Fig. 7.1
Number of male genomes detected per 100,000 female genomes in the thyroid of women with Hashimoto’s thyroiditis, nodular goiter, and normal thyroid [9]. Reprinted with permission from Bioscientifica Ltd.
Maternal autoimmune responses may also target the fetus when autoantibodies cross the placenta, as evidenced by transplacental transfer of maternal thyroid autoantibodies to the TSH receptor (TRAbs) in some infants born to mothers with GD (see section “Neonatal Thyrotoxicosis (“Neonatal Graves’ Disease”)”). Typically, neonatal thyrotoxicosis occurs in the first few days of life and can last up to 4 months, by which time the maternal antibodies are cleared from the infant’s circulation [12].
Graves’ Disease in Pregnancy and Postpartum
Hyperthyroidism occurs in 0.1–0.4 % of pregnancies [13]. Autoimmune thyroid dysfunction represents the most common cause of hyperthyroidism in pregnancy [14, 15]. While GD accounts for 80 % of these cases and the majority were present prior to pregnancy, GD can rarely develop de novo during pregnancy [16, 17]. The hyperthyroidism of GD is caused by thyroid stimulating immunoglobulins (TSI) binding and activating the TSH receptor, and inducing excess synthesis and secretion of thyroid hormone [17, 18].
The clinical presentation of GD in pregnancy and postpartum is similar to the general adult population. Classic clinical manifestations include diffuse goiter, anxiety, palpitations, tremor, heat intolerance, and thyroid associated manifestations such as ophthalmopathy and dermopathy. Multiple thyroid autoantibodies target the thyroid epithelial cell TSH receptor. Patients with GD can produce activating (TSI), blocking (TBIIs), and neutral thyroid receptor autoantibodies (TRAbs). The interplay of these autoantibodies during the immune tolerance of pregnancy is complex.
During pregnancy, the differential diagnosis of hyperthyroidism in the majority of cases involves distinguishing between GD and gestational hyperthyroidism, which is limited to the first half of pregnancy. Both entities are characterized by elevated free T4 (FT4) and suppressed serum TSH levels. The presence of serum markers of thyroid autoimmunity (TSI, thyroid autoantibodies) can help distinguish between these two diagnoses.
In the postpartum period, new onset GD should be distinguished from postpartum and subacute thyroiditis. New onset postpartum GD occurs 20-fold less commonly than postpartum thyroiditis [19]. The majority of cases of postpartum GD (95 %) exhibit serum positivity for TRAbs [20]. The presence of ophthalmopathy can also help to distinguish postpartum GD from postpartum thyroiditis.
Although exacerbations of GD early in pregnancy are sometimes observed, the disease most often abates after the first trimester. TRAbs are present in over 95 % of patients with active GD and high titers may remain following radioiodine treatment [21]. Pregnancy is associated with a decrease in thyroid autoantibody titers [14, 22] with TRAbs appearing to reach their lowest levels during the third trimester (Fig. 7.2) [23, 24]. A number of studies have examined changes in TRAbs [23–25] and other antithyroid antibodies [26] during pregnancy. Kamijo studied changes in TRAb levels during pregnancy, using four different methods of detection [24]. The author was able to demonstrate a significant decrease in TRAb titers as pregnancy progressed [24]. Similarly, Gonzalez-Jimenez et al. reported that TRAb levels decreased significantly (p < 0.01) during pregnancy in active GD patients [23]. However Tamaki et al. demonstrated that TRAb levels do not always decline during pregnancy in active GD [25]. The immune tolerance of pregnancy is believed to play a role in the decrease of thyroid autoantibody titers, including anti-peroxidase antibodies (anti-TPOAb), thyroglobulin autoantibodies (Anti-TgAb), and TRAbs. The decline of autoantibody levels is anticipated to begin by approximately 20 weeks of gestation. Persistence of maternal TRAbs is important as they have the potential to impact the fetal and neonatal thyroidal status. As noted above, maternal antithyroid antibodies may cross the placenta, particularly when they remain at high levels after week 25. The transfer of stimulatory or inhibitory TRAbs to the fetus may lead to a transient fetal and neonatal hyper- or hypothyroidism.
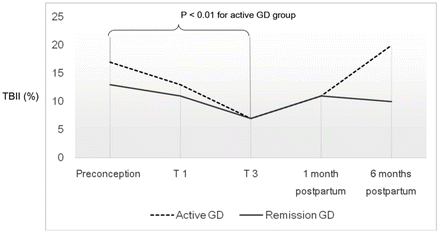

Fig. 7.2
TBII levels nadir in the third trimester of pregnancy. Preconception, first trimester (T1), third trimester (T3), early (1 month) and late (6 months) postpartum levels in pregnant women with active-GD and remission-GD are shown. There was a statistically significant decrease in mean levels from preconception (17 %) to third trimester (7 %) in the active GD group (p < 0.01). Adapted from [23]
The American Thyroid Association recommends a determination of serum TRAbs by 24–28 weeks gestation in the following cases:
Pregnant women with active hyperthyroidism
Previous history of radioiodine treatment
Previous history of delivering an infant with hyperthyroidism
Total thyroidectomy treatment of hyperthyroidism in pregnancy
This recommendation intends to identify pregnancies at risk of maternal–fetal transfer of antithyroid autoantibodies. A TRAb value over three times the upper limit of normal is an indication for close monitoring of the fetus. High TRAb titers between 22 and 26 weeks of gestation represent a risk factor for fetal or neonatal hyperthyroidism [21].
Thyroid Eye Disease in Pregnancy
TED is responsible for approximately 50 % of orbital disorders in the general population [27], and is characterized by inflammation, edema, secondary fibrosis, and extra-ocular muscle involvement. This classic presentation is seen in 70 % of TED patients [28]. The development of TED during pregnancy is extremely rare. Germain et al. reported a case of a 23-year-old euthyroid primigravida at 30 weeks of gestation who developed severe sight-threatening TED. Functionally active TRAbs were demonstrated using a cyclic adenosine monophosphate (cAMP) biological assay [29].
The rare occurrence of worsening TED and the rare development of new onset TED in pregnancy is attributed to the immune tolerant state and decreasing TRAb levels [2].
Studies of TED During Pregnancy and Postpartum
TED progression in pregnancy is rare and to our knowledge, there are no published studies of TED during pregnancy and the postpartum period. Only a very limited number of cases have been reported [29–32]. The severity of TED in the adult ranges from mild to severe, with only 5 % considered severe [33]. Data on prevalence or severity of TED in pregnancy are not available. The peak female age distribution of TED is from 40s to 60s, representing the post-reproductive age [34]. One case report of severe TED in pregnancy described a 22-year-old primigravida, who presented at 30 weeks of gestation with progressive worsening of vision and swollen eyes over a period of 2 months. The patient had been diagnosed with GD 3 years prior and had been treated with radioiodine and rendered euthyroid after the treatment. Physical examination revealed marked bilateral periorbital edema, exophthalmos, limitation of left abduction, decreased visual acuity, and dyschromatopsia bilaterally. Her thyroid function tests were within normal range and TSI = 434 % (normal < 130 %) were detected. On brain CT, bilateral optic nerve compression was suspected. The patient was treated with high-dose steroids, methylprednisolone 300 mg twice daily, without significant improvement. The concern for permanent nerve damage secondary to optic nerve compression prompted surgical orbital wall decompression within 3 days of admission. Steroid therapy was continued and tapered postoperatively. In follow-up, the patient demonstrated improvement of visual acuity, proptosis, lid lag, as well as color discrimination [30].
Differential Diagnosis of Ophthalmopathy in Pregnancy
TED is by far the most common cause of extraocular muscle enlargement in adults [27]. As in any adult, other causes of ophthalmopathy should be considered and excluded before diagnosing TED in pregnancy (Table 7.1). The differential diagnosis of TED in pregnancy includes myasthenia gravis (MG), retro-ocular tumors, carotid-cavernous fistula, and other inflammatory ophthalmopathies.
