Chapter 64 Radio-Guided Parathyroid Exploration
Introduction
Contemporary parathyroid operations are successful: surgery for primary hyperparathyroidism has evolved to a procedure with a greater than 95% success rate and a complication rate of less than 2%. A favorable outcome depends on an accurate diagnosis, appropriate patient selection, and good operative technique. Use of adjunctive approaches such as radio-guided parathyroidectomy and intraoperative parathyroid hormone (PTH) measurements can facilitate success (see Chapter 63, Intraoperative PTH Montioring during Parathyroid Surgery).
Radio-guided parathyroidectomy is to be viewed as a version of minimally invasive parathyroidectomy.1,2 We believe that this is a useful adjunct, especially in difficult parathyroidectomies, that leads to an improved outcome with minimal added cost and morbidity (see Chapters 60, Minimally Invasive Single Gland Parathyroid Exploration, 61, Minimally Invasive Video-Assisted Parathyroidectomy, and 62, Local Anesthesia for Thyroid and Parathyroid Surgery).
Principles of Radio-Guided Parathyroid Surgery
Radio-guided parathyroidectomy is closely related to other radio-guided techniques used for breast cancer, malignant melanoma, thyroid cancer, and colon cancer.3,4 A radiotracer is administered, which accumulates preferentially in the target tissue. Radio-guided techniques are used to localize specific tissue through the detection of radioactivity, theoretically minimizing dissection by a direct approach and decreasing overall operative time.
In the case of parathyroid tissue, 99mTc-sestamibi is utilized. Because of increased mitochondrial density in hypercellular parathyroid glands, the hyperfunctional parathyroid tissue becomes “hot” relative to surrounding thyroid.25 99mTc-sestamibi is not truly specific to parathyroid tissue, as it also accumulates in salivary, thyroid, and cardiac tissues, as well as some malignancies. This phenomenon was first described by Coakley in 1989 to preoperatively localize hyperactive parathyroid with 99mTc-sestamibi imaging.5 This finding was then utilized in the operative setting by Martinez et al. in 1995.6 Their series of three pediatric patients was the first description in the literature of radio-guided parathyroid surgery.
Radio-Guided Parathyroidectomy
Preoperative Evaluation
Preoperatively, patients are evaluated and diagnosed in the manner standard to the institution at which they are being treated. Typically, this includes a history and physical exam, as well as serum calcium, serum PTH, and preoperative localization imaging (see Chapters 56, Primary Hyperparathyroidism: Pathophysiology, Surgical Indications, and Preoperative Workup, 57, Guide to Preoperative Parathyroid Localization Testing, and 58, Principles in Surgical Management of Primary Hyperparathyroidism).
Preoperative Localization
If the 99mTc-sestamibi scan localizes a single parathyroid adenoma, a minimally invasive parathyroidectomy utilizing an intraoperative gamma probe would be feasible (see Chapter 57, Guide to Preoperative Parathyroid Localization Testing).7 It is important that surgeons review all preoperative imaging to localize abnormal glands, as other specialties may be more reluctant to commit to a diagnosis of localization.7 In patients with a negative 99mTc-sestamibi scan, a cervical ultrasound is advised. The sensitivity of cervical ultrasound for the identification of parathyroid adenomas has been demonstrated to be between 70% and 80%.8,9,29 This also has the advantage of assessing for synchronous thyroid disease.10 If the ultrasound also proves to be negative, the decision is presented to the patient. Further imaging with a CT scan may be pursued, or the parties may proceed to radio-guided parathyroidectomy with negative preoperative localization studies. If desired, a perioperative bilateral internal jugular venous sampling of parathyroid hormone for localization may be of some utility when all localization scans are negative.11 We feel this test, generally reserved for reoperative cases, is rarely necessary.
Anesthesia and Other Operative Considerations
Minimally invasive parathyroidectomy can easily be performed under general endotracheal anesthesia. Either because of contraindications or patient preference, it can also be performed under local anesthesia with intravenous sedation. Initially described by LoGerfo,12 utilization of local anesthesia with monitored anesthesia care (MAC) has been validated for parathyroidectomy. Snyder et al. have shown comparable operative results, clinical results, and patient satisfaction between local anesthesia with monitored anesthesia care and general anesthesia.13 MAC may include the use of laryngeal mask anesthesia.
In cases of known bilateral disease, local anesthetic can be extended to the opposite side. This also applies to patients who are shown to have multigland disease intraoperatively. If local anesthesia has been chosen, conversion to general anesthesia should not be delayed in case of complications, unexpected findings, or patient discomfort (see Chapter 62, Local Anesthesia for Thyroid and Parathyroid Surgery).
Radiotracer Injection
Basic Principles
Radio-guided resection is also feasible for patients with secondary and tertiary hyperparathyroidism.17 Because of the high likelihood of multigland disease, preoperative 99mTc-sestamibi scans are typically not obtained for these patients unless there is suspicion of ectopic disease. They have a lower sensitivity and specificity for localization of multigland disease.29 Thus, patients with secondary or tertiary hyperparathyroidism will get only one dose of 99mTc-sestamibi the day of surgery for intraoperative localization.
Protocol 1: Wisconsin
On the day of surgery, patients are injected intravenously with 10 mCi 99mTc-sestamibi approximately 1 to 2 hours before surgery.3 An important consideration is that 99mTc decays with an approximate half-life of 6 hours. It has been shown that intraoperative gamma probe localization is most successful within a very narrow time window of 1 to 3 hours after radiotracer injection.1,15 In our series, the median time from injection to surgery was 90 minutes, with a range from 30 to 420 minutes. Despite this large range, intraoperative radio-guided localization was successful in all cases. More consistent protocols are recommended, but these data suggest that an additional injection or procedure cancellation are not required if delays occur.16
Alternatively, a same-day protocol can be pursued with a full dose of 20 mCi 99mTc-sestamibi in the nuclear medicine department on the day of surgery; scintigraphy for localization is performed at 20 minutes and 2 hours postinjection.17 Gamma probe–directed minimally invasive parathyroidectomy was performed approximately 3 hours following initial radiotracer injection. This protocol is advantageous in that it offers a same-day experience for the patient. At our institutions, this is occasionally utilized for patients who live some distance away or for patients with hypercalcemic crisis requiring urgent operative intervention.
Background Counts
The localization of hyperfunctioning parathyroid tissue by radioguidance is based on the detection of radiotracer counts above background counts. 99mTc-sestamibi washes out of the thyroid gland faster than hyperfunctioning parathyroid tissue. Consequently, an enlarged parathyroid gland should have higher counts than the thyroid. The background counts are based on thyroid gland activity and is set based on the thyroid isthmus (see figure 64-1). Therefore, our first step intraoperatively is to place the gamma probe on the thyroid isthmus to determine background counts (see Figure 64-1).
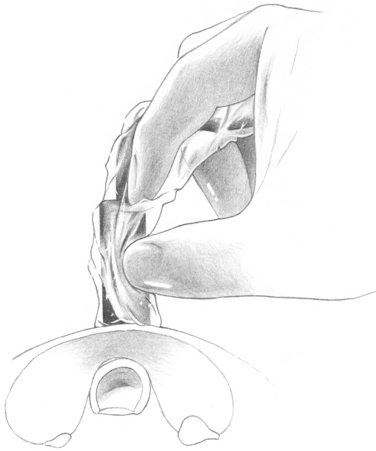
Figure 64-1 Background counts. The gamma probe is placed over the thyroid isthmus to measure the background counts.
Finding the Hyperfunctioning Gland: In Vivo Counts
Once the incision is made and the sternohyoid and sternothyroid muscles are retracted, the gamma probe is inserted into the skin incision directly over the presumed location of the adenoma (Figure 64-2). Intraoperative scanning is performed looking for radionuclide counts greater than background in order to localize abnormal parathyroid glands. It is crucial to position the probe at different angles to triangulate counts to ensure specificity. The counts obtained by scanning on the identified enlarged parathyroid gland in situ are recorded as in vivo counts and expressed as a percentage of the background counts.
An in vivo-to-background percentage higher than 150% strongly suggests the presence of a parathyroid adenoma.15 In more than 180 enlarged parathyroid glands, a mean in vivo count of 150% of background was demonstrated;18 moreover, all hyperfunctioning glands had in vivo counts that were greater than background. The neck is routinely scanned during the dissection to determine the trajectory to the hyperfunctioning parathyroid gland, minimizing the amount the tissue dissected.
Removal of the Parathyroid Gland: Ex Vivo Counts and the “20% Rule”
After excision of the enlarged parathyroid, the tissue is placed on top of the gamma probe while directed away from the patient to determine ex vivo counts (Figure 64-3). Ex vivo counts are also expressed as a percentage of background counts. If the ex vivo parathyroid count is greater than 20% of background, the tissue is identified as parathyroid adenoma (i.e., the “20% rule”).12 In an earlier study of 180 resected parathyroid glands, 179 had ex vivo counts greater than 20% of background. Importantly, no fragments of fat, thyroid, or lymph node had counts greater than 20% of background, emphasizing the specificity of the cutoff.19–21 Thus, the 20% rule functions quite well overall.
Protocol 2: Arkansas
After the induction of either endotracheal or laryngeal mask airway (LMA) anesthesia, the neck is extended with a roll placed between the shoulders, and the head is placed on a circular headrest.22 A 2-cm horizontal incision is created in the midline halfway between the cricoid and the sternal notch. Subcutaneous electrocautery dissection is performed, undermining in all directions, and the subcutaneous fat is grabbed with a forceps. This subcutaneous fat is removed off of the strap muscles. The adipose tissue is reserved as a radiation count negative control. A Gelpe retractor is then used to expose the surgical site, the handle placed opposite the side of the anticipated adenoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree