 Premalignant and Malignant Lesions of the Conjunctival Epithelium
Premalignant and Malignant Lesions of the Conjunctival EpitheliumConjunctival Keratotic Plaque and Actinic Keratosis
General Considerations
Several types of benign and malignant lesions can arise from the squamous epithelium of the conjunctiva. These lesions tend to form a spectrum ranging from those that are entirely benign, to those with low malignant potential, to more aggressive, malignant neoplasms. It is often difficult to differentiate clinically between the benign and malignant lesions. The majority of epithelial lesions seen in a clinical practice are low grade proliferations with only a small chance of evolving into frank squamous cell carcinoma. Two of these are keratotic plaque and actinic keratosis (1,2,3,4). Because they may be impossible to differentiate clinically, they are considered together for this discussion. In the authors’ clinical series of 1,643 conjunctival tumors, there were four keratotic plaques and four cases of actinic keratosis each representing less than 1% of the 1,643 cases (2).
Clinical Features
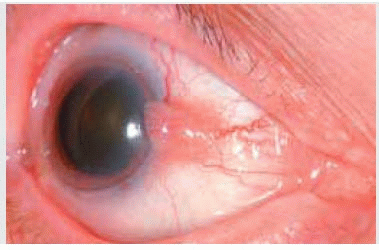
Both keratotic plaque and actinic keratosis can develop on the limbal or bulbar conjunctiva usually in the interpalpebral region. Clinically, the lesion appears as a flat, white plaque that appears gradually and shows no tendency toward aggressive growth.
Pathology
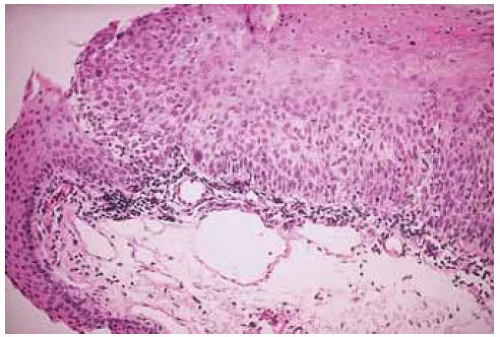
Pathologically, keratotic plaque consists of acanthosis of the epithelium with keratinization of the conjunctival epithelium and parakeratosis. Actinic keratosis (senile keratosis) is a similar proliferation of epithelium with prominent keratosis, often over a chronically inflamed pingueculum or pterygium. It can have a frothy or leukoplakic appearance and may be clinically similar to a keratotic plaque.
Management
Keratotic plaque and actinic keratosis of the conjunctiva are usually indistinguishable clinically from conjunctival intraepithelial neoplasia (CIN), which has slightly greater potential to evolve into invasive squamous cell carcinoma. Therefore, the finding of leukoplakia in the conjunctiva is a relative indication for surgical excision and supplemental cryotherapy. However, it is also acceptable to follow some such lesions until progression is documented, particularly in elderly patients, because the prognosis is generally excellent, even for CIN.
Selected References
1. Shields CL, Demirci H, Karatza E, et al. Clinical survey of 1643 melanocytic and nonmelanocytic tumors of the conjunctiva. Ophthalmology 2004;111: 1747-1754.
2. Spencer WH. Conjunctiva. In: Spencer WH, ed. Ophthalmic Pathology. An Atlas and Textbook. 4th ed. Philadelphia: WB Saunders; 1996:112-113.
3. Volker HE, Naumann GOH. Conjunctiva. In: Naumann GOH, Apple DJ, eds. Pathology of the Eye. New York: Springer-Verlag; 1986:287.
4. Mauriello JA Jr, Napolitano J, McLean I. Actinic keratosis and dysplasia of the conjunctiva: a clinicopathological study of 45 cases. Can J Ophthalmol 1995; 30:312-316.
Conjunctival Keratotic Plaque and Actinic Keratosis
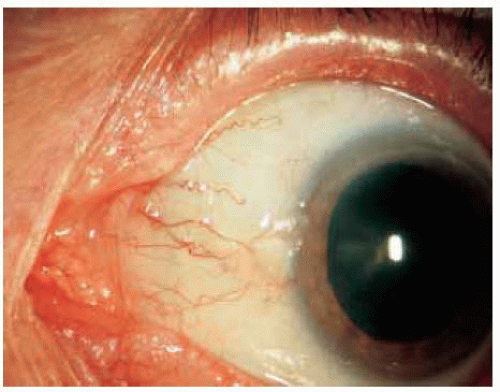
These benign lesions can simulate neoplasia because of the leukoplakia produced by the keratosis.
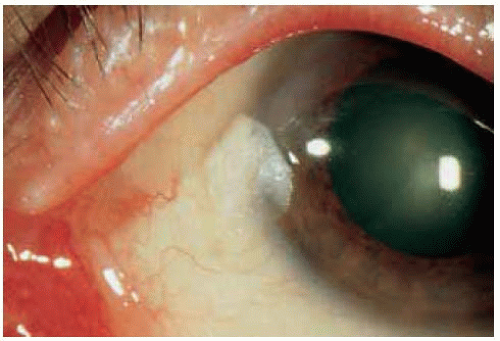 Figure 18.4. Keratosis associated with a pingueculum with slight involvement of the adjacent cornea. |
Conjunctival Intraepithelial Neoplasia
General Considerations
Squamous cell neoplasia can be a localized, minimally aggressive lesion confined to the surface epithelium or a more aggressive tumor that transgresses the basement membrane and invades the adjacent tissues. The former has no potential to metastasize and is called CIN (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28). The latter is called invasive squamous cell carcinoma and generally has a low potential for metastasis. In recent years, some authors have used the umbrella term “ocular surface squamous neoplasia” to include dysplasia, CIN, and invasive squamous cell carcinoma (10). CIN is initially confined to the epithelium but can progress to invasive squamous cell carcinoma; therefore, it is considered a “precancerous” condition, rather than a true malignancy. The main predisposing factors include sunlight and human papillomavirus (HPV). Although results are conflicting, a recent study using polymerase chain reaction established the presence of HPV 16 or 18 in cases of CIN (14).
In the authors’ clinical series of 1,643 conjunctival tumors, there were 71 cases of CIN, accounting for 39% of all premalignant and malignant epithelial lesions and for 4% of all conjunctival lesions (2).
Clinical Features
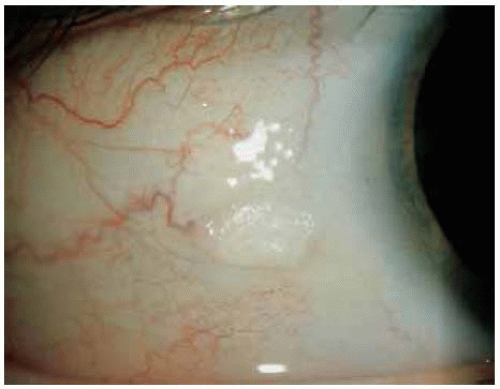
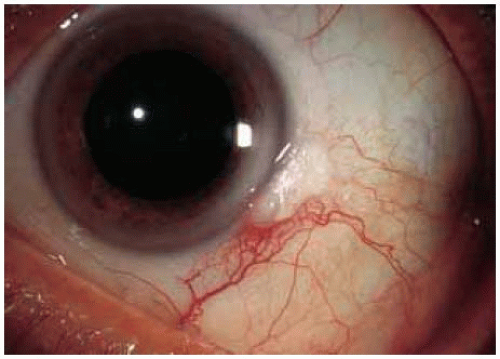
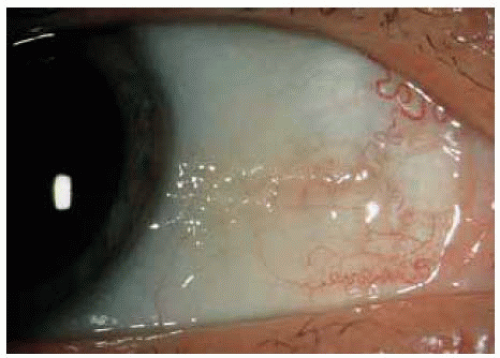
CIN is usually unilateral in fair-skinned, middle-aged or older patients who have had considerable exposure to sunlight (1,2,3,4,11). It can develop in children (6). Both CIN and invasive squamous cell carcinoma occur more frequently in immunosuppressed patients, particularly those with acquired immunodeficiency syndrome (AIDS; 14). Clinically, CIN has several variations, but usually appears as a fleshy, sessile, or minimally elevated lesion usually at the limbus in the interpalpebral fissure and less commonly in the forniceal or palpebral conjunctiva. Secondary inflammation can lead to the misdiagnosis of atypical conjunctivitis before the neoplasm is suspected (12). Leukoplakia is usually absent or minimal; extensive leukoplakia should raise suspicion for invasive squamous cell carcinoma. Although greater thickness is believed to be a sign of malignant transformation we have seen thick tumors that remain within the epithelium. Hence, there are no consistent clinical criteria for distinguishing CIN from invasive squamous cell carcinoma. CIN can extend for a variable distance into the adjacent corneal epithelium, where it appears as a subtle advancing, gray, superficial opacity that may be relatively avascular or may have fine blood vessels (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27). When the involvement is limited to the corneal epithelium with only minimal limbal involvement, it is called “primary corneal dysplasia” (9).
Diagnosis
Although the diagnosis of CIN is made histopathologically, it has sufficiently characteristic features that a diagnosis can be made clinically in most cases. Usually, conjunctival/corneal CIN is small and localized enough that primarily complete excision (discussed later) is the best method of establishing the diagnosis, and incisional biopsy is rarely indicated. However, some have advocated aspiration cytology (13) or impression cytology to establish the diagnosis (28).
