally, potential nonsurgical adjuvant therapies and their current indications for use are discussed. Complications of the disease itself as well as of its treatment are covered. A staging system for following patients with RRP is presented along with other new horizons for research and treatment of this frustrating entity.
RRP is a disease of viral etiology, caused by HPV types 6 and 11, associated with exophytic lesions of the airway. Although it is a benign disease, RRP has potentially morbid consequences due to its involvement of the airway, propensity to recur, and to the risk of malignant conversion.
RRP is both the most common benign neoplasm of the larynx among children and the second most frequent cause of childhood hoarseness (1). The disease is often difficult to treat because of its tendency to recur and spread throughout the respiratory tract. Although it most often involves the larynx, RRP may involve sites anywhere in the entire aerodigestive tract. The course of the disease is variable, with some patients experiencing spontaneous remission and others suffering from aggressive papillomatous growth, requiring multiple surgical procedures over many years.
In most pediatric series, RRP is diagnosed between 2 and 4 years of age with a delay in diagnosis from the time of onset of symptoms averaging about 1 year (2,3). Seventy-five percent of the children have been diagnosed before their fifth birthday (4). It is estimated that 1,500 to 2,500 new cases of childhood-onset RRP occur in the United States each year. The incidence among children in the United States is estimated at 4.3 per 100,000 children, translating into more than 15,000 surgical procedures at a total cost of more than $100 million per year (5). Anecdotal observations suggest that most patients are first born; have young, primigravid mothers; and come from families of low socioeconomic status (5,6,7). The clinical course is unpredictable, with malignant transformation possible in patients with long-standing aggressive disease.
RRP may have its clinical onset during either childhood (juvenile onset recurrent respiratory papillomatosis, JORRP) or adulthood (adult onset recurrent respiratory papillomatosis, AORRP). Of these two distinct forms, JORRP is generally more aggressive. The aggressive form of RRP, although most prevalent in children, can also occur in adults. Children whose RRP was diagnosed at younger ages (less than 3 years) have been found to be 3.6 times more likely to have more than four surgeries per year and almost 2 times more likely to have two or more anatomic sites affected than were children whose RRP was diagnosed at later ages (greater than 3 years) (8,9).
ETIOLOGY
Human Papillomavirus
HPV is a small DNA-containing, nonenveloped icosahedral (20-sided) capsid virus with a double-stranded circular deoxyribonucleic acid 7,900 base pairs long. Until the 1990s, HPV had been suspected but not confirmed as the causative agent in RRP. This uncertainty developed from an inability to culture the virus in vitro, and from the failure to demonstrate viral particles consistently in papilloma lesions using electron microscopy or HPV antibodies. Today, with the use of viral probes, HPV DNA has been identified in virtually every papilloma lesion studied. The
most common types identified in the airway are HPV 6 and HPV 11, the same types responsible for genital warts. Specific viral subtypes may be correlated with disease severity and clinical course. Children infected with HPV 11 appear to have a more obstructive airway course early in the disease and a greater need for tracheotomy (9,10).
most common types identified in the airway are HPV 6 and HPV 11, the same types responsible for genital warts. Specific viral subtypes may be correlated with disease severity and clinical course. Children infected with HPV 11 appear to have a more obstructive airway course early in the disease and a greater need for tracheotomy (9,10).
HPVs are grouped on the basis of genetic homology, with viruses that exhibit less than 90% identity in specific regions of the viral genome being defined numerically as separate types. On this basis, nearly 100 different HPV types have been identified. These groupings correlate with tissue preference as well as similarities in pathophysiology. Groups associated with mucosal lesions in the aerodigestive and genital tracts include HPV types 6 and 11 with a low malignant potential; HPV 16 and 18 with a much higher malignant potential; and HPV 31 and 33 with a malignant potential that lies somewhere in between. At least 90 different types of HPV have been identified and are designated by numbers (i.e., HPV 6). The closer the numbers, the more similar the viral subtypes are in their clinical manifestations.
The induction of cellular proliferation is a fundamental property of HPV though its mechanism of action remains unclear. The present understanding is that HPV establishes itself in the basal layer, where viral DNA enters the cell and is transcribed into RNA which is then translated into viral proteins. The genome consists of three regions: an upstream regulatory region and the two regions named according to the phase of infection in which they are expressed, the early (E) and late (L) regions. The E region genes are involved in potent oncogenes that are responsible for the replication of the viral genome, interaction with host cell intermediate filaments, and transforming activities. The L region genes encode the viral structural proteins (11). Several factors are involved in the regulation of cell proliferation.
It is likely that the host immune system plays an important role in the pathogenesis of HPV-induced lesions. Both humoral and cellular immune responses may be compromised in children with RRP and a patient’s immunocompetence may also influence the clinical course of disease. The role of cytokines such as interleukin-2, interleukin-4, and interleukin-10 and expression of the major histocompatibility complex antigens in the malfunction of the cell-mediated immune response in children with RRP have been demonstrated (12). Papillomas with a reduced expression of these antigens may evade immune surveillance and allow the disease to progress more rapidly.
HPV is thought to infect stem cells within the basal layer of epithelium (13,14,15). Following infection of the stem cells, the viral DNA can either be actively expressed, or it can exist as a latent infection in epithelium that remains clinically and histologically normal. During latency, there is very little viral RNA present. In fact, HPV DNA can be detected in normal-appearing mucosa in RRP patients who have been in remission for years, explaining why reactivation and clinical recurrence can occur following many years of remission (16,17,18). Thus, reactivation of viral expression can occur any time following establishment of a latent infection. Adult-onset respiratory papillomas could reflect either activation of virus present since birth or an infection acquired in adolescence or adult life. Gene products of early genes E6 and E7, and possibly E5, are required for papilloma induction, but the details of the mechanism of HPV activation are unknown. In order to “cure” RRP, it is necessary to modulate the host response to the virus and, ideally, eliminate the latent infection.
The universality of HPV in the lower genital tract rivals that of any other sexually transmitted disease in humans. It is estimated that at least 1 million cases of genital papillomas occur per year in the United States, affecting about 1% of the population (19). These most often manifest as condylomata acuminata involving the cervix, vulva, or other anogenital sites in women or the penis of male sexual partners of affected women. In addition, it is estimated that colposcopic (subclinical) changes affect nearly 5 million US women. An additional 14 million women, or about 10% of the female population of child-bearing age, are DNA positive but have no visible lesions while more than 80 million women, or 60% of the at-risk population, are HPV antibody positive but DNA negative. The incidence of HPV infections in sexually active young college women is high, with a cumulative incidence of 43% over a 36-month period in a recent study (20). Clinically apparent HPV infection has been noted in 1.5% to 5% of pregnant women in the United States (21). As in RRP, HPV 6 and 11 are the most common subtypes identified in cervical condylomata.
Recurrent Respiratory Papillomatosis
Histologically, RRP appears as pedunculated masses with fingerlike projections of nonkeratinized stratified squamous epithelium supported by a core of highly vascularized connective tissue stroma (Fig. 94.1). The basal layer may be either normal or hyperplastic, and mitotic figures are generally limited to this layer. Cellular differentiation appears to be abnormal, with altered expression and production of keratins. The degree of atypia may be a sign of premalignant tendency.
