Prenatal Development of the Eye and It’s Adnexa
Cynthia S. Cook
Victoria Ozanics
Frederick A. Jakobiec
In this text, we attempt to provide an overview of ocular embryology by describing essential developmental events in a concise fashion. Fine structural data on human and primate eye components have become available since the appearance of the original publications on ocular embryology by Mann,1 Barber,2 Dejean et al.3 and Duke-Elder et al.4 These observations aid in reconfirming or reevaluating the functional development of ocular structures as expressed by morphologic changes. Our descriptions are based on mammalian tissues, including both humans and other species that serve to model human development. Comparisons have demonstrated that the sequence of developmental events is similar across species. Factors that must be taken into consideration when making interspecies comparisons include: duration of gestation; differences in anatomic endpoint (such as the absence in other species of a macula, Schlemm’s canal, or Bowman’s membrane); and when eyelid fusion breaks (during the sixth month of gestation in the human versus 2 weeks postnatally in the mouse). Within the limits of these species variation, mice have proven to be a valuable model in the study of normal and abnormal ocular morphogenesis. In particular, the study of effects of acute exposure to teratogens during development has provided valuable information about the specific timing of events leading to malformations.
In development of the eye, as in other organs, the multiplication of cells as well as directional change in shape, structure, and function of the cells govern growth. Gene determination decides the direction in which a change can occur, whereas the reciprocal demands of the individual cells or parts determine how far that direction must be followed. Fundamentally, the process consists of these two activities: change in structure and shape due to relatively different rates of growth and also change in structure and function due to differentiation and functional specialization.
Induction of one ocular tissue by another and interrelations between these developing tissues have been extensively reinvestigated in many laboratories using various experimental techniques.5,6,7,8,9,10,11,12,13,14 One example is the lens, which arises in direct response to induction by the optic vesicle. The developing lens, in turn, promotes normal morphogenesis of neural ectodermal and mesenchymal elements in the eye. It has an inducing influence on corneal differentiation and promotes vitreous growth. Moreover, a strong organogenetic connection exists between the lens and iris. The reciprocal interactions between the optic cup and lens bring about the functional adjustment of the ocular axes.
Although the neural retina grows and differentiates independently of the lens, the presence of the lens may influence the normal growth and change in shape of the pigment epithelium, choroid, and sclera. The pigment epithelium, however, directs the deposition of the mesenchyme around it; subsequently, all three layers grow in unison. The pigment epithelium also depends on the vitreous body for increase in its area.
Early Morphogenesis
Although events occurring during the first few weeks after fertilization, before the appearance of identifiable ocular primordia, may seem to have little significance to the clinical ophthalmologist, evidence indicates that abnormalities that originate during this period may be responsible for many ocular malformations that occur in humans.
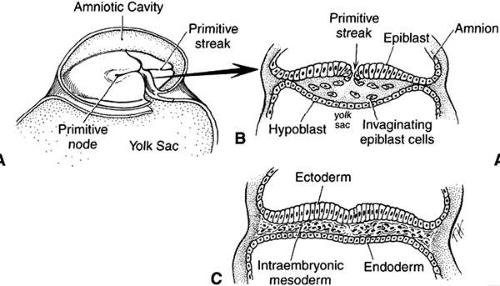
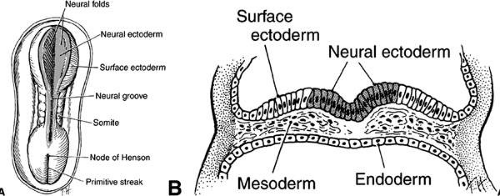
Gastrulation (formation of the mesodermal germ layer) occurs early in gestation (day 7 in mice, day 20 in humans). The primitive streak forms as a longitudinal groove within the epiblast (future ectoderm) of the bilaminar embryonic disc. Epiblast cells migrate medially toward the primitive streak where they invaginate to form the mesodermal layer (Fig. 1). This forms the classic three germ layers: ectoderm, mesoderm, and endoderm. Gastrulation progresses in a cranial to caudal direction. Concurrently, cranial surface ectoderm proliferates forming bilateral elevations called neural folds (Fig. 2). Columnar surface ectoderm in this area now becomes neural ectoderm.
Experimental studies in mice using acute exposure to teratogens have demonstrated the significance of the period of gastrulation to later ocular development. Exposure to ethanol or retinoic acid during a short period equivalent to the third week of human gestation causes primary damage to the forebrain neural ectoderm.15,16,17 This results in a spectrum of malformations including microphthalmia, anterior segment dysgenesis (Peters anomaly), iris and optic nerve colobomas, and persistent hyperplastic primary vitreous.18,19
As the neural folds elevate and approach each other (neurulation), a specialized population of mesenchymal cells, the neural crest, emigrates from the neural ectoderm at its junction with the surface ectoderm. In the development of the eye, the neural ectoderm (deriving from the neural plate and neural folds), the surface ectoderm, the neural crest, and, to a lesser extent, the mesoderm are of importance (Table 1).
TABLE 1. Embryonic Origins of Ocular Tissues | |
|---|---|
|
The cranial neural crest contributes most of the connective tissues of the eye and its adnexal structures.13,20,21,22,23,24,25,26,27,28 The hyaluronic acid-rich extracellular matrix influences migration and differentiation of the neural crest cells. This acellular matrix is secreted by the surface epithelium as well as the neural crest cells and forms a space through which crest cells migrate. Fibronectin secreted by the noncrest cells forms the limits of the mesenchymal migration. Interactions between the migrating neural crest and the associated mesoderm appear to be essential for normal crest differentiation. Many congenital malformations of the anterior segment and cornea probably arise from derangements in the axial migration of ocular neural crest.
Experimental embryologic studies have shown that the mesoderm actually contributes little to head and neck mesenchyme. The cranial correlates to the paired paraxial somites are called somitomeres. Seven pairs of cranial somitomeres have been identified in the mouse.23,28,29,30,31,32,33,34,35,36,37,38 In the eye, the mesoderm contributes only to the striated extraocular muscles and vascular endothelia. To these limited primary mesodermal elements come associated neural crest satellite cells (surrounding the striated muscles) and pericytes (surrounding the vascular endothelium). Circulating blood elements originate from mesoderm. The term mesenchyme broadly refers to any embryonic connective tissue and should not be confused with mesoderm. With respect to the head and neck, most of this connective tissue derives from the cranial neural crest, with the exceptions mentioned.
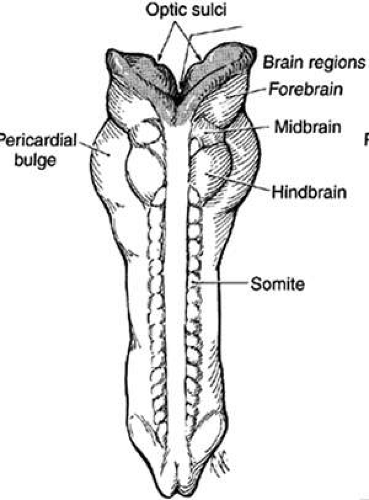
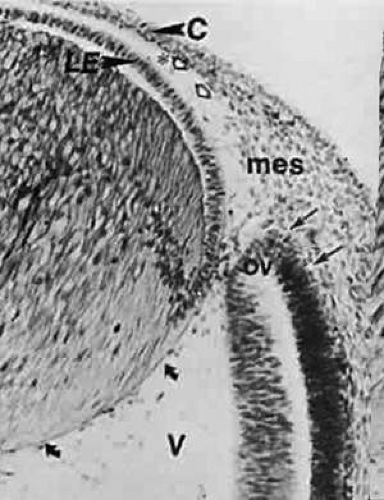
The optic primordium is a thickened zone in the differentiating central nervous system that forms the neural folds of the early embryo. Some of the neuroepithelium composing the optic primordium becomes the future optic cup and stalk; some cells may delaminate to contribute to the neural crest. The optic sulcus or groove arises in the primordium at the time when the neural folds are still open in the forebrain (8 to 15 somite pairs, approximately 2 to 3.5 mm) (Figs. 3 and 4A). With enlargement of the sulcus, the optic evaginations and, later, the optic pits appear in the region of the future forebrain (see Fig. 4B). The portion of the evaginations adjacent to the midbrain contacts the mesencephalic neural crest cells, which will form the mesenchymal envelope isolating neural from surface ectoderm (see Fig. 4C).
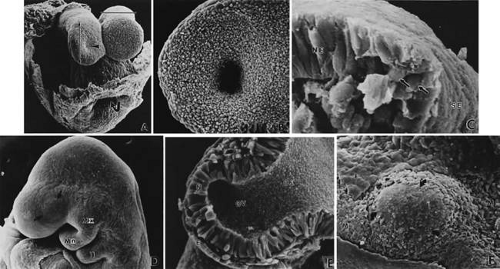 FIGURE 4. Formation of optic sulcus and vesicle. Mouse embryos are illustrated and follow a sequence nearly identical to that observed in human embryos. A. A scanning electron micrograph of a mouse embryo at day 8 (six somite pairs) equivalent to the human at 4 weeks of gestation. The amnion has been removed. Arrowhead indicates the optic sulcus, an evagination of the forebrain (FB). The entrance to the foregut is indicated by the curved arrow (165×). B. Optic sulcus continues to deepen through a process of microtubule-mediated cell elongation and microfilament-mediated apical constriction (387×). C. Embryo fractured through the midbrain in a plane indicated by the horizontal line in Figure 2A. Note the junction between the columnar neural ectoderm (NE) and the flattened surface ectoderm (SE). This is the area from which the mesenchymal neural crest cells emigrate (arrows) to join the underlying mesoderm. These mesencephalic neural crest cells will populate the area around the optic primordia and give rise to most of the ocular connective tissue (1,071×). B. Embryo of 25 somite pairs (fifth week in a human), the bulge of the enlarging optic vesicle (arrows) can be appreciated externally. Mx, maxillary prominence of the first visceral arch: Mn, mandibular prominence of the first visceral arch; II, second visceral arch (447×). E. Frontal fracture at the level of the optic vesicle (OV). The optic vesicle is lined by the columnar neural ectoderm (N) and enlarges, approaching the surface ectoderm (E). The optic stalk (OS) is continuous with the cavity of the forebrain (440×). F. Removal of the surface ectoderm (E) from an embryo of 25 somite pairs reveals the exposed basal lamina of the optic vesicle (arrows). Enlargement of the optic vesicle has displaced the adjacent mesenchyme (M) so that the basal lamina of the surface ectoderm (E) is in direct contact with that of the optic vesicle (214×). |
At approximately the 24th day (2 to 4 mm) with the closure of the neural tube, the optic pits are pushed outward away from the central nervous system and toward the surface ectoderm. The two lateral bulges, caused by the outward extension of the growing optic pits, become pouch-shaped vesicles at about the 25th day of development (20 somite pairs) (Fig. 5; see Fig. 4D and E).
The optic vesicles become sheathed with cells of neural crest origin that, except for a small region in the center of the bulge, separate them from the surface ectoderm (see Fig. 4E). The future primordium of the retina is present before closure of the neural tube when the neural ectoderm is still open to the amniotic cavity. The optic stalk is formed by a constriction of the area between the vesicles and the future forebrain. At this time, all cells lining the inner surface of the vesicle’s cavity are ciliated, and its outer surface, as well as the inner aspect of the surface ectoderm overlying it, is covered by a thin basal lamina.
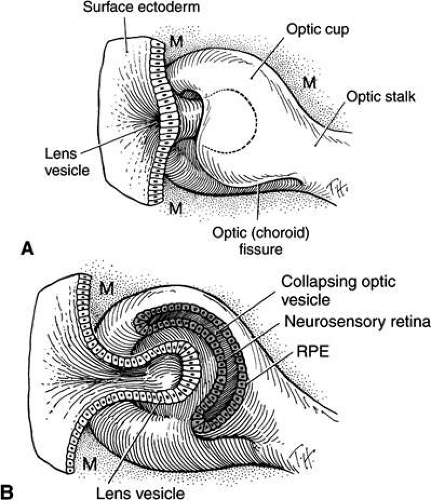
The next event is invagination of the optic vesicles by differential growth and buckling to form the optic cup (Figs. 6,7,8,9). The temporal and lower walls move inward against the upper and posterior walls. This process also involves the optic stalk so that the optic (choroid/embryonic/retinal) fissure is formed where the two laterally growing edges of the cup and stalk meet. Mesenchyme (primarily neural crest) penetrates immediately into the cup by filling up the fissure.
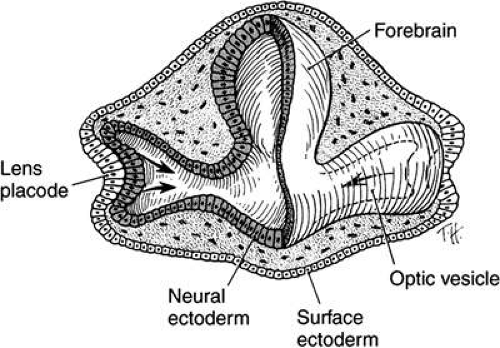 FIGURE 6. Drawing of a transection through a 28-day-old embryo showing invaginating lens placode that is pushing into the optic vesicle (arrows), thus creating the optic cup. Note the orientation of the eyes 180 degrees from each other. This is also illustrated in Figures 9B and C. (Cook CS, Sulik KK, Wright KW: Embryology. In Wright KW (ed): Pediatric Ophthalmology and Strabismus. St Louis: Mosby, 3–43, 1995) |
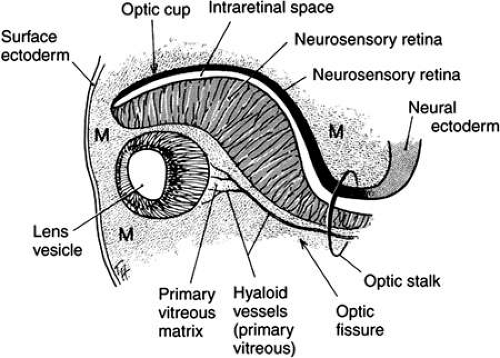 FIGURE 8. Drawing of cross section at approximately 5 weeks of gestation through optic cup and optic fissure. The lens vesicle is separated from the surface ectoderm. Mesenchyme (M) surrounds the developing lens vesicle, and the hyaloid artery is seen with the optic fissure. See also Figure 9F. (Cook CS, Sulik KK, Wright KW: Embryology. In Wright KW (ed): Pediatric Ophthalmology and Strabismus. St Louis: Mosby, 3–43, 1995) |
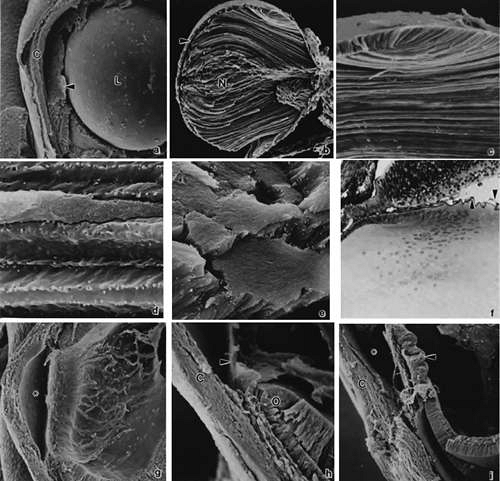
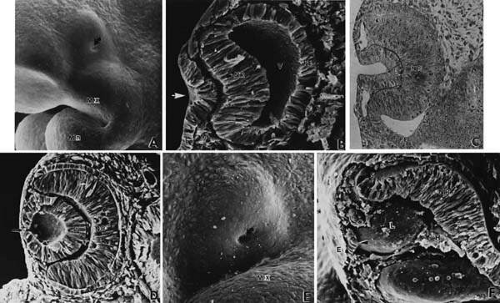 FIGURE 9. Invagination of the optic cup and lens vesicle. Mouse embryos are illustrated. A. Embryo of somite pairs (fifth week in a human). Upon external examination, the invaginating lens placode can be seen (arrow). Note its position relative to the maxillary (Mx) and mandibular (Mn) prominences of the first visceral arch (106×). B. Embryo of the same age as in Figure 3A. Frontal fracture through the lens placode (arrow) illustrates the associated thickening of the surface ectoderm (E). Mesenchyme (M) of neural crest origin is present adjacent to the lens placode. Distal portion of the optic vesicle thickens concurrently, as the precursor of the neural retina (NR), whereas the proximal optic vesicle becomes a shorter, cuboidal layer that is the anlage of the retinal pigmented epithelium (PE). The cavity of the optic vesicle (V) becomes progressively smaller (367×). C. Epithelium of the lens placode continues to invaginate (L). There is an abrupt transition between the thicker epithelium of the placode and the adjacent surface ectoderm, which is not unlike the transition between the future neural retina (NR) and the future pigmented epithelium (PE). (Periodic acid-Schiff’s stain; 443×) D. As the lens vesicle enlarges during the eleventh day, the external opening, or lens pore (arrow), becomes progressively smaller. The lens epithelial cells at the posterior pole of the lens elongate to form the primary lens fibers (L). NR, anlage of the neural retina; PE, the anlage of the pigmented epithelium (now a very short cuboidal layer) (300×). E. External view of the lens pore (arrow) and its relationship to the maxillary prominence (Mx)—32 somite pairs (260×). F. Frontal fracture reveals the optic fissure (*) where the two sides of the invaginating optic cup meet. This forms an opening in the cup allowing access to the hyaloid artery (H), which ramifies around the invaginating lens vesicle (L). The former cavity of the optic vesicle is obliterated except in the marginal sinus (S) at the transition between the neural retina (NR) and the pigmented epithelium. E, surface ectoderm (307×). |
The optic vesicle and optic stalk invaginate through differential growth and infolding. Local apical contraction39 and physiologic cell death40 have been identified during invagination. This process progresses from inferior to superior so that the sides of the optic cup and stalk meet inferiorly in the optic fissure. The two lips of the optic fissure meet and initially fuse anterior to the optic stalk with fusion progressing anteriorly and posteriorly. Failure of normal closure of this fissure may result in inferiorly located defects (colobomas) in the iris, choroid, or optic nerve.
Closure of the optic cup through fusion of the optic fissure allows establishment of intraocular pressure. Studies have demonstrated that, in the chick, the protein in the embryonic vitreous humor is derived from plasma proteins entering the eye by diffusion out of permeable vessels in the anterior segment.41 After optic fissure closure, protein content in the vitreous decreases, possibly through dilution by aqueous humor produced by developing ciliary epithelium.
Table 2 lists the chronologic sequence of ocular development and comparative body-eye measurements in relationship to embryonic time intervals.
TABLE TWO. Revised Sequence of Human Ocular Development | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Lens Induction and Differentiation
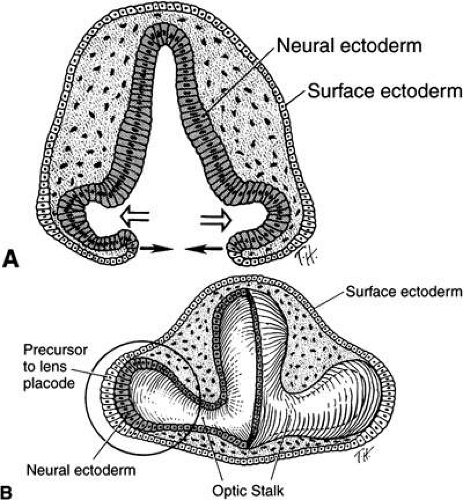
As the optic vesicles enlarges, it contacts the overlying surface ectoderm. The first manifestation of lens induction is the appearance of a disc-shaped thickening of surface epithelial cells (27 days of gestation) (see Figs. 5B, 6, and 9A and B). A tight, extracellular matrix-mediated adhesion between the optic vesicle and the surface ectoderm has been described. This anchoring effect on the mitotically active ectoderm results in cell crowding and elongation and formation of a thickened placode. Adhesion between the optic vesicle and lens placode serves to ensure alignment of the lens and retina in the visual axis. Although adhesion between the optic vesicle and surface ectoderm exists, the respective basement membranes remain separate and intact throughout the contact period (see Fig. 4F). Arrested development at the lens stalk stage resulting in aphakia in mice is associated with deletion n the promoter of the homeobox transcription factor Pitx3.42 Otx2, Pax6 and Lens1 found within the lens ectoderm are induced by the anterior neural plate. Pax6 forms complexes with transcription factors and is required for expression of cMaf, MafA/L-Maf, Six3, and Prox1. Contact between the optic vesicle and surface ectoderm promotes expression of mafs, Soxs, and Prox1. These transcription factors are responsible for crystalline expression, cell elongation, and arrest of the cell cycle in the primary lens fibers.43 Other transcription factors identified in this process include: Meis, Six3, FoxE3, and Prox1.44,45 Inducers for lens formation may act on the regulation of structural genes or they may act directly on the cell cytoplasm. Lens induction, therefore, may involve transfer of inductor substances from the optic cup to the surface cells across both basement membranes. Invagination of the lens placode (29 days) is accomplished by a synergistic elongation of the placode cells with contraction of their apical cytoplasm and terminal bar system (see Figs. 7 and 9C). The processes of differentiation into a lens pit, cup, and then a vesicle have been studied in detail.55,56,57,58
As the lens placode invaginates, it forms a hollow vesicle (see Figs. 8 and 9D). The area of contact of the optic vesicle and the surface ectoderm determines the size of the lens vesicle, orbit, and palpebral fissure. The lens separates from the surface epithelium at about 33 days of gestation (7 to 9 mm; see Fig. 9D). The vesicle consists of a single layer of cells, which is covered by a basal lamina. Through appositional growth to its epithelial surface, the basal lamina acquires more layers that become the lens capsule. At first, the posterior capsule is more prominent than the anterior; the outer layers may have components from the mesodermal tissues forming the hyaloid vascular network. A zone of necrosis develops, displacing the lens placode from the surface ectoderm (see Fig. 9E and F). The process of lens vesicle detachment is accompanied by active migration of epithelial cells out of the keratolenticular stalk, cellular necrosis, and basement membrane breakdown.59,60 Coordinated interaction between the neuroepithelium of the optic cup and the neural crest derived mesenchyme is mediated by transcription factors, in particular Pax6, found in both cell types.61 Cup formation is achieved by contraction of the apical filaments. The process of induction is thus localized.
Primary Lens Fibers
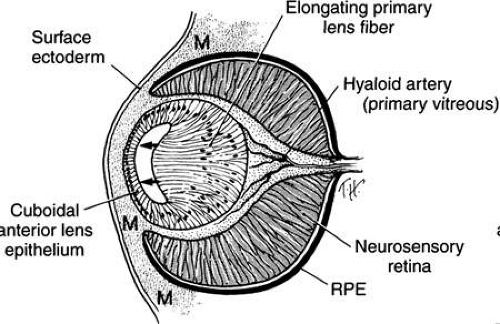
The hollow lens vesicle consists of a single layer of epithelial cells with cell apices directed toward the center. Following detachment from the surface ectoderm, the lens vesicle is surrounded by a basal lamina, the future lens capsule. The cells lengthen (Figs. 10 and 11A) until the lumen of the vesicle is filled (45 days, 17 mm). These constitute the primary lens fibers. The apical ends of the newly formed fibers become firmly attached to the apical surface of the anterior lens epithelium.
The retinal anlage promotes primary lens fiber formation in the adjacent lens epithelial cells. Surgical rotation of the lens vesicle in the chick’s eye by 180 degrees results in elongation of the lens epithelial cells nearest the presumptive retina, regardless of the orientation of the transplanted lens.58 Thus, the retina develops independently from the lens, whereas the lens appears to rely on the retina for cytodifferentiation. This transformation of primary lens fibers is accompanied by ultrastructural changes in the nucleus and cytoplasm, decreased numbers of organelles, and increased numbers of fibrillar materials composed of the characteristic lens proteins. The primitive lens filled with primary lens fibers forms the embryonal nucleus, visible in the adult. This portion of the lens lacks sutures.
Secondary Lens Fibers
The cells nearest the corneal primordium remain cuboidal and become the lens epithelium, which remains mitotic throughout life and gives rise to future lens fiber cells. Production of the secondary lens fibers is initiated by migration of the anterior epithelial cells toward the equator and their elongation at various degrees with a shift in their nuclear distribution, thus resulting in the lens bow (Fig. 12B, C, and F, and 13; see Figs. 11B and C). The basal ends of the fibers remain tightly attached to the basal lamina; their apical ends extend anteriorly to the center, thus forming the anterior suture. The tips of these secondary fibers are not yet tapered. Coordinated regulation of cell-matrix and cell-cell adhesion is required to precise migration and elongation of the secondary lens fibers.62 A corresponding increase in cell volume and decrease in intercellular space within the lens accompany lens fiber elongation.47 The lens fibers exhibit surface interdigitations. They extend around the primary fibers beneath the capsule and meet in planes, the lens sutures, arranged essentially vertically to the surface. The basic anatomy of the lens is established after the first layer of secondary fibers has been placed (seventh week of gestation).63
Lens Sutures
Succeeding generations of cells extend anteriorly and posteriorly from the equator beneath the capsule. The anterior suture line is shaped like a Y that is inverted in the posterior aspect. The posterior suture is formed when the posterior central cells lose their nuclei, become separated from their basal lamina, and migrate inward.56 Curved lens fibers result with the superficial ones being the longest. Linear and triradiate sutures form, which represent different stages in lens development.
Maturation
The shape of the lens and its orientation with respect to the optic axis continually adjust to the developing eye. This is partly regulated by the neural retina and peripheral mesenchyme.7 Through the third month of gestation, the anteroposterior diameter is greater than the equatorial. Mainly because of the continued generation of secondary fibers, the equatorial diameter increases rapidly, thus making the lens more and more ellipsoid. The lens, still somewhat spherical at birth, grows throughout life.
A general structural densification occurs progressively during maturation. Fibrillar material is increased within the cytoplasm and cell organelles are decreased. The successive parallel layers of interdigitating, elongated lens fibers become tightly apposed (see Fig. 12D and E). Deeper nuclei become homogenous and dense. By the end of the third month, the innermost cells have lost their nuclei and simultaneously show disintegration of the chromatin and the ribosomes, leaving a finely filamentous cytoplasm.
Connective Tissue Coats
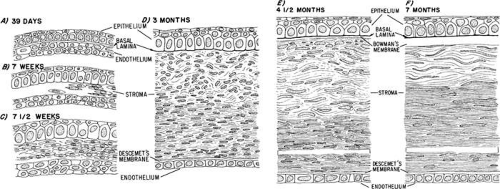
Cornea
Among the many publications on the morphogenesis of the cornea (Fig. 14) and the development of its constituents in various vertebrates, only a few can be cited in this general review.
Epithelium
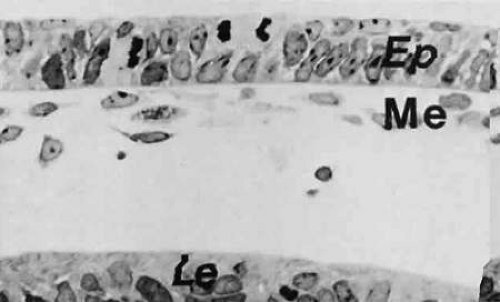
When the lens cup separates from the surface ectoderm in embryos at approximately 33 days of postfertilization (7 to 9 mm in length), development of the cornea can be said to have begun. The surface ectoderm becomes continuous covering the optic cup and lens vesicle and later develops into the corneal epithelium.
Descemet’s Membrane and Endothelium
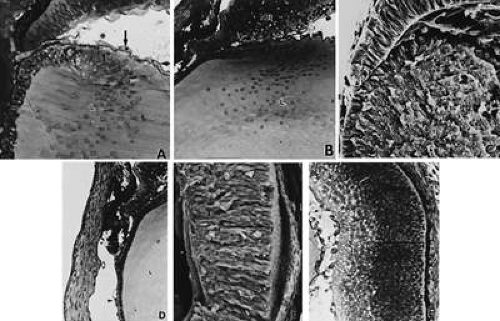
During the next week, mesenchymal cells grow centrally between the basal laminae of the lens and corneal epithelium (Fig. 15; see 14A-C). Posterior to the basal lamina of the corneal epithelium, the mesenchyme has produced a double row of flattened cells, the future corneal endothelium (see Fig. 14A).
Descemet’s membrane first appears at 8 weeks as a patchy accumulation resembling basement membrane material.68 The patches become confluent and thickened due to the synthetic activity of the endothelial cells. Evidence of organization is seen early during the fourth month when four or five superimposed lamellae interspersed with collagen fibrils appear on the stromal side of the endothelial basal lamina. The endothelium has zonulae occludentes at the cell apices by the middle of the fourth month of development. Their appearance corresponds to the onset of aqueous humor formation.
Stroma
Following formation of the corneal endothelium, mesenchyme (neural crest) continues to migrate axially over the rim of the optic cup during the seventh week (17 to 18 mm) (Fig. 16). At 8 weeks (18 to 22 mm), migrating mesenchymal cells from the periphery invade the space between epithelium and endothelium. This mesenchyme, as well as that which will give rise to the sclera and iris stroma, is of neural crest origin.21 The central portion of the future stroma is still acellular (see Fig. 14B). The endothelium merges with the stratified cells at the periphery over the lips of the optic cup. This mass of cells, in turn, is continuous with the cellular scleral condensation extending to the equator of the globe. The developing keratocytes begin to produce glycosaminoglycans.
In the early 8-week-old embryo, approximately 22 mm in length, the mesenchymal stroma consists centrally of five to eight rows of cells (Fig. 14C) within a fibrillar matrix containing collagen. Nerves have been identified within the corneal stroma and between epithelial cells at 3 months.79
The most posterior layers of the corneal stroma are confluent peripherally with a condensed band of mesenchyme that is gradually spreading backward to enclose the eye. The mesenchyme destined to form the sclera is not distinct from that which will form the four oculomotor muscles.
The cornea at 2 months (about 20 mm) consists of an epithelium of outer squamous and basal columnar cells. The middle polygonal or wing cells of the adult do not appear until the fourth or fifth month. The stroma has about 15 layers of cells with rapidly developing collagen fibrils with most in the posterior portion. At 3 months, the endothelium of the central area consists of a single row of flattened cells that seem to rest on an interrupted basal lamina, which is the first indication of a thin Descemet’s membrane. With the exception of Bowman’s membrane, all corneal components are present (see Fig. 14D).
Bowman’s Membrane
Arising relatively late in gestation (see Fig. 14E and F), Bowman’s membrane is observed by light microscopy during the fifth month, but somewhat earlier by electron microscopy. It is always acellular, presumably formed by the most anterior fibroblasts of the stroma, which move posteriorly as Bowman’s fibers and the ground substance are synthesized. The epithelium may play a partial role in the local polymerization of the collagen precursors presumably produced by the most anterior stromal fibroblasts.
Transparency
Perhaps the most important and unique corneal characteristic is its transparency, which also develops during fetal life. The early embryonic and fetal cornea is translucent rather than transparent and is more hydrated than in the adult. Condensation begins in the posterior stroma during fetal maturation.70 At about the time that the most anterior stromal lamellae are formed, corneal transparency reaches adult quality. During this development, the water content of the cornea is being reduced so that the adult level of corneal hydration is attained at the same time as transparency.
Coincident with eyelid opening, there are alterations in gene expression within the cornea, differentiation of the epithelium, and thinning of the stroma.80
Sclera
The sclera forms first anteriorly, by mesenchymal condensation at the limbus near the future insertion of the rectus muscles and grows gradually posteriorly. Fibrocytes are involved in the synthesis of the elastic foci in the sclera.81 In contrast, the cornea lacks elastic components.
Inspection of the sclera at 60 to 65 mm or 12 weeks reveals it as a mesenchymal condensation that has reached the posterior pole of the eye and surrounds the optic nerve. Some cells have entered among the optic nerve fibers and are arranged transversely, forming the first stages of the connective tissue lamina cribrosa. The scleral spur appears at 4 months as circularly oriented fibers; at 5 months, it is visible behind the anterior chamber. At this time the sclera is well differentiated all around the eye.
Although the corneal and scleral cells are derived from the same mass of mesenchyme surrounding the anterior part of the optic cup, they behave differently when in their definitive position. Corneal fibroblasts form collagen faster than the scleral cells and differ in the rate and amount of noncollagenous protein that they synthesize.
Structures of the Aqueous Outflow Pathways
Iridocorneal Angle
Light and scanning electron-microscopic studies reveal the anterior chamber angle of the human eye to have a continuous endothelial lining during the third and fourth months (Figs. 17 and 18). The tissues in the angle later differentiate into a loose reticulum with large enclosed spaces near the iris and ciliary body; outside of this trabecular tissue, a tighter aggregation of cells is oriented toward the sclera.82,83,84,85 With the growth of surrounding structures, Schlemm’s canal comes to lie at the level of the apex of the angle. Descemet’s membrane and the corneal endothelium still cover a portion of the trabecular meshwork, but the endothelial lining of the chamber has become discontinuous (Figs. 19 and 20). The loose reticular tissue of the earlier stages now occurs only in the deepest part of the angle where it has large intercellular spaces (see Figs. 17C and 20).
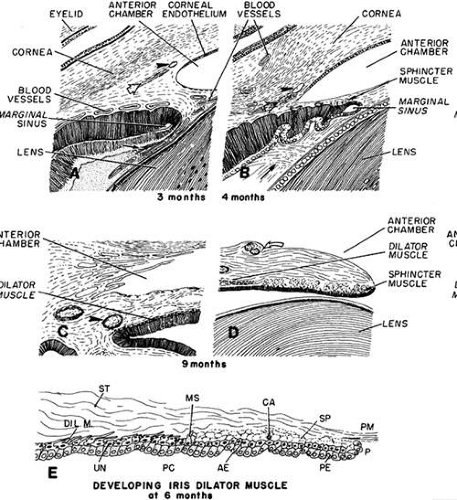 FIGURE 17. Schematic diagram of the progressive deepening of the angle; its relation to the neighboring tissues. A. At 3 months, corneal endothelium extends nearly to the angle recess: an incipient Schlemm’s canal (arrowhead) and a more posterior scleral spur condensation (hollow arrow) appear to its left. Pigment epithelium of the forward growing ectodermal optic is indented by blood vessels. The secondary vitreous fibrils run parallel to its surface (arrow). This is the faisceau isthmique or marginal bundle of Druault. B. At 4 months, the angle recess has deepened, and the endothelial lining has receded somewhat. There is a small aggregate of differentiating sphincter muscle fibers near the tip of the optic cup. The arrowhead points to Schlemm’s canal. The condensed tissue just posterior to Schlemm’s canal is the developing scleral spur (hollow arrow). The arrow points to the developing “tertiary vitreous” or zonular fibers. They originate from the nonpigmented ciliary epithelium and pass at right angles through Druault’s bundle toward the lens capsule. C. The iris has grown and only its ciliary portion is presented. The angle recess has deepened and is occupied by loose connective tissue separated by many spaces. The dilator muscle of the iris has reached its root, which is still thick. The arrowhead points to the major arterial circle. D. Sphincter muscle is fully developed and is separated by connective tissue septa into several groups of cells. The collarette is represented as a surface stromal bulge containing two blood vessels (curved hollow arrow). E. Schematic diagram of the developing iris dilator muscle at 6 months. During the sixth month, dilator muscle fibers (Dil. M) start to differentiate from extensions of the anterior epithelial cells (AE) into the stroma (ST). These cells are located peripherally to the developing von Michel’s spur (MS), which itself is a pigmented projection of the anterior epithelium, demarcating the posterior limit of the sphincter muscle (SP). In the developing dilator muscle, myofilaments within the elongating processes become arranged parallel to the stromal axis. Some undifferentiated anterior epithelial cells (UN) are present. In the sphincter, which had originated earlier from the same layer of anterior epithelial cells, connective tissue septa and a capillary (CA) start to grow between clumps of cells, but connective tissue has not yet invaded between the muscle cells and the anterior pigment epithelium beneath it. Eventually, the sphincter muscle bundles become completely separated from the anterior epithelium, whereas the dilator muscle sheet remains as the multilayered stromal projection of a part of this epithelium never separating from it. Therefore, the dilator muscle is not a separate cellular layer, but rather a partial myoid differentiation of cellular processes of the anterior neuroectodermal pigment epithelial cells. P, pupillary margin; PC, posterior chamber; PE, posterior epithelium; PM, pupillary membrane.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|