Table 7.1
Differential diagnosis of TED in pregnancy
Diagnosis | Clinical manifestations |
|---|---|
TED | Usually bilateral and associated with hyperthyroidism MRI: Orbital fat and extraocular muscle involvement with tendon sparing |
Ocular Myasthenia Gravis | Weakness usually involves one or more ocular muscles without overt pupillary abnormality. Ptosis which shifts from one eye to the other is virtually pathognomonic of Myasthenia Gravis |
Orbital inflammation of muscle (orbital myositis or orbital pseudotumor) | Clinical diagnosis is made by exclusion of other conditions. Unilateral disease is very common but bilateral presentations are not uncommon, especially in children. Symptoms usually acute in onset with proptosis and inflammatory sign such as pain, swelling, and erythema |
CT scan: Moderately enhanced focal or diffuse mass frequently accompanied by infiltration of orbital fat | |
MRI: Proptosis, optic nerve thickening, uveal-scleral thickening, lacrimal gland infiltration, and extraocular muscle involvement with muscle tendon and sheath enlargement can differentiate this diagnosis from TED | |
Carotid-cavernous fistula | History: Preceding trauma and surgical manipulations, recent childbirth or pregnancy. The onset is rapid with pulsating exophthalmos, ocular bruit, diplopia, headache, conjunctival chemosis, increased intraocular pressure, and a reduction in vision |
CT scan: Enlargement of the superior ophthalmic vein and to a lesser extent, enlargement of extraocular muscles | |
Orbital lymphoid tumor | Orbit, lacrimal glands, eyelids, or conjunctiva can be affected. Most often presents as a (1) nontender, firm, subcutaneous mass in the anterior orbit; (2) conjunctival salmon-patch infiltrate; or (3) eyelid infiltration |
Orbital cellulitis | Should be suspected in any pregnant patient with a preceding history of upper respiratory tract infection, trauma, dental infection, or dental procedure. Commonly presents with fever, malaise, and pain with eye movement. CT scan: preseptal cellulitis appears as an area of hyper-attenuation with swelling of anterior orbital tissues and obliteration of adjacent fat planes. MRI: hypointense on T1-weighted, hyperintense on T2-weighted images |
MG is important to consider in the differential because of the overlapping patient demographics and clinical findings with TED. Women are affected by MG nearly three times more often than men before age 40, but the incidence is higher in males after age 50, and roughly equal in men and women during puberty [35]. MG usually affects women in their third decade of life. Pregnancy is considered a precipitating factor for MG. Although the disease course is variable, pregnant patients face risk of exacerbation, respiratory failure, adverse drug response, crisis, and death. A study by Batocchi et al. reported that MG worsened in 10 (19 %) of 54 patients followed during pregnancy. Approximately 60 % of exacerbations occurred during the first trimester, and approximately 28 % of patients deteriorated immediately after delivery [36]. Thyroid disorders also represent an exacerbating factor for MG. The clinical presentation of MG associated with ATD is frequently limited to double vision [37]. Diplopia, intermittent ptosis, and incomplete eyelid closure are the most common presenting symptoms occurring in approximately 85 % of patients with MG [38]. The symptoms are a result of the involvement of extraocular muscles, levator, and orbicularis oculi in MG. Symptoms can fluctuate in severity, worsening with exertion, and improving with rest. Fatigue upon exertion is essential to making the diagnosis and differentiating MG from TED. Gaze-evoked nystagmus and Cogan’s lid twitch represent clinical signs characteristic of MG. Ptosis and/or exotropia are rarely seen in TED. If ptosis, exotropia, or weakness of the orbicularis oculi muscle develops in a patient with TED, coincidence of MG should be considered.
Due to the trophic factors elaborated in pregnancy, orbital tumors such as lymphoid tumors, cavernous hemangiomas, and meningiomas can proliferate and become manifest during pregnancy. Any of these can present like TED.
Carotid-cavernous fistula is an abnormal communication between the internal and external carotid arteries in the cavernous sinus and represents a rare cause of exophthalmos in the adult, which can also manifest in pregnancy. Pertinent histories include recent trauma, surgical manipulations as well as pregnancy or recent childbirth. Patients could present with increasing proptosis, conjunctival chemosis, ocular pulsation, and progressive vision loss [27].
Orbital cellulitis can also masquerade as TED during pregnancy. Patients may present with proptosis, double vision, fever, malaise, and pain with eye movement. Orbital cellulitis should be suspected in any pregnant patient with a preceding history of upper respiratory tract infection, trauma, dental infection, or dental procedure. A thorough history and physical examination are crucial in establishing the diagnosis [27].
Therapeutic Options for TED During Pregnancy
The majority of GD patients exhibit mild and nonprogressive ocular involvement that tends to improve spontaneously [39]. Therefore, most TED pregnant patients would be expected to require only local measures for symptomatic relief.
For the rare case of severe TED that does not improve during pregnancy, more aggressive medical, and possibly surgical, therapy may be indicated. As noted elsewhere, the severity and activity of TED are not synonymous, but both are important in the assessment of TED patients including those who are pregnant. Similar to the nonpregnant adult, patients with active eye disease during pregnancy are expected to respond to medical treatment, whereas surgical treatment is indicated for the rare case of unresponsive, sight-threatening TED.
It is necessary to identify patients with sight-threatening TED, including compressive optic neuropathy (CON), corneal breakdown, or occasionally, globe subluxation [40, 41]. This group of patients must be treated urgently and aggressively [34, 42], and treatment should not be delayed on account of pregnancy. The optimal initial treatment for CON is high-dose systemic steroids (see medical management below). Suboptimal response to steroids is an indication for surgical decompression.
The Therapeutic Interventions for TED During Pregnancy Include
Local Measures
Eye shades/sunglasses help alleviate photophobia and shield the eyes from dust and wind. Saline artificial tears can alleviate foreign body sensation, dry eyes, and the gritty sensation. Raising the head of the bed at night may be of some help to reduce periorbital edema in addition to diuretics. Elimination of controllable risk factors for TED progression such as smoking is critical, not only for the benefit of TED but also for the pregnancy. Nocturnal taping of the eyes can be helpful when lagophthalmos is present and prisms can be used to improve cases of mild diplopia. Understanding the natural history of the disease and providing reassurance is one of the most important therapeutic measures since the chance of progression of TED to more severe forms is very low [40]. All the above supportive measures can be safely implemented during pregnancy. Diuretic use to decrease swelling is debatable for routine treatment of TED.
Medical Therapy
Both hyper- and hypothyroidism can influence the course of TED. Patients with uncontrolled thyroid hormone levels are more likely to exhibit severe TED than euthyroid patients. Anti-thyroid drugs (anti-TDs) do not appear to have a negative influence on the course of TED. Medical treatment is usually followed by a fall in serum antibody concentrations, suggesting a waning of autoimmunity [43]. Since obstetrical and medical complications are directly related to thyroid status during pregnancy, patients should be euthyroid before conceiving. Anti-TDs are the mainstay of treatment during pregnancy. The American Thyroid Association recommends propylthiouracil (PTU) for the first trimester, due to the risk of methimazole (MMI) embryopathy [44]. Following the first trimester, patients on PTU should be switched to MMI in order to decrease the incidence of hepatotoxicity related to PTU. Thyroid function tests should be monitored monthly during pregnancy [15, 21].