Pathology
Histopathologically, mild CIN (dysplasia) is characterized by a partial thickness replacement of the epithelium by mildly anaplastic cells that lack normal maturation. Severe CIN (carcinoma in situ) is characterized by full-thickness replacement of the epithelium by similar cells. The basement membrane is intact (1). Both variants show a characteristic abrupt demarcation between the affected epithelium and the adjacent normal epithelium (1,2). In contrast with invasive carcinoma, hyperkeratosis and dyskeratosis are relatively uncommon findings.
Management
There are many reports that discuss management of CIN (15,16,17,18,19,20,21,22,23,24,25,26,27,28). Complete excision with adequate margins is the preferred initial treatment. For most localized lesions, we remove the growth with alcohol corneal epitheliectomy, partial lamellar sclerokeratoconjunctivectomy, and double freeze thaw cryotherapy (15). This is the same technique used for localized squamous cell carcinoma and melanoma and it is discussed and illustrated in Chapter 25. Cryotherapy is almost always used as supplemental treatment and seems to provide better control (15,16,17); low-dose irradiation with strontium-90 has been used successfully (18). Most recently, we and others have used extensively chemotherapy with topical mitomycin C for recurrent or persistent cases (19,21,22). Others used local interferon-a-2b (24) and 5-fluorouracil (25,26). A recent report described making the diagnosis of conjunctival/corneal CIN by impression cytology and treating with topical mitomycin C and interferon-a-2b in cycles (21). An antiviral drug, cidofovir, has been used with some success (23). Incomplete removal is associated with greater recurrence.
Selected References
1. Spencer WH. Conjunctiva. In: Spencer WH, ed. Ophthalmic Pathology. An Atlas and Textbook. 4th ed. Philadelphia: WB Saunders; 1996:38-125.
2. Shields CL, Demirci H, Karatza E, et al. Clinical survey of 1643 melanocytic and nonmelanocytic tumors of the conjunctiva. Ophthalmology 2004;111: 1747-1754.
3. Shields CL, Shields JA. Overview of tumors of the conjunctiva and cornea. In: Foster CS, Azar DT, Dohlman CL, eds. Smolin and Thoft’s The Cornea. 4th ed. Philadelphia: Lippincott, Williams & Wilkins; 2005:735-755.
4. Shields CL, Shields JA. Tumors of the conjunctiva and cornea. Surv Ophthalmol 2004;49:3-24.
5. Grossniklaus HE, Green WR, Luckenbach M, et al. Conjunctival lesions in adults. A clinical and histopathologic review. Cornea 1987;6:78-116.
6. Linwong M, Herman SJ, Rabb MF. Carcinoma in situ of the corneal limbus in an adolescent girl. Arch Ophthalmol 1972;87:48-51.
7. Dark AJ, Streeten BW. Preinvasive carcinoma of the cornea and conjunctiva. Br J Ophthalmol 1980;64:506-514.
8. Erie JC, Campbell RF, Liesegang TJ. Conjunctival and corneal intraepithelial and invasive neoplasia. Ophthalmology 1986;93:176-183.
9. Waring GO, Roth AM, Ekins MB. Clinical and pathological descriptions of 17 cases of corneal intraepithelial neoplasia. Am J Ophthalmol 1984;97:547-559.
10. Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol 1995;39:429-450.
11. Tunc M, Char DH, Crawford B, et al. Intraepithelial and invasive squamous cell carcinoma of the conjunctiva: analysis of 60 cases. Br J Ophthalmol 1999;83:98-103.
12. Akpek EK, Polcharoen W, Chan R, et al. Ocular surface neoplasia masquerading as chronic blepharoconjunctivitis. Cornea 1999;18:282-288.
13. Grossniklaus HE, Stulting RD, Gansler T, et al. Aspiration cytology of the conjunctival surface. Acta Cytol 2003;47:239-246.
14. Scott IU, Karp CL, Nuovo GJ. Human papillomavirus 16 and 18 expression in conjunctival intraepithelial neoplasia. Ophthalmology 2002;109:542-547.
15. Shields JA, Shields CL, De Potter. Surgical approach to conjunctival tumors. The 1994 Lynn B. McMahan Lecture. Arch Ophthalmol 1997;115:808-815.
16. Fraunfelder FT, Wingfield D. Management of intraepithelial conjunctival tumors and squamous cell carcinomas. Am J Ophthalmol 1983;95:359-363.
17. Divine RD, Anderson RL. Nitrous oxide cryotherapy for intraepithelial epithelioma of the conjunctiva. Arch Ophthalmol 1983;101:782-786.
18. Elkon D, Constable WC. The use of strontium-90 in the treatment of carcinoma in situ of the conjunctiva. Am J Ophthalmol 1979;87:84-86.
19. Frucht-Pery J, Rozenman Y. Mitomycin C therapy for corneal intraepithelial neoplasia. Am J Ophthalmol 1994;117:164-168.
20. Tabin G, Levin S, Snibson G, et al. Later recurrences and necessity for long-term follow-up in corneal and conjunctival intraepithelial neoplasia. Ophthalmology 1997;104:485-492.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree