Principles of Radiation Oncology
Umamaheswar Duvvuri
Gregory J. Kubicek
Radiation oncology plays an important role in the treatment of head and neck cancers, both in the adjuvant and the definitive setting. Radiation is an important modality and has a large influence in terms of both survival and toxicity for patients. The field of radiation oncology is rapidly evolving, and head and neck oncologists should be aware of some of the important and changing features of this field to ensure that the patients they refer are treated appropriately. The goals of this chapter are to review the fundamental basis of radiation oncology (radiobiology), to describe some of the everyday aspects of radiation therapy delivery and toxicity, and finally to describe some of the new innovations in the field of radiation oncology.
RADIOBIOLOGY
Radiobiology describes the effects of ionizing radiation on the cellular level. Ionizing radiation induces cell death via DNA damage. DNA damage occurs when radiation interacts directly with the DNA strand or, more commonly, when the radiation interacts with a water molecule with secondary DNA damage. When ionizing radiation hits a water molecule, it forms free radicals; these free radicals are very unstable and, if formed in the vicinity of the DNA strand, are able to inflict damage.
DNA damage does not always lead to cell death; the cell has the capability of repairing radiation-induced damage to DNA. The rate and capability for repair are different for different types of cells. In general, more rapidly growing cells are more susceptible to damage from radiation (the process of cell division makes the DNA more susceptible to damage and also DNA damage is declared during DNA division). This is why the mucosa of the oral cavity (which is always dividing and being replaced) is very sensitive to the effects of radiation. In general, normal tissues have greater capacity for DNA repair than cancer tissues; one of the rationales for fractionation of radiation is to allow repair of normal tissues during fractions.
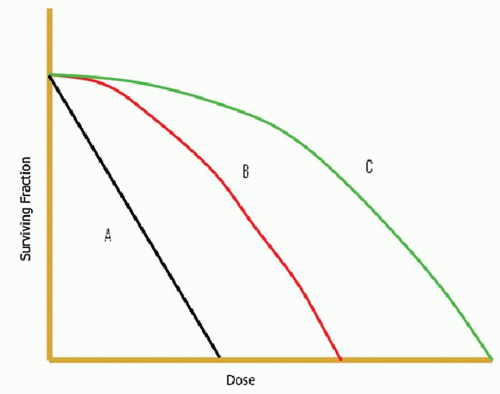
The more radiation damage that is inflicted upon a population of cells, the more cell death; the initial radiation dose causes damage to the cell population but does not necessarily cause cell death. As the radiation damage accumulates, cell death starts to occur. A simplistic way to view this is to imagine that both strands of the DNA must be damaged before the cell is irreversibly killed; the initial dose of radiation is most likely to damage a number of single strands and occasionally both strands in a single cell. As radiation dose continues, it becomes more likely that double-strand damage occurs in more cells and thus causes cell death. During the initial (single strand) DNA damage, there is potential for the cell to repair the single-strand break and undo the radiation damage. The differential ability for DNA repair has led to the terms “radiosensitive” and “radioresistant.” Although the exact terms are very subjective, the basic principle is that some types of cells and tumors have very little repair ability and die rapidly from even low doses of radiation. On the other side are “radioresistant” tumors; such tumors have ability to repair DNA, which makes them less likely to undergo cell death from radiation damage. Some typical radiosensitive tumors include lymphomas, while radioresistant tumors include sarcomas and melanomas. In Figure 110.1 we have three cell populations; cell population A would be radiosensitive (note the lack of a shoulder region and the immediate linear decline in surviving cells; such a curve is seen in blood cells that lack any repair mechanisms). Population C has a wide shoulder representing a radioresistant cell population; the initial portion of the curve is where cell repair is occurring preventing death from the radiation.
In addition to the capacity for DNA repair, another component that affects radiation sensitivity (and the slope of the cell death curve) is oxygenation. Under hypoxic conditions, it is thought that the free radicals, which are
responsible for DNA damage, can be scavenged by the hypoxia-induced acidic environment and that oxygen itself may play a role in continuing the cascade of the free radical formation. Large tumors tend to grow faster than their blood supply, leading to hypoxic areas in the center of large tumors. This phenomenon is thought to contribute to the relatively poor control rates for large head and neck cancers treated with ionizing radiation.
responsible for DNA damage, can be scavenged by the hypoxia-induced acidic environment and that oxygen itself may play a role in continuing the cascade of the free radical formation. Large tumors tend to grow faster than their blood supply, leading to hypoxic areas in the center of large tumors. This phenomenon is thought to contribute to the relatively poor control rates for large head and neck cancers treated with ionizing radiation.
DOSE AND DELIVERY
The most commonly used form of radiation is photons, but other particles can also be used, including electrons, protons, neutrons, and carbon ions. Electrons are widely available but not as commonly used as photons. Electrons have a short depth of penetration; this allows for good dose to the surface and minimal dose to deeper levels (e.g., 6 MV electrons will deposit 90% of their energy 2 cm into tissue). Because of the dose distribution, electrons are a very good treatment option for the treatment of skin cancers. Protons and carbon ions are other forms of radiation and are less frequently used. Protons, neutrons, and carbon ions (referred to as heavy particle radiation) are not produced by normal radiation oncology linear accelerators and require special (and very expensive) accelerators to produce this type of radiation (see below for a more detailed description of proton radiation oncology).
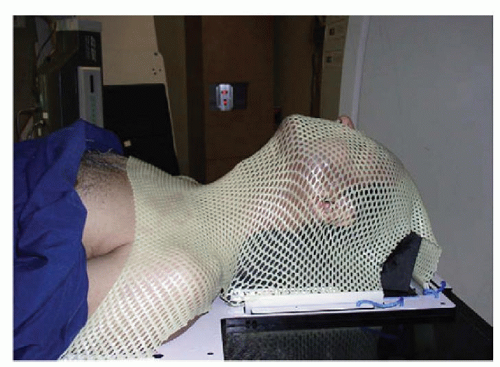
It should be noted that all types of radiation work via the same basic mechanisms although some of the logistics of delivery are different. For photon radiation (usually just described as radiation), the radiation is produced by a linear accelerator (Fig. 110.2). The patient lies on the treatment table, and the linear accelerator rotates around the patient to deliver the radiation. In order to ensure that the patient is not moving while the radiation is being delivered, an immobilization device is used; for HNC this typically consists of the thermoplastic head mask (Fig. 110.3).
Radiation is described in terms of dose; the common convention for dose is the gray (Gy). The definition of a Gy is the amount of energy dose absorbed per unit mass (1 Gy = 1 J/kg). Less often used is rads, 1 rad is equal to 0.01 Gy. The typical total dose for treatment of HNC is 60 to 66 Gy for adjuvant radiation and 70 to 74 Gy for definitive radiation. By comparison, doses for lymphoma would be 30 to 40 Gy, and the dose for a computed tomography (CT) scan is 0.1 to 0.2 Gy.
The total dose of radiation is divided into smaller doses called fractions. The dose for a fraction of radiation is usually between 1.8 and 2.0 Gy, although there can be a wide range (e.g., the dose for radiosurgery in the treatment of
trigeminal neuralgia can be as high as 80 Gy in a single fraction). The purpose of delivering the total dose of radiation as a number of smaller fractions is to take advantage of normal tissue repair of DNA damage. For the most part, normal tissues have a much greater ability for DNA repair than do cancer cells. Thus, if damage is incurred to both normal tissues and cancer cells, the normal tissues will repair themselves while the cancer cells will die (this is not completely accurate as there is still a lot of normal tissue damage as discussed later).
trigeminal neuralgia can be as high as 80 Gy in a single fraction). The purpose of delivering the total dose of radiation as a number of smaller fractions is to take advantage of normal tissue repair of DNA damage. For the most part, normal tissues have a much greater ability for DNA repair than do cancer cells. Thus, if damage is incurred to both normal tissues and cancer cells, the normal tissues will repair themselves while the cancer cells will die (this is not completely accurate as there is still a lot of normal tissue damage as discussed later).
The interplay between dose and fraction is sometimes referred to as “radiobiologically equivalent dose.” Several mathematical models exist to convert from one dose and fractionation to another dose at a different fractionation (e.g., in terms of spinal cord tolerance, at 3 Gy per fraction, the maximum dose is 30 Gy, whereas at 2.0 Gy per fraction, the maximum dose is 45 Gy). The differential in repair between normal and cancer cells is magnified as radiation is delivered over a long period of time. This allows for improvement of the toxicity profile and minimizes toxicity to normal tissues.
FRACTIONATION AND TREATMENT TIME
The most common fractionation pattern is to deliver the radiation every day (Monday through Friday) and is referred to as “conventional” or standard fractionation. Hypofractionation is delivering less than one fraction per day (typically done in radiosurgery where the fraction is given every other day). Hyperfractionation is delivering more than one fraction per day. While there is repair of normal tissue DNA between radiation fractions, there is also (hopefully to a lesser extent) repair of cancer DNA and potential growth of the cancer itself. In order to decrease this risk, there have been several studies that have investigated the potential advantages of hyperfractionation.
The most well-known study is RTOG 90-03 (1). In this study, patients with stage III and IV HNC where randomized to conventional radiation (standard arm), hyperfractionation with twice daily treatments, hyperfractionation with twice daily treatments with a planned 2-week break during the radiation, and accelerated hyperfractionation or concomitant boost (which consists of daily radiation until the final 12 treatments at which time radiation is given twice daily). The results of this study showed an improvement in local control in the hyperfractionation and accelerated hyperfractionation arms (survival had a trend toward improvement, but did not reach statistical significance). Several other studies have shown similar results including DHANCA where patients were randomized to five versus six treatments per week, and local control was found to be improved with the six fraction regimen (5 year local control 70% vs. 60%) (2).
It should be noted that while tumor control appears to be improved with a reduction in treatment time, there is also an increase in acute toxicity (the normal tissues now have less ability to repair). However, the studies have not found a significant increase in late toxicity.
While hyperfractionation appears to be one method to improve outcome, chemotherapy is another. Chemotherapy has been shown to improve outcome versus radiation alone in both the definitive and the adjuvant setting. Brizel et al. (3) found that concomitant chemotherapy delivered with hyperfractionation provided better locoregional control than hyperfractionated radiation alone (70% vs. 44%). Brizel et al. (4) performed a mathematical analysis of the data obtained from RTOG 90-03 to determine what the impact of chemotherapy was on the radiobiologically equivalent dose. They determined that a 1% increase in radiobiologically equivalent dose lead to a 1.1% increase in locoregional control. Similarly, the addition of chemotherapy effectively increased the total radiobiologically equivalent dose by about 10 Gy. The authors concluded that this increase in effective radiation dose could not be safely achieved by simply increasing the amount of radiation delivered to the patient.
RTOG 0129 (5) set out to examine if chemotherapy (cisplatin 100 mg/m2 every 3 weeks) plus hyperfractionated radiation (concomitant boost) was better than the same chemotherapy plus standard (once daily) radiation. The study did not find a difference between the two arms, and given the increase in acute toxicity and logistical difficulties with hyperfractionation, most institutions use once daily treatments for locally advanced HNC treated with chemoradiotherapy.
In summary, based on the known benefits of chemotherapy and the negative RTOG 0129 study, hyperfractionation is typically only used for patients who cannot get chemotherapy and have to be treated with radiation alone.
The overriding principle for the benefits of hyperfractionation is that a reduction in overall treatment time corresponds to an improvement in tumor control. The opposite is also true; when the overall treatment time is extended, tumor control decreases (6,7,8,9). RTOG (6) looked at the importance of treatment time in HNC radiation and found that local failure increased when the total treatment time (from surgery to completion of radiation) exceeded 11 weeks. This is why treatment breaks should be avoided unless absolutely necessary.
RADIATION ONCOLOGY PLANNING
The first step in radiation oncology planning is consultation with a radiation oncologist to examine the patient and discuss the risks and benefits of therapy. After this, a radiation planning CT scan is performed. This is similar to a diagnostic CT scan except that the CT scan is performed with the patient in the same position as they will be when receiving the daily radiation treatments. In order to reduce setup error between and during treatments, patients will have a plastic mask made that conforms to their facial
anatomy (Fig. 110.3). Because of the special positioning (including the mask), a diagnostic CT scan cannot be used as a substitute for a radiation planning scan (although the information from a diagnostic CT scan can certainly be used to help in planning). Sometimes a positron emission tomography (PET) scan can be done at the same time as radiation planning (see section on PET scans).
anatomy (Fig. 110.3). Because of the special positioning (including the mask), a diagnostic CT scan cannot be used as a substitute for a radiation planning scan (although the information from a diagnostic CT scan can certainly be used to help in planning). Sometimes a positron emission tomography (PET) scan can be done at the same time as radiation planning (see section on PET scans).
After the radiation planning scan has been performed, the radiation oncologist will begin planning the radiation beams. This process has evolved over time (see below) but in all cases involves distinguishing normal structures from areas that are targeted for radiation. Several terms are commonly used to describe this process (Table 110.1). Gross tumor volume (GTV) represents the areas of actual tumor; this can be defined either radiographically (CT, PET, MRI) or clinically. In patients who have had surgery, there should not be a GTV since there is no longer any gross disease. Clinical tumor volume (CTV) defines areas that are at risk for harboring microscopic disease (such as lymph node basins); there can be discrepancy between different physicians in deciding the CTV volumes (e.g., does the contralateral neck need to be covered in a patient with tonsil cancer). Planning tumor volume (PTV) is a small expansion on the CTV or GTV to account for errors in daily setup. A typical PTV expansion is 0.3 to 1.0 cm. At our institution, we use daily imaging verification to reduce random setup error and are able to use a smaller PTV margin (3 to 5 mm). Organ at risk (OAR) is defined as normal tissue that is not involved with cancer and needs to be protected from radiation (examples include the spinal cord, parotid glands, mandible, etc.). Depending on the degree of risk, different areas will receive different doses of radiation. For example, the gross disease (GTV) will typically receive 70 to 74 Gy; areas of low risk (uninvolved lymph node basins) are treated to 50 to 54 Gy, while intermediate risk regions receive 60 to 66 Gy (Table 110.2).
TABLE 110.1 ORADIATION TERMS | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|