Glucocorticoid therapy has been shown to reduce interferon gamma-induced HLA-DR expression in vitro [43–46]. Current recommendations for the treatment of acute and moderate-to-severe TED in adults is a course of 0.5 g of methylprednisone IV once weekly for 6 weeks, followed by 0.25 g/week for 6 weeks. If the patient fails to respond, treatment may be stopped after 6 weeks of 0.5 g/week dosing [47]. Currently there are no specific dosing recommendations for pregnancy. Glucocorticoids, however, have been widely used during pregnancy for the treatment of a variety of medical conditions. They are categorized as a class C pregnancy risk and should be given only if the potential benefits justify the potential risk to the fetus. Various animal studies showed that high doses of glucocorticoid can lead to an increased incidence of cleft palate in exposed offspring [48, 49]. However, several subsequent reports in which pregnant women were treated with glucocorticoids throughout the first trimester for various medical conditions did not reveal a consistent pattern of embryopathy [50]. Glucocorticoids do not appear to have major teratogenic potential [51]. Glucocorticoids during pregnancy, however, may increase the risk of premature rupture of the membranes and intrauterine growth restriction [52, 53]. Antenatal exposure to a relatively large dose of synthetic glucocorticoids is associated with suppression of the hypothalamic-pituitary-adrenal axis [54]. Other maternal adverse effects include pregnancy-induced hypertension, gestational diabetes, osteoporosis, and infection [51]. Therefore, when systemic glucocorticoids are needed in pregnancy, it is generally recommended to use the lowest effective dose for the shortest duration of time, avoiding high doses during the first trimester [55]. Glucocorticoids are metabolized in the placenta by a crucial enzyme, 11-beta-hydroxysteroid dehydrogenase-2. Prednisolone-related drugs (methylprednisone, prednisolone, and prednisone) are mostly metabolized to inactive forms by this enzyme and thus only approximately 10 % of the total dose compared to 33 % of betamethasone and 50 % of dexamethasone will enter the fetal circulation [56].
Prednisone and prednisolone have been measured in breast milk. Levels of prednisolone, the active form of prednisone measured in the milk, are typically less than 0.1 % of the total prednisone dose, even for a dose up to 80 mg. This corresponds to less than 10 % of an infant’s endogenous cortisol production [57] and therefore does not pose a clinically significant risk to a nursing infant. Nonetheless, exposure may be minimized if nursing is performed 3–4 h after a dose [57]. The American Academy of Pediatrics considers steroid therapy compatible with breastfeeding [57]. Guidelines from the National Heart, Lung and Blood Institute for the management of asthma also note that maternal use of systemic glucocorticoids is not a contraindication to breastfeeding [58].
Other Alternative Medical Treatments
In nonpregnant women, some studies have found that selenium may lower anti-TPOAb titers. The European Group on Graves’ Orbitopathy (EUGOGO) consortium conducted a trial to compare selenium use with placebo or pentoxifylline. Several ocular parameters (eye involvement (P = 0.01), progression of TED (P = 0.01) as well as quality of life outcome measures (P < 0.001)) improved in the selenium-treated group compared to placebo without significant side effects (see Chap. 6 for a full discussion) [59]. The selenium levels of the study population were marginally low and this may have biased in favor of a positive selenium treatment response [59]. Although selenium levels can be low in full-term pregnant women compared with nonpregnant women, the risk to benefit comparison does not support routine selenium supplementation during pregnancy [21]. Currently, there is an ongoing study on selenium supplementation in pregnancy where secondary outcomes will examine maternal and fetal risks.
Somatostatin Analogs
Four large double-blind, placebo-controlled studies of somatostatin analogs have shown no significant effects on TED compared with placebo [60–63]. Somatostatin analogs are listed as a category B pregnancy drug. Since somatostatin analogs cross the placenta, potential adverse fetal outcomes must be considered. Several papers reported the safe and effective use of these analogs in pregnancy for various therapeutic indications [64–66].
Rituximab: Rituximab has been assigned a pregnancy category C by the FDA. Because human IgG is known to traverse the placenta, rituximab may potentially cause fetal B-cell depletion. Post-marketing data indicate that B-cell lymphocytopenia generally lasts less than 6 months in infants exposed to the medication in utero. It is only recommended for use in pregnant women when the drug is clearly necessary. Rituximab use during breastfeeding is also not recommended since the drug is excreted in breast milk.
Azathioprine: Azathioprine is a pregnancy category D drug. Azathioprine should not be given during pregnancy and breastfeeding.
In summary, rituximab and azathioprine are contraindicated in pregnancy for the treatment of TED.
Orbital Radiotherapy
Surgical Therapy
Orbital decompression can be safely performed during pregnancy for severe CON unresponsive to a short course of systemic glucocorticoids or if sight is threatened by corneal exposure. If ophthalmopathy is severe but inactive, orbital decompression may also be performed. Reducing proptosis and decompressing the optic nerve can be achieved by a variety of decompression techniques (see Chaps. 11 and 12). One of the most feared complications of orbital decompression is postoperative diplopia. Extraocular muscle or eyelid surgery is often needed for full rehabilitation but can be delayed until after delivery.
Although orbital decompression is sometimes performed as first-line treatment for CON, most patients will still require glucocorticoid therapy; whereas less than half of patients treated with glucocorticoids initially will require orbital decompression [67]. Although it is preferable to operate on a pregnant patient during the second trimester [68, 69], emergencies require intervention regardless of gestational age.
Thyroidectomy
Whether or not thyroidectomy affects the course of TED remains unsettled. In many studies [41, 70, 71], different thyroid surgeries (subtotal versus total thyroidectomy) seem to carry very low risk for progression of TED [40]. Thyroidectomy is not recommended as first-line therapy for hyperthyroid GD, regardless of pregnancy status, but it should be considered in the event of contraindications to anti-TD therapy [21]. If thyroidectomy is indicated during pregnancy, the optimal time to perform surgery is in the second trimester [21].
With regard to the need for oculoplastic referral, it is the authors’ opinion that because of the rare occurrence of TED during pregnancy and the limited therapeutic options, there should be a very low threshold for referral to oculoplastic specialists.
Summary
TED during pregnancy is a relatively uncommon occurrence in clinical practice. The prevalence of moderate-to-severe TED during pregnancy should decrease with treatment of preexisting TED prior to conception. Studies are needed to better understand the natural course of TED during pregnancy as well as the short- and long-term consequences of the differing therapeutic options. An endocrinologist, ophthalmologist, and obstetrician should be members of a multidisciplinary patient care team.
Pediatric Thyroid Eye Disease
Neonatal Thyrotoxicosis (“Neonatal Graves’ Disease”)
Neonatal thyrotoxicosis (referred to as “neonatal Graves’ disease” in the pediatric literature) is uncommon, comprising no more than 1 % of childhood cases of hyperthyroidism, and presenting in less than 2 % of infants born to mothers with a history of GD [72]. Most cases of neonatal thyrotoxicosis result from transplacental passage of maternal thyroid-stimulating immunoglobulins in utero. Even more rarely, there have been case reports of neonatal thyrotoxicosis due to activating mutations of the TSH receptor (TSHR) [73]. According to Wiersinga et al., true clinically significant TED has never been observed in neonatal thyrotoxicosis [74]. However, a prominent stare may be present (Fig. 7.3), providing a clinical clue to neonatal thyrotoxicosis. A maternal history of GD is often not elicited if the mother is being treated for hypothyroidism after radioactive iodine (RAI) ablation. The characteristic stare may prompt the clinician to inquire further into the maternal history, observe the infant for additional clinical signs such as tachycardia and poor feeding, and perform a laboratory evaluation.
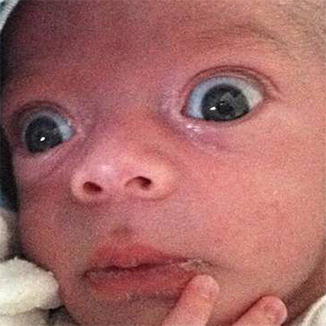

Fig. 7.3
Characteristic stare of an infant with neonatal thyrotoxicosis. The infant was born to a mother with a history of Graves’ disease, treated with radioactive iodine a decade earlier. Tachypnea, tachycardia, poor feeding, and the ocular appearance of the infant prompted evaluation for neonatal thyrotoxicosis (Figure courtesy of Moran Gotesman, MD)
Pediatric Graves’ Disease
In children, the signs and symptoms of GD may develop gradually, often leading to a delayed diagnosis. Nonspecific clinical features such as difficulty concentrating and faltering academic achievement may be attributed to other conditions such as attention deficit hyperactivity disorder (ADHD). In children, long-term treatment with anti-TDs represents first-line therapy. Most children who are treated appropriately do not suffer long-term sequelae. However, delayed diagnosis has been associated with abnormal neurodevelopment (hyperactivity, psychomotor delay, and mental retardation) and altered skeletal maturation, including craniosynostosis and advanced bone age in younger children [75, 76].