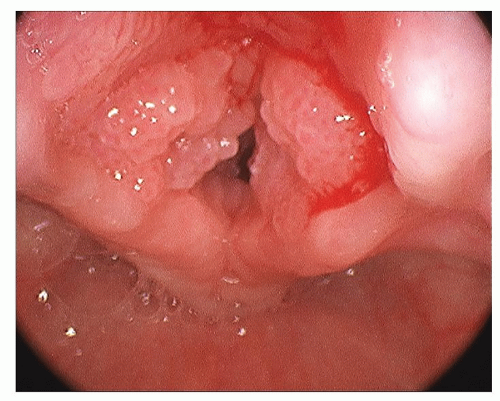
RRP lesions occur most often at anatomic sites in which ciliated and squamous epithelium are juxtaposed (22). The most common sites for RRP are the limen vestibuli, the nasopharyngeal surface of the soft palate, the midline of the laryngeal surface of the epiglottis, the upper and lower margins of the ventricle, the undersurface of the vocal folds, the carina, and at bronchial spurs (6). In tracheotomized patients, RRP is often encountered at the stoma and in the mid-thoracic trachea, areas that might be considered iatrogenic squamociliary junctions. Papilloma lesions may be sessile or pedunculated and often occur in irregular exophytic clusters (Fig. 94.2). Typically, the lesions are pinkish to white in coloration. Iatrogenic implantation of papilloma may be preventable
by avoiding injury to nondiseased squamous or ciliated epithelium adjacent to areas of frank papilloma. Ciliated epithelium undergoes squamous metaplasia when exposed to repeated trauma and is replaced with nonciliated epithelium that creates an iatrogenic squamociliary junction. This may also explain the observation that RRP flourishes in the presence of uncontrolled gastroesophageal reflux with an increased incidence of laryngeal scarring (23).
by avoiding injury to nondiseased squamous or ciliated epithelium adjacent to areas of frank papilloma. Ciliated epithelium undergoes squamous metaplasia when exposed to repeated trauma and is replaced with nonciliated epithelium that creates an iatrogenic squamociliary junction. This may also explain the observation that RRP flourishes in the presence of uncontrolled gastroesophageal reflux with an increased incidence of laryngeal scarring (23).
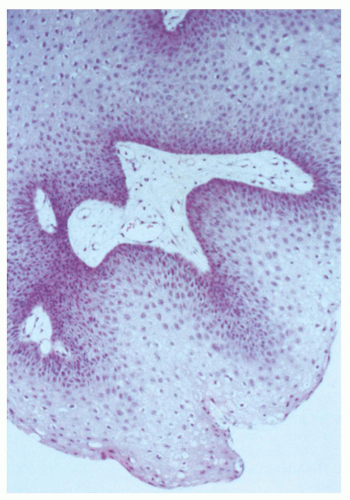 Figure 94.1 Histologic section of papilloma, demonstrating fingerlike projection of nonkeratinized stratified squamous epithelium and vascularized connective tissue stroma. |
EPIDEMIOLOGY
RRP may affect people of any age, with the youngest patient identified at 1 day of age and the oldest at 84 years (5). JORRP (arbitrarily defined as patients diagnosed at less than 12 years of age) is most often diagnosed between 2 and 4 years of age. The distribution of RRP among boys and girls is approximately equal, and there are no apparent differences in surgical frequencies by gender or ethnicity (8). AORRP peaks between the ages of 20 and 40 years and has a slight male predilection. The mode of transmission for AORRP is unknown. While it is postulated that transmission is from either latent virus present from birth or adult exposure through sexual contact, these theories have yet to be fully substantiated.
Numerous studies have been performed to elucidate the true incidence of RRP. The national registry of children with RRP, composed of the clinical practices at 22 pediatric otolaryngology sites (arguably a skewed population of more severely affected children), calculates a mean number of procedures at 19.7 per child, with an average of 4.4 procedures per year (8). Children diagnosed before 3 years of age were found to be 3.6 times more likely to require more than four surgical procedures per year and 2.1 times more likely to have two or more anatomic sites involved than those diagnosed after their fourth birthday (8). Children with disease progression are diagnosed at younger ages than those who remain stable or become disease-free (24). Younger children are more likely to have persistent disease and more often experience an increased number of surgeries in the first year following diagnosis (25).
In a survey of practicing otolaryngologists in the United States, half of the adults with RRP had required fewer than five procedures over their lifetime compared with less than 25% of the children. Approximately equal percentages of children and adults (17% children vs. 19% adults) had very aggressive RRP (defined as requiring more than 40 lifetime operations), although adults had more years to accumulate these operations (5). The true incidence and prevalence of RRP are uncertain. In a Danish subpopulation incorporating 50% of the population of that country, the incidence of laryngeal papillomatosis was 3.84 cases per 100,000 (26). The rate among children was 3.62 per 100,000, whereas adult-onset cases occurred at a rate of 3.94 per 100,000. These figures are comparable with those found in a US survey, which estimated an incidence in the pediatric population of 4.3 cases of RRP per 100,000 children and 1.8 cases of RRP per 100,000 adults (5). Armstrong et al. (27) estimated that there are 80 to 1,500 incident cases of JORRP annually in the United States and 700 to 3,000 prevalent cases of JORRP based upon a 1999 epidemiologic analysis of two US cities.
Recently, Campisi et al. created a national database incorporating all children (less than 14 years old) with RRP in Canada treated by Pediatric Otolaryngologists. This study found the national incidence of JORRP from 1994 to 2007 to be 0.24 per 100,000 with a prevalence of 1.11 per 100,000 (28). These estimates are significantly lower than several previous studies, but similar to a population-based study of RRP patients in Seattle and Atlanta (27). The authors attribute this discrepancy to either over-estimation by other studies based on extrapolated data or higher incidences in other countries.
Interestingly, a recent pilot study of a large database of publicly and privately insured patients in the United States consistently showed that RRP incidence was higher in publicly insured patients compared to those with private insurance (3.21 vs. 1.98 per 100,000, respectively) (29). An explanation for this finding is that patients with public insurance often come from a lower socioeconomic level than those with private insurance. A cross-sectional study of all active JORRP patients from the Hospital for Sick Children in Toronto showed that nearly half of these patients were below the poverty line in Canada (30). This study, however, showed no correlation between socioeconomic status and severity of disease.
RRP places a large economic burden on individual patients and their families as well as our society as a whole. On average, a child presenting to an academic center in the United States with RRP requires 19.7 procedures over their lifetime, with a mean frequency of procedures being 4.4 per year (5). Approximately equal numbers of adults and children with RRP (17% vs. 19%, respectively) will have aggressive disease requiring more than 40 lifetime procedures. The average lifetime cost to treat one patient with RRP has been estimated at $60,000 to $470,000 in the United States (29).
In addition to the economic burden, RRP significantly impacts quality of life. Lindman et al. (31) utilized a validated quality of life instrument to assess patients with JORRP. Compared with healthy controls, children with RRP self-reported significantly poorer quality of life in the areas of psychosocial health, social functioning, and school functioning. Children with RRP had quality of scores similar to children with other chronic medical conditions.
Transmission
The precise mode of HPV transmission remains unclear. Several studies have convincingly linked childhood-onset RRP to mothers with genital HPV infections, whereas circumstantial evidence suggests that adult disease may be associated with oral-genital contact. Retrospective and recent prospective studies have confirmed that HPV may be passed by vertical transmission from mother to child (32,33,34). Silverberg showed that children born to mothers with active condylomata had a 231-fold increased risk of developing RRP when compared to children born to disease-free mothers (35). In addition, they showed that children born to women with active condylomata had a twofold higher risk of developing RRP if labor lasted more than 10 hours. Kashima found that childhood-onset RRP patients were more likely to be first born and vaginally delivered than were control patients of similar age (36). The authors hypothesized that primagravid mothers are more likely to have a long second stage of labor and that the prolonged exposure to HPV in the birth canal leads to a higher risk of infection in the first born child. They also suggested that newly acquired genital HPV lesions are more likely to shed virus than long-standing lesions. This would explain the higher incidence of RRP observed among the offspring of young mothers of low socioeconomic status—the same group that is more likely to acquire sexually transmitted infections such as HPV.
Hallden showed that 54% of JORPP patients were born to mothers with a history of vulvar condylomata at the time of delivery (37). Despite this apparent close association, few children exposed to genital warts at birth actually develop clinical disease (38). It is not well understood why RRP develops in so few children whose mothers have condylomata. Although HPV could be recovered from the nasopharyngeal secretions of 30% of infants exposed to HPV in the birth canal the number of infants expected to manifest evidence of RRP is only a small fraction of this (39). Based on these data, it appears that secondary factors including patient immunity; timing, length, and volume of virus exposure; and local traumas (intubation, extraesophageal reflux) must be important in the development of RRP. Reports of neonatal papillomatosis suggest that, in at least some cases, development of the disease may occur in utero. Because caesarean section does not seem to prevent the development of RRP in all cases, a better understanding of the risk factors associated with RRP is needed before the efficacy of caesarean delivery in preventing papilloma disease can be fully assessed (40).
A case-control study found that patients with AORRP had more lifetime sexual partners and a higher frequency of oral sex than matched controls (36). This data would suggest patients with AORRP are exposed later in life than patients with JORRP. However, HPV has the disturbing capability to form latent infections in the basal cell layer of otherwise healthy appearing mucosa (16,17,18). It has been suggested that AORRP may represent a reactivation of HPV infection acquired during birth instead of a de novo exposure during adulthood.
Viral DNA has been detected in areas of “normal appearing mucosa” adjacent to papilloma lesions, suggesting a possible explanation for the recurrence of the disease following thorough surgical removal (18,38). Brandsma reported finding HPV DNA in 4% of random, clinically normal biopsies of the airway (41). AORRP, thus, could reflect either activation of virus present since birth or an infection acquired in adolescence or adult life. Nosocomial acquisition of virus by patients in the operating room
(OR) also may result theoretically from improper equipment sterilization or room preparation. However, current Occupational Safety and Health Administration recommendations have virtually eliminated this risk.
(OR) also may result theoretically from improper equipment sterilization or room preparation. However, current Occupational Safety and Health Administration recommendations have virtually eliminated this risk.
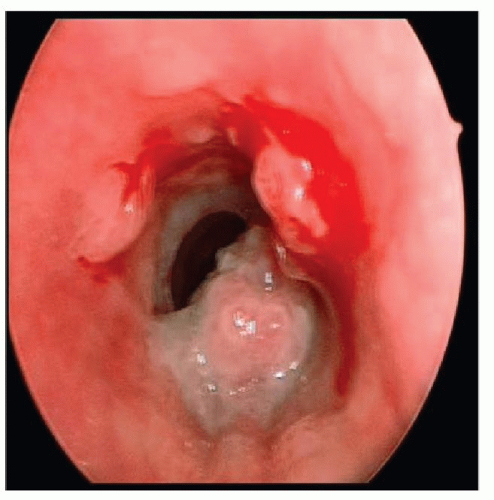
Although any site along the upper aerodigestive tract can be affected, HPV shows a predilection to infect sites where ciliated columnar and squamous epithelia are juxtaposed (8). This explains the frequent involvement of the false vocal folds, the upper and lower margins of the ventricle, and the undersurface of the true vocal folds. In tracheotomized patients, papillomas often are encountered at the stoma and in the mid-thoracic trachea, which are areas where iatrogenic trauma to ciliated epithelium often induces squamous metaplasia (see Fig. 94.3). It is postulated that an eddying flow of the mucous blanket at squamociliary junctions may concentrate infectious viral particles at these sites. Children with bronchopulmonary dysplasia who require prolonged endotracheal intubation also may be at increased risk for development of RRP. An endotracheal tube may play the same role in the mechanical dissemination or implantation of papilloma virus as a tracheotomy in this setting, through the interruption of the continuous respiratory mucosal surface. Gastroesophageal reflux disease (GERD) also has been identified as a potential risk factor for disease persistence, although additional research is necessary to verify this anecdotal observation.
CLINICAL FEATURES
Because the most common symptoms of RRP are related to airway obstruction, it is not uncommon for children to be misdiagnosed initially as having asthma, croup, or chronic bronchitis. The hallmark of RRP in children is the triad of relentlessly progressive hoarseness, stridor, and respiratory distress. Hoarseness in children tends to be overlooked or at least accepted until it reaches a certain level of severity. However, any infant or young child with symptoms of voice change, along with obstructive airway symptoms or recurrent croup, warrants laryngoscopy to rule out neoplasia, with RRP being the most likely lesion.
Children with RRP most often present with some degree of dysphonia. Stridor is often the second clinical symptom to develop, beginning as an inspiratory noise and becoming biphasic with progression of the disease. Less commonly, chronic cough, recurrent pneumonia, failure to thrive, dyspnea, dysphagia, and acute life-threatening events may be the presenting symptoms. The duration of symptoms prior to diagnosis varies with a typical period of about 1 year from onset of symptoms till a diagnosis of RRP is made. Not uncommonly, a mistaken diagnosis of asthma, croup, allergies, vocal nodules, or bronchitis is entertained before arriving at the definitive diagnosis of RRP.
Because of the rarity of RRP and the slowly progressive nature of the disease, some cases may go unrecognized until respiratory distress results from papillomas obstructing the airway. The result is a relatively high need for tracheotomy to be performed in these children. Shapiro et al. (42) noted that RRP tracheotomy patients presented at a younger age and with more widespread disease, often involving the distal airway prior to tracheotomy. In the Centers for Disease Control and Prevention (CDC) registry, children with tracheotomy were initially diagnosed with RRP at a younger age (2.7 years) than those without a tracheotomy (3.9 years) (7). Both prolonged tracheotomy and the presence of subglottic papilloma at the time of tracheotomy have been associated with an increased risk of distal tracheal spread.
Extralaryngeal spread of respiratory papillomas has been identified in 13% to 30% of children and in 16% of adults with RRP (5,43). The most common sites of extralaryngeal spread were, in order of frequency, the oral cavity, trachea, and bronchi (5). In a recent review of the experience at three academic medical centers, 12% of the children with RRP had distal tracheal spread of their disease and 7% had pulmonary dissemination. One common thread in the children with distal spread was the use of jet ventilation anesthesia techniques in the surgical management of their disease (44). Pulmonary RRP begins as asymptomatic, peripheral nodules and progresses to enlarging lesions with central cavitation and necrosis. Pulmonary RRP is insidious and progresses over years. It can eventually manifest as respiratory failure due to destruction of the lung parenchyma and malignant transformation.
The course of RRP is variable with some patients experiencing spontaneous resolution. However, RRP is frequently characterized by relentless recurrence and most patients necessitate frequent surgical debridements. Certain patient factors such as younger age at diagnosis and HPV 11 subtype are linked to aggressive progression of RRP. A possible
link between RRP and immunodeficiency states also has been observed. Both children and adults with acquired immunodeficiency syndrome, congenital immunodeficiencies, or those on immune suppression after organ transplantation have been identified with RRP (7). Respondents in a recent American Society of Pediatric Otolaryngology (ASPO) survey related at least one death from RRP in their clinical practices (43). When death occurs, it is usually associated with a complication of frequent surgical procedures or caused by respiratory failure due to distal disease progression.
link between RRP and immunodeficiency states also has been observed. Both children and adults with acquired immunodeficiency syndrome, congenital immunodeficiencies, or those on immune suppression after organ transplantation have been identified with RRP (7). Respondents in a recent American Society of Pediatric Otolaryngology (ASPO) survey related at least one death from RRP in their clinical practices (43). When death occurs, it is usually associated with a complication of frequent surgical procedures or caused by respiratory failure due to distal disease progression.
Dysplastic and malignant transformation have been reported in RRP. In a recent study by Sajan et al. (45), RRP biopsy specimens of 20 patients with JORRP were examined. Only 1 of 123 specimens (less than 1%) had evidence of dysplasia. Dysplasia in AORRP appears to be more prevalent with 22% to 55% of patients demonstrating evidence of dysplasia on histopathology (46,47). An RRP task force survey reported malignant transformation of RRP into squamous cell carcinoma (SCC) in 26 cases (43). The mechanism of RRP transformation remains poorly understood. In a recent study by Lee in which six AORRP patients developed laryngeal carcinoma, the only risk factor associated with malignant transformation was an absence of HPV DNA on polymerase chain reaction of papilloma specimens (48).
PATIENT ASSESSMENT
History
Persistent or progressive stridor and dysphonia, with the possible development of respiratory distress, are the most consistent signs and symptoms of RRP in children (Table 94.1). In the absence of severe respiratory distress, a careful history should be obtained. Information regarding the time of onset of symptoms, possible airway trauma including a history of previous intubation, and characteristics of the cry are obviously important. Hoarseness, although a common and often benign clinical complaint in young children, always indicates some abnormality of structure or function. Because of the precision of laryngeal mechanics, hoarseness may result from a remarkably small lesion and thus be an early sign in the course of a disease process. On the other hand, if the lesion’s origin is remote from the vocal cords, hoarseness may present as a late sign. Although histologically the same lesion, a papilloma that produces hoarseness in one patient may produce stridor and obstruction in another, depending on the size and location of the lesion. The quality of the voice change may give only limited clues to its etiology, whereas other characteristics such as age of onset, rate of progression, associated infection, history of trauma or surgery, and the presence of respiratory or cardiac distress may be of much greater significance. A low-pitched, coarse, fluttering voice suggests a subglottic lesion, whereas a high-pitched, cracking voice, aphonia, or a breathy voice suggests a glottic lesion. Associated high-pitched stridor also suggests a glottic or subglottic lesion. Although stridor that has been present since birth is more often associated with laryngomalacia, subglottic stenosis, vocal cord paralysis, or a vascular ring, it should be realized that neonates also can present with papillomatosis.
TABLE 94.1 COMMON PRESENTING SYMPTOMS IN CHILDREN WITH RRP | |||
|---|---|---|---|
|
Associated symptoms such as feeding difficulties, allergic symptoms, vocal abuse, and the presence of hereditary congenital anomalies may help distinguish RRP from alternative diagnoses, including vocal fold nodules, vocal fold paralysis, subglottic cysts, subglottic hemangioma, and subglottic stenosis. In the absence of any history suggesting these lesions, review of the perinatal period may reveal a history of maternal or paternal condylomata. If the onset of stridor and dysphonia is gradual and progressive over weeks or months, then neoplastic growth compromising the airway must be considered and investigated.
Certainly not every child with a hoarse voice or cry merits investigation beyond an assessment of the symptom. However, in the presence of hoarseness with respiratory distress, tachypnea, decreased air entry, tachycardia, cyanosis, dysphagia, chronic cough, failure to thrive, recurrent pneumonia, or dysphagia, the larynx must be visualized and a firm diagnosis of the cause of hoarseness must be made. Any child with slowly progressive hoarseness merits investigation and the clinician should not wait until total aphonia or airway problems occur.
Physical Examination
Children presenting with symptoms consistent with RRP must undergo a thorough and organized physical examination. The child’s respiratory rate and degree of distress must first be assessed. The physician should observe the child for tachypnea or the onset of fatigue that may indicate impending respiratory collapse. The child should be observed for flaring of the nasal ala and the use of accessory neck or chest muscles. Increasing cyanosis and air hunger may cause the child to sit with the neck hyperextended in an attempt to improve airflow. If a child is gravely ill, additional examination should not be undertaken outside the OR, the emergency room, or the intensive care unit, where resuscitation equipment for intubation of the airway, endoscopic evaluation, and possible tracheotomy are readily available.
In the stable, well-oxygenated child, additional examination can proceed. The most important part of the examination is auscultation with the aid of a stethoscope. The physician should listen over the nose, open mouth, neck, and chest to help localize the probable site of the respiratory obstruction. A useful technique is to pull the bell off the stethoscope and listen over these areas with the open tube. The respiratory cycle, which is normally composed of a shorter inspiratory phase and a longer expiratory phase, should then be observed. Stridor of a laryngeal origin is most often musical and may begin as inspiratory, but will progress to biphasic with worsening airway narrowing. Infants with stridor should be placed in various positions to elicit any changes in the stridor. A child with RRP would not be expected to demonstrate much change in the stridor with position change in contrast to infants with laryngomalacia, a vascular ring, or a mediastinal mass. Pulse oximetry can add an accurate quantitative analysis of the child’s respiratory state. In the stable patient in whom asthma is a likely diagnosis, pulmonary function testing combined with arterial blood gas evaluation also may be helpful.