Epidemiology of Pediatric Graves’ Disease
The frequency of thyrotoxicosis ranges from 0.1 in 100,000 in young children (i.e., prior to adolescence) to 3.0 in 100,000 in adolescents [7]. Although GD represents the most common cause of thyrotoxicosis, it only affects 0.02 % of all children [78]. GD comprises 10–15 % of thyroid disorders in patients under 18 years of age, is rare under the age of 5, has a peak incidence at 10–15 years of age, and more commonly affects female pediatric patients [75]. Mild signs and symptoms of TED, including lid retraction and lid lag, are common (25–60 %) in pediatric thyrotoxicosis [78]. However, severe TED is rarely seen in pediatric patients. Bartley et al. studied subjects in Olmsted County, Minnesota, and found that only 6/120 cases of TED occurred in patients under the age of 20 years [79]. The reported incidence rate of TED in male children is 0 per 100,000 for ages 5–9 and 15–19 years, and 1.7 per 100,000 for ages 10–14 years. The reported incidence rate of TED in female children is 3.5 per 100,000 for ages 5–9 years, 1.8 per 100,000 for ages 10–14 years, and 3.3 per 100,000 for ages 15–19 years [74, 79].
Clinical Features of Pediatric Graves’ Disease
Pediatric GD may present with many of the same signs and symptoms as in adults, including heat intolerance (61 %), tremor (49 %), and ophthalmopathy (43 %) [80]. Additionally, children with GD may often present with decreased academic performance (50 %) [80]. Favorable predictors of long-term remission include age less than 12 years old, free thyroxine (FT4) level <35–50 pmol/L at diagnosis, initial response to therapy, TSH receptor antibody levels ≤4 times the upper limit of normal, Caucasian ethnicity, and having other autoimmune condition(s) [81, 82].
Thyrotoxicosis has deleterious physiologic effects on children, including low bone mineral density and increased bone turnover, advancement of bone age leading to a lower than predicted final height, diminished left ventricular reserve, thyrotoxic ophthalmologic signs, and cognitive/neuropsychiatric deficits (hyperactivity, irritability, anxiety, and impaired school attention). Correction of the thyrotoxicosis in a timely manner usually reverses the adverse physiologic effects of thyrotoxicosis. Thus, in most children there do not seem to be long-term sequelae as long as the hyperthyroidism is corrected without delay [83].
Treatment of Pediatric Graves’ Disease
In adult GD, the standard of care is a 12–18-month trial of anti-TDs followed by definitive therapy (i.e., RAI or thyroidectomy) if remission is not achieved. In contrast, pediatric GD is commonly treated for years with anti-TDs – typically methimazole – as long as the patient tolerates the therapy and euthyroidism can be achieved. Propylthiouracil (PTU) is avoided in the treatment of hyperthyroidism in children due to reports of PTU-induced liver failure (also discussed in chapter 1) [54]. The preference for medical management arises from the theoretical fear of radioiodine-induced thyroid malignancy, as well as the fear of potential morbidity and mortality associated with thyroidectomy [75, 80, 84]. After discontinuation of anti-TD therapy, over 50 % of pediatric patients will remain in remission for at least 2 years; however, relapse rates remain high and on the order of 36–47 % [75, 85–88]. Sustained disease remission for over 2 years post anti-TD therapy is less common in children (15–30 %) compared to adults (40–60 %), and may take up to 10 years to achieve [75, 83, 84, 88]. Gruniero-Papendieck et al. reported that in children who did not receive definitive treatment, the rate of persistent GD at 10 years was 31 % [77]. Up to 28 % of pediatric patients on anti-TD therapy experience adverse effects, and severe complications such as agranulocytosis, aplastic anemia, hepatotoxicity, and mortality have been reported, for PTU in particular [83].
Thyroidectomy and radioiodine represent definitive treatment options. Sherman et al. retrospectively investigated 78 patients under 18 years of age who underwent thyroidectomy for GD. The surgical indication for more than half the patients was failed medical therapy (60 %). Other indications included intolerance to anti-TDs (9 %), severe thyrotoxicosis, severe TED (concern for worsening with RAI), significant thyromegaly (gland >30 g), suspicious/malignant thyroid nodule, and unwillingness to receive RAI. Postoperative morbidity was transient and consisted of hypocalcemia (96 %) and unilateral recurrent laryngeal nerve palsy (1 %). It should be noted that the surgery was performed by experienced thyroid surgeons in a high-volume center [80]. Additional indications leading to thyroidectomy over RAI included age less than 10 years (due to theoretical higher risk of thyroid malignancy with RAI in this age group) and consideration of pregnancy in late adolescent females (TRAb levels can transiently increase and persist for at least 5 years after ablation, thus increasing risk of fetal/neonatal GD in the offspring) [75, 89–91].
Although radiation-induced thyroid malignancy as evidenced by the Chernobyl nuclear accident is a concern, retrospective studies suggest a very low risk of malignancy associated with RAI therapy. Patients with GD who are over 10 years old can be treated with RAI with the consultation of a pediatric ophthalmologist prior to ablation [75, 91]. Published retrospective studies with up to 36 years of follow-up have not supported the concerns of increased thyroid malignancy post RAI in the pediatric population [90–92]. In a retrospective review of the medical records of 78 pediatric patients who underwent thyroidectomy at Mayo Clinic, 4 patients (5 %) were incidentally found to have thyroid malignancies on histologic examination [80]. Thus, pediatric patients may harbor occult thyroid malignancy irrespective of RAI therapy.
TED in Children
Definition of TED
The definition of TED with respect to adults has been reviewed elsewhere in this textbook. In children, Antoniazzi et al. defined TED as clinical signs of ocular involvement in patients with GD (lid retraction, lid lag, or stare), who also have exophthalmos or signs of enlarged extraocular muscles on orbital MRI [93].
Epidemiology, Risk Factors, and Pathogenesis
In a 2004 review by Wiersinga, 23 % of pediatric patients with GD were found to have TED, which is very similar to that seen in adult patients with GD (18 %) [74]. However, TED prevalence rates up to 39 % have been reported in childhood GD [94]. A few small studies have attempted to further characterize the epidemiology of pediatric TED and to identify risk factors for the development of clinically significant TED. Durairaj et al. found that 10.0 % (2/20) of pediatric patients with TED had a family history of TED, and 60.0 % (12/20) had a family history of thyroid dysfunction [95]. Young, however, did not find any specific HLA Ag to be prevalent in patients with TED versus patients without TED [96]. Thus, currently there are no reliable genetic markers for identifying which pediatric patients with GD are at increased risk of developing TED.
According to Gogakos and coauthors, although severe TED is less common in children than adults, the epidemiology overall is similar [102]. Furthermore, TED has a similar female-to-male predominance in both children and adults (87 % and 83 %, respectively), and is associated with more severe thyrotoxicosis at the time of diagnosis in all ages. There is often a 2-year lag between the onset of thyroid dysfunction and TED, and a notable family history of thyroid disease in both children and adults. In late adolescence, the clinical features of TED resemble those of the adults [74, 97]. A 2005 European survey of 23 pediatric and 44 adult endocrinologists suggested that smoking was a potential risk factor for the development of TED, as countries with higher smoking prevalence had a higher prevalence of TED among children and adolescents with GD [84]. Although it is not possible to draw conclusions about causality from a survey study, in adults, smoking is an established risk factor for TED (see Chap. 5). Wiersinga postulates that the lower frequency of smoking in children may at least in part explain the tendency for milder clinical manifestations in pediatric TED [74].
Two studies have examined the severity of TED in a total of 163 prepubertal versus postpubertal children and adolescents with GD. Holt et al. did not find evidence of restrictive strabismus in prepubertal subjects. Severe TED seemed to occur less frequently in prepubertal subjects, but the number of subjects in that group was too small to achieve statistical significance (total N = 41; subjects with TED N = 6) [98]. In their prospective study of pediatric patients with GD followed by orbital MRI, Antoniazzi et al. reported a better prognosis in subjects who were prepubertal at the time of diagnosis. The authors hypothesized that increasing orbital volume with advancing age and puberty serves as a physiologic decompression [93].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


