(1)
Department of Ophthalmology and Visual Sciences, University of Iowa Hospitals and Clinics, Iowa City, IA, USA
The posterior ciliary arteries (PCAs) supply the choroid and the optic nerve head. As discussed in Chap. 4, information on the vascular pattern of the choroid and of the PCAs was previously drawn entirely from postmortem cast studies, ever since the first description by Frederik Ruysch [1] in 1700. Extensive anatomical studies of the choroidal vascular bed have been conducted, mostly by studying casts prepared by the postmortem injection of a variety of materials [2–8] and also by studying casts by scanning electron microscopy [9–12]. These studies formed the basis of the classical anatomical textbook description of the choroidal vasculature. According to most of these descriptions: (1) PCAs have no segmental distribution, (2) they anastomose freely with one another as well as with the anterior ciliary arteries, (3) there are inter-arterial and arteriovenous anastomoses in the choroid, and (4) the choriocapillaris forms a freely communicating and an uninterrupted vascular bed in the entire choroid [8, 11]. From these postmortem anatomical descriptions, it was generally concluded that occlusion of a PCA or one of its branches should not produce an ischemic lesion.
Experimental a PCA Occlusion Studies
After the advent of fluorescein fundus angiography in the 1960s, in 1970 I decided to investigate PCAs experimentally in rhesus monkeys (since the vascular pattern in rhesus monkeys is similar to that in the human) by fluorescein angiography. This included in vivo circulation and distribution pattern of the PCAs and their branches and also the types of ischemic lesions produced by cutting various PCAs and their branches. I proceeded by cutting one or another or all the PCAs at a time in 113 monkey eyes [13–17], one or the other short PCA in 87 eyes [18], and a long PCA in 22 eyes [19]. That had never been done before and provided wholly new information.
In Vivo Circulation and Distribution Pattern of the PCAs and Their Branches
As discussed at length in Chap. 4, my in vivo fluorescein angiographic studies [13, 16, 18, 19] on the PCAs and their various branches provided totally new information. This included information about (1) the in vivo circulation and distribution pattern of the PCAs and their branches, right down to the terminal choroidal arterioles, and the choriocapillaris. (2) They showed for the first time that the PCAs and their branches, right down to the terminal choroidal arterioles, and also the choriocapillaris have a segmental distribution in the choroid and that the PCAs and choroidal arteries function as end arteries. These findings have tremendous clinical importance in explaining various choroidal lesions. Thus, these fluorescein angiographic studies showed that the postmortem injection cast studies had misled us greatly for centuries.
Choroidal Ischemic Lesions Produced by Occlusion of the PCAs
I investigated this by experimental occlusion of the PCAs in rhesus monkeys [14]. This was the first comprehensive study of the choroidal ischemic lesions produced by PCA occlusion in large number of eyes. Based on this study, I feel it is essential to describe the nature of these lesions and their evolution with time in detail, because there is little information on this subject elsewhere in the ophthalmic literature, and that has resulted in frequent misdiagnoses and mismanagement of these lesions. Following is a detailed account of the ophthalmoscopic, fluorescein fundus angiographic and histopathological findings of the choroidal ischemic lesions and their evolution, from my study.
Ophthalmoscopic Findings
About 1 h after occlusion of the PCA, ophthalmoscopic examination of the fundus showed no detectable lesions. When the eyes were examined 1–2 days after that, white patches were seen in the part of the fundus supplied by the occluded artery (Fig. 18.1a–c). These patches assumed a large variety of shapes and sizes. Their size varied from a tiny spot to a large area of the fundus. They were irregular in shape, many being elongated with pointed ends, and frequently tended to be triangular and sectoral (Figs. 18.2a, 18.3a, and 18.4). These patches were separated from one another by narrow strips of normal colored fundus and sometimes by the big visible choroidal vessels. The white patches were infarcts, lying deep to the retina.
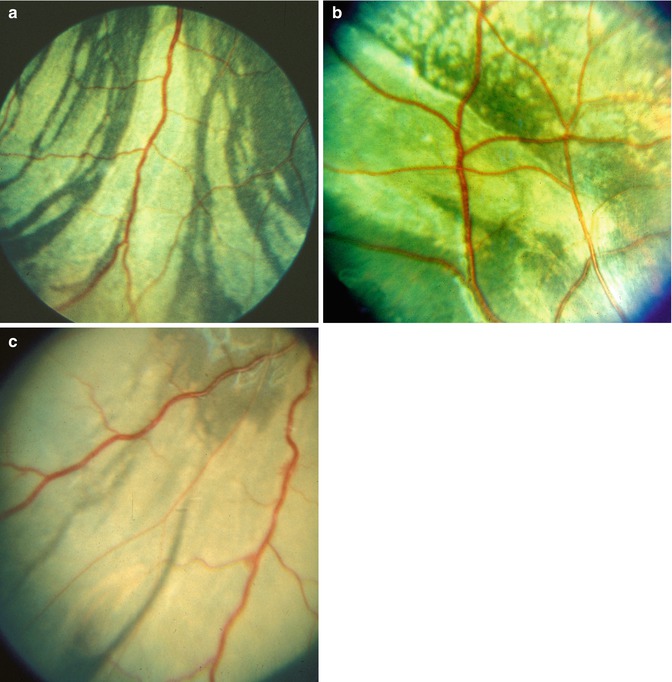
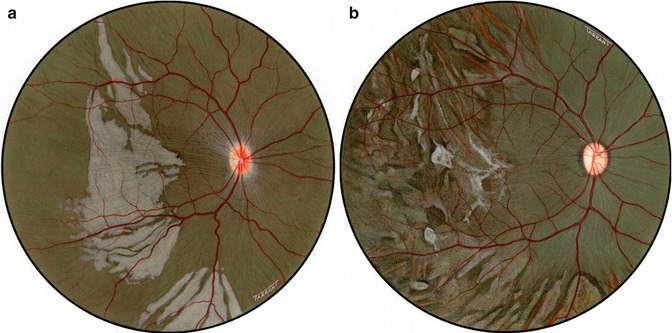
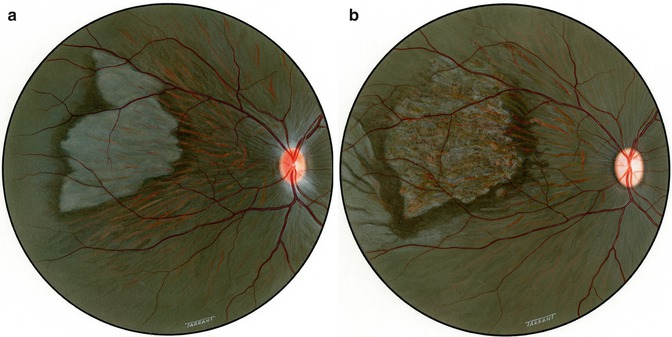
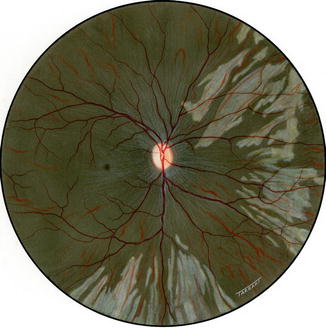

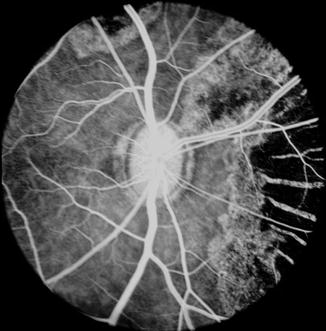
Fig. 18.1
Fundus photograph shows white ischemic choroidal lesions in rhesus monkey eyes. (a) In inferotemporal region 2 days after lateral PCA occlusion (Reproduced from Hayreh and Baines [14]). (b) In superotemporal region 1 day after occlusion of all the PCAs (Reproduced from Hayreh and Baines [14]). (c) In inferotemporal region 2 days after occlusion of all PCAs

Fig. 18.2
Fundus drawings of the right eye show lesions after lateral PCA occlusion in rhesus monkey after (a) 5 and (b) 90 days of occlusion

Fig. 18.3
Fundus drawings of the left eye of rhesus monkey show lesions after occlusion of medial PCA in rhesus monkey – (a) during the acute phase and (b) after resolution of acute phase

Fig. 18.4
Fundus drawing of the right eye of rhesus monkey shows lesions after medial PCA occlusion during the acute phase
On follow-up, these lesions did not increase significantly in size. However, they became more clearly defined over the following few days. At the beginning of the second week, they began to resolve. They became less white and assumed a granular appearance, becoming less dense and gray. After 2–3 weeks, these lesions appeared as granular, grayish, depigmented scars in the fundus (Fig. 18.5a). With the passage of time, some of the patches tended to become ill defined.
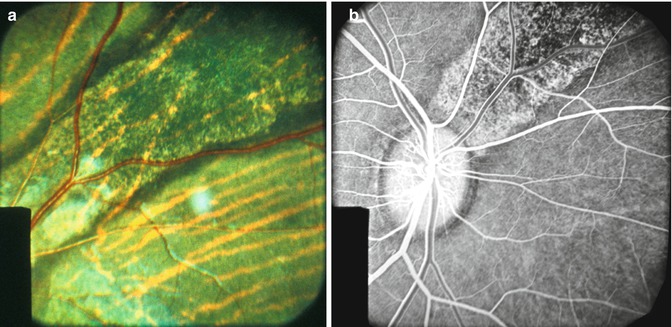

Fig. 18.5
Fundus photograph (a) and fluorescein fundus angiogram during the retinal arterial phase (b) of the right eye of rhesus monkey show lesions situated in superior nasal region, after occlusion of all the PCAs (Reproduced from Hayreh and Baines [14])
Another interesting change appeared toward the end of the second week. The gray lesions became surrounded by a narrow zone, like a dark-colored halo, which was darker than the surrounding fundus (Figs. 18.2b, 18.3b, 18.5a, and 18.6). These patches became more marked with time and their size increased, extending to involve the adjacent normal-looking fundus. This was a slowly progressive phenomenon, and the lesion was found to be still increasing in size when followed for more than 3 months, but became stationary by about 5–7 months. Thus, the involved part of the fundus had large, irregular dark brown areas, with steel-gray patches, in addition to the grayish-white granular depigmented patches scattered among them and the pigmented areas encroaching upon the normal-looking fundus (Figs. 18.2b, 18.3b, 18.6, and 18.7).
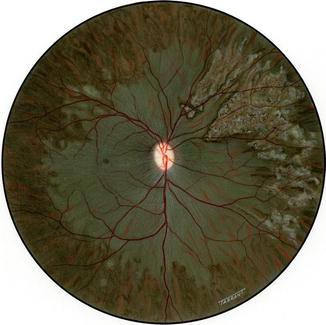
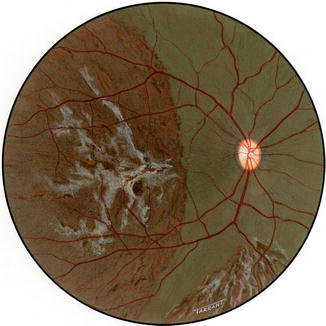

Fig. 18.6
Fundus drawing of the right eye of rhesus monkey shows late lesions after occlusion of all PCAs

Fig. 18.7
Fundus drawing of the right eye of rhesus monkey shows late lesions after occlusion of all PCAs
In the depigmented areas, the main choroidal vessels were sometimes more conspicuous than in the normal fundus. Some of these patches, when associated with choroidal destruction, had a mottled, shining, grayish-white appearance, with prominently visible large choroidal vessels (Fig. 18.8).
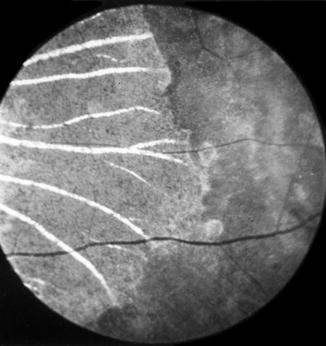

Fig. 18.8
Fundus picture of rhesus monkey eye shows lesions 58 days after occlusion of all PCAs situated nasal to the optic disc (Reproduced from Hayreh and Baines [14])
In addition to the characteristic fundus lesions seen in these eyes, a few showed the following pattern:
(a)
Instead of the ophthalmoscopically well-defined patches of lesions mentioned above, only a diffuse, scattered, irregular mottling of the pigment was visible in some areas. These patches were seen better on fluorescein angiography than by ophthalmoscopy.
(b)
In others no depigmented patches were seen, but large areas of dark brown discoloration of the fundus (similar to the haloes) were seen, usually situated in the region extending from the outer limits of the central part of the fundus (the central part includes the macular region, optic disc, peripapillary choroid, and adjacent areas) to the equator of the eye. Examination of the peripheral fundus showed that in the equatorial region of the affected part of the fundus, there was a scalloped border between the posterior dark-colored fundus and the anterior normal fundus. This indicated that the pigmentary disturbance in the involved region was diffuse and extensive and was associated with increased pigmentation of the fundus.
I found differences in the distribution of the fundus lesions in occlusion of the lateral, medial, or all PCAs, as is evident from the following description.
After Lateral Posterior Ciliary Artery Occlusion
The lesions involved the temporal part of the fundus in every eye (Figs. 18.1, 18.2, 18.3, and 18.7). In two eyes, they also encroached upon the adjacent part of the inferior nasal sector (Fig. 18.2). In no eye did the lesions involve the peripapillary choroid or the area extending from the optic disc to the nasal margin of the macula. The macula was involved in three eyes. The temporal lesions were of extremely varied distribution, but were usually situated in the area between the equator and the central region (Figs. 18.2, 18.3, and 18.7). In the macular region, particularly the region between the optic disc and the macula, horizontal retinal folds were frequently seen for the first 2–3 weeks.
After Medial Posterior Ciliary Artery Occlusion
The lesions appeared in the nasal part of the fundus in one half of the eyes, no lesion being seen in the rest. Their distribution was also very varied, and they were situated in the area between the equator and the central region of the fundus. In almost all cases, the lesions seen in medial PCA occlusion were much smaller in size and more localized than those seen in lateral PCA occlusion.
After All Posterior Ciliary Artery Occlusion
The PCA supply to the eye was completely cut off. Much more extensive lesions were seen than in either of the previous groups (Figs. 18.4 and 18.9). In this group, the lesions were seen in every eye and involved many parts of the fundus. In eyes in which a small short PCA escaped cutting, no fundus lesions were seen in the corresponding sector.


Fig. 18.9
Diagrammatic representation of the various fundus lesions seen in the rhesus monkey eyes after occlusion of all PCAs. Dark areas represent the distribution of white patches during the acute phase and the shaded areas that of subsequent discoloration of the fundus (Reproduced from Hayreh and Baines [14]). X position of the macula
Fluorescein Fundus Angiographic Findings
These ischemic choroidal lesions were also investigated by fluorescein fundus angiography.
Fluorescein Pattern at the Initial Stages of PCA Occlusion
During the transit of the dye through the eye, no background choroidal fluorescence was seen in the region of the white patches; these patches, however, showed late fluorescence a few minutes after the transit of the dye (Fig. 18.10a). The late fluorescence was due to staining of the necrotic tissue of the white patches by the fluorescein, which had leaked very slowly from the underlying choroid into the overlying tissue. This may be due to the following two mechanisms:
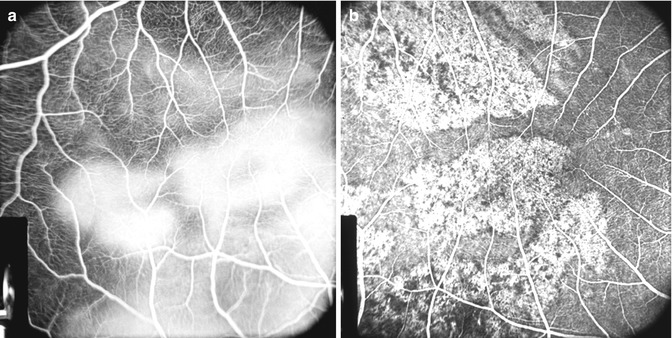

Fig. 18.10
Fluorescein fundus angiograms of the macular region and the area temporal to it after lateral PCA occlusion 2 days after the occlusion in rhesus monkey. (a) Shows late fluorescence of the white patches. (b) The same after 26 days of occlusion shows unmasking of the choroidal fluorescence (Reproduced from Hayreh and Baines [14])
1.
Late Filling of the Choroid in the Region of the Occluded PCA Soon After the Occlusion: The various mechanisms responsible for this were discussed in detail elsewhere [13]. Briefly, these include the following modes, singly or in combination:
(a)
Via Retrograde Choroidal Circulation from the Vortex Veins: The territory of the uveal tract drained by one vortex vein is usually supplied by three sets of arteries, medial and lateral PCAs and the anterior ciliary arteries, as shown in Fig. 18.11. Thus, occlusion of the medial and/or lateral PCA reduces the blood pressure in the venous tributaries draining the non-filling sector of the choroid. Blood could regurgitate from the main stem of the vortex vein (which still has venous return from the anterior segment of the eye) into these empty tributaries, which leads to retrograde filling of the choroidal vascular bed. This is further supported by the fact that these isolated filling areas appear during the late venous phase of the dye transit (Fig. 18.12 and 18.13). The normal ocular pulsation, by acting as a pumping mechanism, would help in the filling and emptying of the vortex veins in this sector.
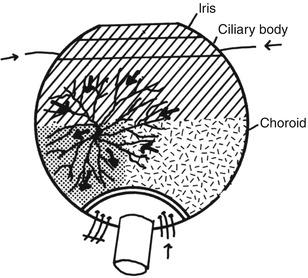
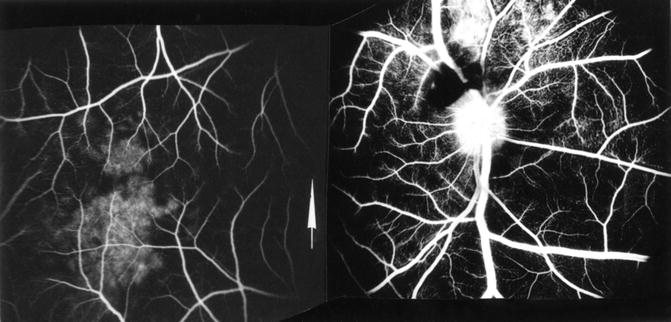
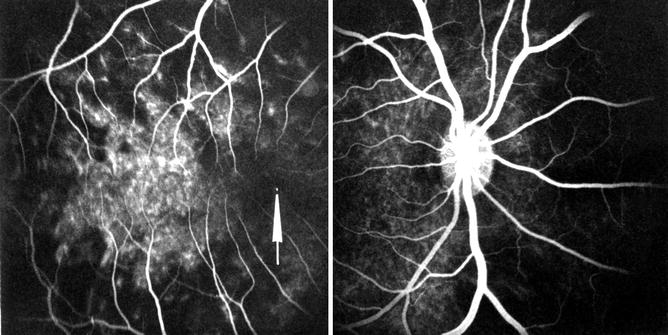

Fig. 18.11
Diagram shows route of retrograde flow of blood via the vortex vein into the part of choroid supplied by an occluded PCA. Arrows indicate direction of flow of blood in various vessels (Reproduced from Hayreh and Baines [13])

Fig. 18.12
A composite fluorescein fundus angiogram, during retinal venous phase, of the right eye of rhesus monkey 45 min after occlusion of all PCAs with superior temporal vortex vein occlusion. It shows patchy choroidal filling with normal retinal filling. Angiogram shows retinal hemorrhage above optic disc. Arrow indicates fovea (Reproduced from Hayreh and Chopdar [16])

Fig. 18.13
A composite fluorescein fundus angiogram of the right eye of rhesus monkey, after occlusion of all PCAs with inferior temporal vortex vein occlusion, after transit of dye, showing faint patchy choroidal fluorescence temporal to the fovea (arrow) (Reproduced from Hayreh and Chopdar [16])
(b)
Via the Posterior Episcleral Arterial Plexus: Anastomoses on the surface of the posterior sclera between the PCAs, optic nerve sheath vessels, and other vessels would enable dye or blood to reach the occluded vessel distal to the occlusion, as demonstrated in the silicone rubber injection studies [13]. The cutting of the PCAs some distance posterior to the sclera would leave these anastomoses intact. The choroidal filling through these anastomoses increased with the passage of time.
(c)
Via the Retrolaminar Optic Nerve Pial Vascular Plexus: The peripapillary choroid anastomoses with the retrolaminar optic nerve pial vascular plexus (Fig. 18.14). With normal blood supply in the pial plexus, isolated, small, patchy filling of the peripapillary choroid slowly starts from that source. This usually appeared in the venous phase. Fluorescence spreads from these patches to adjacent areas of the choroid (Figs. 18.15 and 18.16).
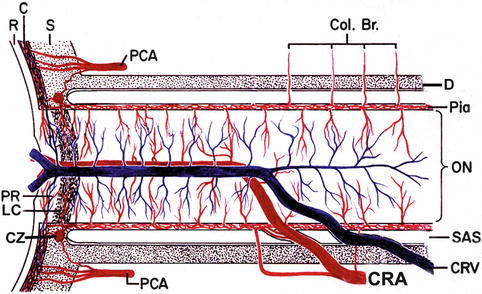
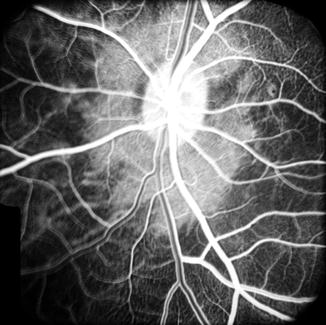

Fig. 18.14
Schematic representation of blood supply of the optic nerve. Abbreviations used: C choroid, CRA central retinal artery, Col. Br. collateral branches, CRV central retinal vein, CZ circle of Zinn and Haller, D dura, LC lamina cribrosa, ON optic nerve, PCA posterior ciliary artery, PR prelaminar region, R retina, S sclera, SAS subarachnoid space

Fig. 18.15
Fluorescein fundus angiogram after occlusion of all PCAs, 1 h after occlusion, during retinal late venous phase. Note no filling of choroid and filling of lower peripapillary choroid only (Reproduced from Hayreh and Baines [13])

Fig. 18.16
Fluorescein fundus angiogram 29 h after occlusion of all PCAs in rhesus monkey, during retinal arterial phase. Most of the peripapillary choroid has been filled and some main choroidal vessels fill from the peripapillary choroid (Reproduced from Hayreh and Baines [13])
2.
Breakdown of Blood–Retinal Barrier in the Retinal Pigment Epithelium (RPE): Normally the RPE cell layer exercises a blood-retinal barrier which prevents diffusion of choroidal fluid into the retina. But, in the region of the infarcted RPE, the blood-retinal barrier breaks down, as shown by our horseradish peroxidase tracer study, discussed below [17]. That results in the diffusion of fluorescein from the choroid into the infarcted pigment epithelium and staining of it.
Fluorescein Pattern at Later Stages of PCA Occlusion
The white patches progressed to become thin, and unmasking of the choroidal fluorescence became evident, indicating a thinning of the RPE (Fig. 18.10b). This unmasking became more prominent with the passage of time and was at its greatest when the lesions assumed the granular depigmented grayish-white appearance (Figs. 18.5b and 18.17). The marginal parts of the lesion showed a bright granular fluorescence, which appeared with the filling of the choroid. The central part of the lesion showed evidence of reduction of choroidal vessels and some granular fluorescence as well. In these patches, the main choroidal arteries showed a distinct filling and unmasking (Fig. 18.17). This fluorescence of the patches persisted only during the transit of dye through the choroid and faded off thereafter. There was no staining of the lesions after about 3 weeks.
The steel-gray patches in the haloes around the depigmented choroidal lesions usually slightly masked the choroidal fluorescence (Fig. 18.5). In the eyes showing no depigmented patches but large areas of dark brown discoloration, fluorescein fundus angiography showed no abnormality. Similarly, when such a discoloration was associated with typical depigmented lesions, no fluorescein angiographic abnormality corresponding to the dark brown patches was evident.
No other fluorescein angiographic abnormality was detected in the fundus. The findings suggested a degeneration of the RPE in the areas of the patches, with some destruction of small and medium-sized choroidal vessels in the central part of these lesions (Fig. 18.5b). The retina showed no abnormality. In areas with marked choroidal degeneration (Fig. 18.8), apart from a few big choroidal vessels, no other choroidal vessels were seen (Fig. 18.17), except at their margins.
Progressive Filling of the Choroid Later on in PCA Occlusion
Fluorescein fundus angiography also showed that with time, the choroidal vascular bed filling in the region of the occluded PCA gradually improved, so that finally the choroidal vascular bed filled almost normally. The mechanism of that filling is discussed above. This fact is important clinically because normal filling of the choroidal vascular bed months later in eyes with PCA occlusion (e.g., in giant cell arteritis) has been misinterpreted as showing that there was no occlusion of the PCA – a common argument. I have seen that in eyes with PCA occlusion due to giant cell arteritis. Therefore, complete occlusion of the PCA can only be demonstrated on fluorescein angiography within the first few days.
Histopathological Findings
The various fundus lesions showed the following histological changes on light microscopy. These changes never involved the anterior part of the choroid supplied by the anterior ciliary arteries.
Retina
In eyes with white patches, necrotic material was present between Bruch’s membrane and the inner layers of the retina in the region of the patches (Fig. 18.18). The necrotic material consisted of dead RPE, rods, cones, and to a variable degree the outer nuclear layer. The pigment granules of the RPE were irregularly scattered in the necrotic mass. The latter varied in thickness and structure, depending upon its age, and disappeared completely in older lesions.
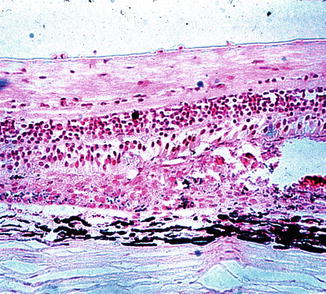

Fig. 18.18
Sections through a white choroidal ischemic patch, 11 days after occlusion of all the PCAs in rhesus monkey, show necrosis of the pigment epithelium, rods, cones, and outer nuclear layer. Hematoxylin and eosin staining (Reproduced from Hayreh and Baines [14])
In old lesions, the most common change observed was the loss of visual cells and outer nuclear layer in the area of the lesion (Figs. 18.19 and 18.20). A few nuclei of the outer nuclear layer might still be seen (Figs. 18.19 and 18.20). Rarely, the inner nuclear layer was somewhat involved and disorganized, but not significantly.
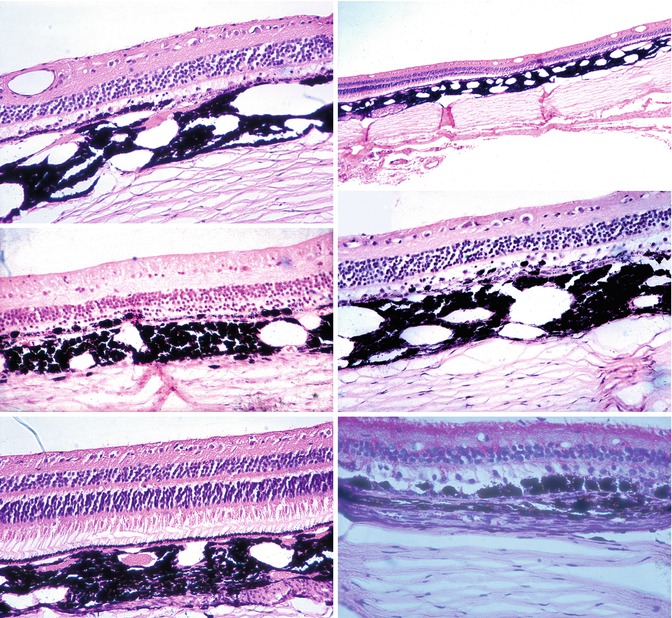

Figs. 18.19, 18.20, 18.21, 18.22, 18.23, and 18.24
Sections through late choroidal ischemic lesions produced by occlusion of PCAs. Hematoxylin and eosin staining
Chorioretinal adhesions were frequently seen in ophthalmoscopically well-marked lesions (Figs. 18.19 and 18.21). In these adhesions, the RPE and Bruch’s membrane were often absent or disorganized. In a few instances, some big pigmented cells were seen to have invaded the retina close to the choroid (Fig. 18.22). No migration of the pigment into the deeper layers of the retina was seen.
Retinal Pigment Epithelium
This was involved significantly in the vast majority of these eyes. There were, however, instances where, with the retina showing absence of the visual cells and outer nuclear layer, no significantly demonstrable changes in the RPE could be seen. The reverse was also true.
The changes in the RPE varied from mild degeneration to complete disorganization (Figs. 18.19, 18.20, 18.21, and 18.22) or even to the complete absence of the RPE. Usually the degeneration of the RPE was more widespread than the changes in the overlying retina. The normal RPE is thin, uniform in thickness, and light brown, having regularly arranged cells and a very finely granular appearance (Fig. 18.23). The changes in the RPE seen in the region of the white patches have already been mentioned under “Retina.” In old lesions, the RPE might be represented by very dark (sometimes even black) clumps or granules. In others the epithelium might be irregular in thickness, with coarse granules. Some of these granules were discrete, rounded, of varying size, and black. At some places, a few of these isolated granules were the sole evidence of the RPE (Fig. 18.22). Big, round, pigmented cells were seen lying over Bruch’s membrane in some areas or even invading the adjacent layers of the retina. These were considered to be grossly hypertrophied RPE cells. Sometimes localized hypertrophy of the RPE was seen around the area of the destruction of the RPE (Fig. 18.24).
In eyes where the ophthalmoscope showed pigmented fundus lesions – e.g., brownish discoloration of the fundus – the only histological evidence was roughening and some thickening of the RPE.
Bruch’s Membrane
Usually Bruch’s membrane showed no demonstrable changes in mild fundus lesions. Where chorioretinal adhesions were seen, the membrane was generally absent (Fig. 18.22). A possible thickening and hypertrophy of Bruch’s membrane was seen in some places and frequently in areas close to chorioretinal adhesions. In some eyes, however, a normal Bruch’s membrane was present, even in the presence of the adhesions (Fig. 18.21).
Choroid
Usually no demonstrable change was seen in the choroid, except for some obliteration of the choriocapillaris. In some of the areas which showed evidence of absence of choroidal vessels on fluorescein fundus angiography, there was localized atrophy of the choroid. In the eyes irrigated with glutaraldehyde, normally no blood was seen in the choroidal vessels if adequate circulation was present, but in eyes with freshly occluded PCAs, the choroid was full of blood on the side of the occlusion.
Light and Electron Microscopy and Horseradish Peroxidase (HRP) Tracer Study
In another study [17], histopathological changes were investigated both by light and electron microscopy. In this investigation, the horseradish peroxidase (HRP) tracer technique was also used to study the RPE blood-retinal barrier. Following were the findings of this study.
Twenty-Four Hours After PCA Occlusion
In the macular region, the choriocapillaris was congested and showed extensive thrombosis. The RPE was focally necrotic, alternating with early focal reactive proliferation (Fig. 18.25). Necrosis of photoreceptor inner and outer segments was evident and was associated with extensive focal pyknosis in the outer nuclear layer. The inner retina appeared unaffected. Leakage of HRP extended through the RPE into the subretinal space and passed the outer limiting membrane and outer nuclear layer to the outer plexiform layer. Regional variation of the ischemic changes was remarkable. Nasally, focal RPE necrosis was seen, associated with macrophage-like cells that were infiltrating into the sub-RPE space, and focal thinning and necrosis of the outer nuclear layer were also noted. However, only mild HRP leakage through the RPE was present at this site. Inferiorly, changes included scattered RPE pyknosis with focal necrosis in the overlying photoreceptor cells.
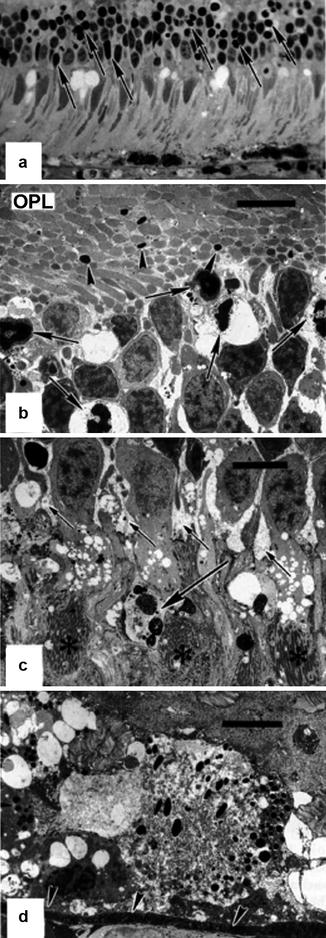

Fig. 18.25
Eye of rhesus monkey 24 h after PCA occlusion with no vortex vein occlusion. (a) Light micrographic illustration of the macular area displaying features of photoreceptor cell damage such as vacuolation of the inner segments, distortion of the outer segments, and marked focal necrosis in the outer nuclear layer (arrows). The retinal pigment epithelium is necrotic and focally absent, and the choriocapillaris is congested (methylene blue-azur II, original magnification ×480). (b) Corresponding electron micrograph of the outer nuclear layer and the outer plexiform layer (OPL) showing marked vacuolation and nuclear pyknosis of photoreceptor cells (arrows) and condensation of axons (arrowheads) in the OPL (uranyl acetate-lead citrate, bar = 5 μm). (c) There is also marked Müller cell edema in the outer nuclear layer (short arrows), and occasional autophagocytosis (long arrow) is seen. Asterisks indicate photoreceptor inner segments (uranyl acetate-lead citrate, bar = 5 μm). (d) The retinal pigment epithelium is markedly necrotic with cytoplasmic swelling and rupture of cell membranes leading to considerable vacuolation. Arrowheads indicate Bruch’s membrane (uranyl acetate-lead citrate, bar = 5 μm) (Reproduced from Loeffler et al. [17])
Two Weeks After PCA Occlusion
In the macula, the choriocapillaris revealed an extensive occlusion and loss of capillaries that was accompanied by a proliferation of pericytes. The remarkable feature was plaques of RPE fibrous metaplasia proliferating in multiple layers on Bruch’s membrane (Fig. 18.26). The HRP tracer infiltrated the RPE plaque and extended into the subretinal space to the opposing outer limiting membrane. Some RPE cells showed marked vacuolation and liquefaction of the cytoplasm. Photoreceptor outer and inner segments were largely lost, and the outer limiting membrane approached the apical RPE surface. The outer nuclear layer was severely thinned, and pyknotic nuclei were observed. The inner retina overlying the RPE plaque appeared mildly gliotic. An extensive RPE plaque was also noted in the superior quadrant of the retina, but HRP leakage was only apparent within the RPE plaque and in the subretinal space. Beneath the plaque, the choriocapillaris exhibited marked replacement fibrosis and the capillaries were extensively reduced in number. The outer nuclear layer was almost completely lost at this site. In the temporal quadrant, the choriocapillaris was patent and there was a focal area of RPE depigmentation and proliferation and a markedly thinned outer nuclear layer.
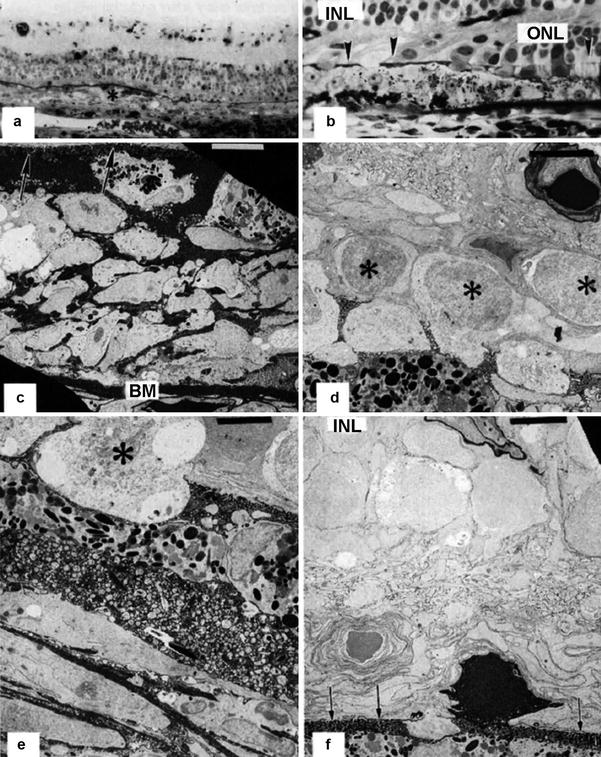

Fig. 18.26
A rhesus monkey eye 2 weeks after PCA occlusion with no vortex vein occlusion. (a) Extensive plaque formation (asterisk) with multiple layers of retinal pigment epithelial cells is seen in the macula overlying an area of absent choriocapillaris. Black horseradish peroxidase product accumulates in Bruch’s membrane (methylene blue-azur II, original magnification ×120). (b) There is widespread loss of the outer nuclear layer (ONL), and the outer limiting membrane (arrowheads) is approaching the inner nuclear layer (INL) and the apical retinal pigment epithelial surface. Black horseradish peroxidase product accumulates in Bruch’s membrane (methylene blue-azur II, original magnification ×480). (c) Corresponding electron micrograph shows retinal pigment epithelial plaque formation with multilayering of retinal pigment epithelial cells with intense intercellular staining of horseradish peroxidase between Bruch’s membrane (BM) and the thickened outer limiting membrane (arrows) formed by villous processes of the retinal pigment epithelial and Müller cell fibers (uranyl acetate-lead citrate, bar = 5 μm). (d) Remaining photoreceptor nuclei (asterisks) are approaching the retinal pigment epithelial plaque. Note the horseradish peroxidase staining around the retinal arteriole (uranyl acetate-lead citrate, bar = 5 μm). (e) Electron micrograph of the upper aspect of the retinal pigment epithelial plaque. Note the macrophage-like cell morphology and the remnant of the photoreceptor inner segment (asterisk) (uranyl acetate-lead citrate, bar = 5 μm). (f) In an adjacent area, only one photoreceptor cell that is intensely stained by intracellular is left. Arrows indicate the site of the outer limiting membrane. Note the horseradish peroxidase staining around the retinal vessel in the inner nuclear layer (INL) (uranyl acetate-lead citrate, bar = S μm)
Three Months After PCA Occlusion
In the macula, there was patchy loss of choriocapillaris, and a RPE plaque had formed, and the outer nuclear layer was mildly reduced in thickness (Fig. 18.27). The regional ischemic changes appeared to be milder than the necrotic changes that were noted in the previously described eyes. Scattered macrophages were observed in the outer plexiform layer. The HRP infiltrated the RPE plaque and extended into the subretinal space. Nasally, a large RPE plaque with focal thinning of the overlying outer nuclear layer and occasional macrophages in the outer plexiform layer were present, but no significant HRP leakage was detected in this area.
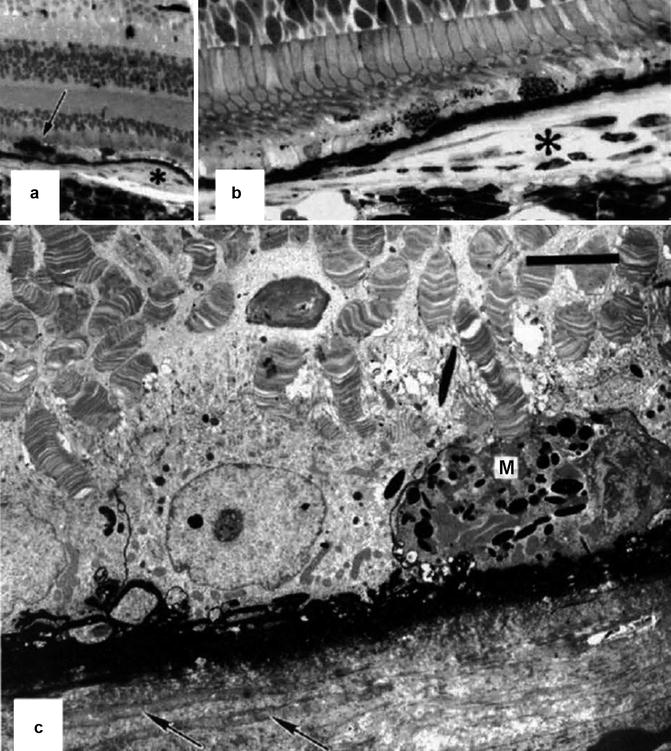

Fig. 18.27
Eye 3 months after PCA occlusion with no vortex vein occlusion (Reproduced from Loeffler et al. [17]). (a) The macula shows a small retinal pigment epithelial plaque (asterisk) and proliferation of the adjacent retinal pigment epithelium (arrow) overlying an area of choriocapillary loss. The horseradish peroxidase product accumulates in Bruch’s membrane, which appears as a black line (methylene blue-azur II, original magnification ×120). (b) Higher magnification of the retinal pigment epithelial plaque (asterisk); no significant amounts of horseradish peroxidase are seen within the plaque. Note also the partial absence of the choriocapillaris (methylene blue-azur II, original magnification ×480). (c) Electron micrograph of the retinal pigment epithelial plaque. Note the replacement of the choriocapillaris by collagen and fibroblast-like cells (arrows) and the depigmentation as well as macrophage-like transformation (M) of the retinal pigment epithelial. The outer segments are distorted. Black horseradish peroxidase product accumulates in Bruch’s membrane and in the interstitial space between retinal pigment epithelial cells (uranyl acetate-lead citrate, bar = 5 μm)
Thus, this study showed that the RPE reaction to ischemia began with intracellular edema and cell necrosis in the area of damage while, over time, adjacent RPE cells migrated to repopulate this region. Plaque formation as a response to ischemia was noted as early as 1 week after PCA occlusion, but it was only seen in those areas with the most severe necrotic changes in the outer retina. The outer nuclear layer became thinned with focal photoreceptor cell death 24 h after vascular occlusion. Damage to the photoreceptor cell layer appeared irrevocable and obvious at 3 months, even overlying fairly normal repopulated RPE. Three months after the ischemic insult, no areas with complete RPE denudation were seen, as was described in a previous study [20]. It appeared that RPE cells could repopulate a previously denuded area within this period. The inner retina appeared largely unaffected, but rarefaction of axons in the outer plexiform layer was frequently noted.
Horseradish Peroxidase (HRP) Tracer Technique Findings
The breakdown of the blood-retinal barrier was evident 6 h after ischemia when leakage of HRP from the choroid into the outer retina was observed. As in the studies by Uyama et al. [20] and Koshibu and ltotagawa [21], penetration of HRP into diseased RPE cells was noticed, while the tight junctions between these cells were still intact and prevented intercellular HRP movement into the outer retina. Continuous disruption of the chorioretinal-blood barrier was noted until 1 month after ischemia, but leakage of HRP through the RPE appeared to decrease after 1 week. Three months later, the chorioretinal-blood barrier was mostly restored, with only minimal HRP leakage at the sites of severe ischemia, whereas the loss of photoreceptor cells from the outer nuclear layer was irrevocable.
Conclusions on PCA Occlusion
When various PCAs were cut, white patches of markedly varied shape, size, and distribution appeared in the parts of the fundus supplied by the occluded artery within 24 h. These patches stained during the late phase of fluorescein in angiography. After 1 week the patches began to change gradually, so that over a 2–3-week period, they became grayish-white, granular, depigmented areas. During the evolution of the lesions, they became surrounded by deeply pigmented haloes, and the adjacent fundus showed similar pigmentation. In the grayish-white depigmented patches, intravenous fluorescein fundus angiography revealed degeneration of the pigment epithelium and partial destruction of the choroidal vasculature. The sites of these white patches corresponded to post-occlusion nonperfusion areas in the choroid. Histological studies of the fundus lesions revealed destruction of visual cells and the outer nuclear layer, with degeneration of the RPE and Bruch’s membrane, but with little change in the choroid. Later on it appeared that RPE cells could repopulate a previously denuded area within a 3-month period. The inner retina appeared largely unaffected, but rarefaction of axons in the outer plexiform layer was frequently noted. HRP study showed breakdown of the RPE blood-retinal barrier initially, but 3 months later it was mostly restored.
Correlation of Site of Lesion with Choroidal Ischemia
In the majority of the eyes, the site and extent of the lesions after PCA occlusion depended to a great extent upon the extent of the choroidal filling, via the routes discussed above and elsewhere [13]. The lesions usually appeared in areas devoid of choroidal filling after the occlusion, as revealed by nonperfusion of the area of the choroid during the transit of the dye in angiographic studies. In some, however, it was difficult to be definite about the exact area of nonperfusion in the choroid. On follow-up studies, a delayed filling of the choroid in the areas of the lesion was usually evident. It is, again, difficult to explain the occurrence of elongated pointed lesions with narrow intervening strips of normal-looking fundus.
Fundus lesions after the section of the PCAs in the rabbit have been described in the literature [22–25]. Nicholls [25] recorded three types of changes, depending upon the number of vessels cut: (a) no change, (b) phthisis bulbi, and (c) localized area of retinal degeneration (this was seen in the region supplied by the bulk of the vessels). Degeneration was produced only after cutting at least two thirds of the PCAs, although Wagenmann [22] noticed these changes on section of only a few ciliary arteries. Nicholls [25] found extreme pallor in the area immediately after occlusion, spreading fanwise from the optic nerve head out to the ora; in a few hours, these were less pale but swollen. In about a week, two changes were noticed:
(i)
A normal appearance returned to certain areas with the restoration of circulation.
(ii)
Where circulation was not restored, the pale area gradually became spotted here and there with round sharply circumscribed dense accumulations of pigment (mostly in the equatorial region), which progressively changed over several weeks to stabilize in 4–5 months, when a contraction of the degenerate area, with shallow folds in the normal areas, was seen.
There is a difference between rabbits and primates in the blood supply of the retina and the rest of the eye. In rabbits the entire thickness of the retina is supplied by the choroid, but in primates the choroid supplies only the outer layers of the retina. Therefore, findings in rabbits are not always applicable to primates, including humans. That would account for some of the differences in my findings. For example, in none of my animals did a circumscribed dense accumulation of pigment or phthisis bulbi develop. No migration of the pigment into the retina was seen in my studies.
Since my first publication about experimental choroidal ischemic lesions in PCA occlusion in monkeys, several similar studies [26–34] – all but one [26] in Japanese [27–35] (which I cannot read) – have been reported with almost similar findings. The Japanese studies [28, 30, 31] mainly deal with ultrastructural changes of the retinal pigment epithelial cells following experimental occlusion of the PCA in the rhesus monkey. Algvere [26] produced choroidal ischemia in owl monkeys on occlusion of a vortex vein, associated with proliferation or degeneration of the RPE, frequently accompanied by photoreceptor degeneration and circumscribed retinal detachment.
Optic Nerve Head Ischemic Lesions Produced by the PCA Occlusion
I investigated the effect of experimental PCA occlusion on the optic nerve head in a large number of rhesus monkey eyes (this was first such study) [15]. PCA occlusion produced anterior ischemic optic neuropathy. Since ischemic optic neuropathy is a commonly seen clinical condition, I feel it is essential to describe the nature of these lesions and their evolution with time in detail, as seen in this comprehensive experimental study. This is because clinically there is a good deal of confusion and controversy on the etiology and evolution of ischemic optic nerve head lesions. Following is a detailed account of the ophthalmoscopic, fluorescein fundus angiographic, and histopathological findings of the optic nerve head ischemic lesions and their evolution seen in this experimental study.
Ophthalmoscopic and Fluorescein Fundus Angiographic Findings
After Lateral PCA Occlusion (31 Eyes)
One Hour After Occlusion
Ophthalmoscopic examination of the fundus at this time showed slight edema of the disc in 7 of 31 eyes. This represented anterior ischemic optic neuropathy. This edema involved the lower part of the optic disc in three eyes, the temporal part in 2, the nasal part in one, and the whole optic disc in one. Eight of 31 eyes showed pallor of the disc, as compared to its pre-occlusion appearance, the pallor being usually more evident on the temporal part of the disc. As discussed in Chap. 4, it is pertinent to point out that there is wide interindividual variation in the supply by PCAs to the optic nerve head, and that may be responsible for different sites of optic disc edema in different eyes.
At this stage, fluorescein fundus angiographic studies revealed very faint or no fluorescence of the disc during the retinal arterial phase and moderate fluorescence during the arteriovenous phase. The fluorescence was usually more marked on the nasal than the temporal part of the disc. This is interesting, because the pre-occlusion studies in these, and in normal eyes, mostly showed a more marked fluorescence on the temporal than the nasal part. Thus, occlusion of the lateral PCA reversed the fluorescence pattern in these discs. During the late phase, blurring of the disc margins was seen in the 7 eyes with optic disc edema.
One to Two Days After Occlusion
Out of the 12 follow-up eyes, eight eyes were examined at this stage. Five of these showed a mild degree of edema of the disc, involving either the whole (Fig. 18.28) or a part of the disc. Three of the discs with pallor 1 h after PCA occlusion remained pale at this stage. Two eyes were normal.
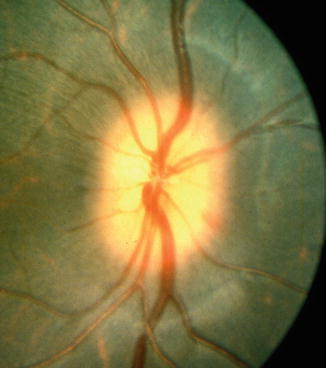

Fig. 18.28
Fundus photograph shows edema of the optic disc 2 days after occlusion of the lateral PCA (Reproduced from Hayreh and Baines [15])
At this stage, fluorescein fundus angiographic studies revealed a slight fluorescence of the disc in the retinal arterial phase, largely uniform in distribution in the majority, which increased during the arteriovenous phase and again decreased in the venous phase. A mild degree of fluorescein leakage was seen in five eyes – usually along the temporal or lower margins.
One Week After Occlusion
There was no significant change.
After 3–4 Weeks’ PCA Occlusion
In the three eyes with edema of the disc, which were followed for more than 1 week, the edema subsided in about 3–4 weeks. Subsequently, these three eyes developed pallor, more marked on the temporal than the nasal part, and showed varying degrees of optic atrophy. None of these three eyes showed any immediate post-occlusion pallor of the disc, though two showed a degree of edema. Fluorescein fundus angiography at the end of the follow-up revealed reduced fluorescence in the atrophic discs.
After Medial PCA Occlusion (17 Eyes)
One Hour After PCA Occlusion
One disc was edematous in its lower part. No hyperemia was seen. Five discs showed a mild degree of pallor.
On fluorescein fundus angiography, the discs showed faint to moderate fluorescence in the retinal arterial phase, with the temporal part usually more fluorescent than the nasal. Fluorescence increased in intensity during the arteriovenous phase and was reduced in the venous phase. In the disc showing edema inferiorly, fluorescein leakage was seen in the same area.
Two Days After PCA Occlusion
Mild edema of the disc was seen in two eyes involving the upper part in one and the lower part in the other. Half of the discs showed a mild degree of hyperemia.
One Week After PCA Occlusion
The edema of the disc cleared.
Two Months After PCA Occlusion
One of the previously edematous discs developed a moderate degree of optic atrophy with pallor, more marked in the nasal part than the temporal part (before the occlusion, the temporal part of the disc had been paler than the nasal part).
After All PCA Occlusion (37 Eyes)
One Hour After PCA Occlusion
The discs showed uniform pallor in about a quarter of the eyes involved.
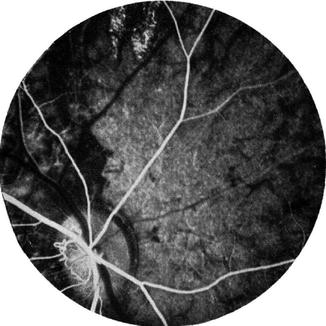
On fluorescein fundus angiography, during the retinal arterial phase, only a faint disc fluorescence was usually seen, with some discs showing no fluorescence at all (Fig. 18.29). A uniform fluorescence of moderate to marked degree was seen during the arteriovenous phase. The late phase showed, in a third of these eyes, a leak of fluorescein with blurring of the lower border of the disc; the earliest filling of the peripapillary choroid occurred in the corresponding sector. This suggests that the fluorescein had leaked from the ischemic vessels on restoration of the peripapillary circulation.
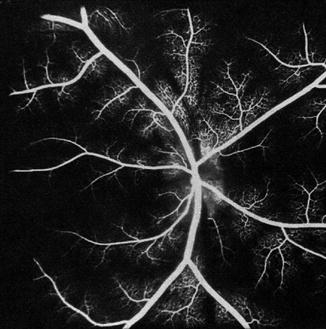

Fig. 18.29
Fluorescein fundus angiogram 1 h after occlusion of all PCAs in a rhesus monkey shows, during the retinal arterial phase, no filling of the choroid (Reproduced from Hayreh and Baines [15])
Two Days After PCA Occlusion
A mild degree of edema of the disc with fluorescein leakage was seen in most of these eyes; this usually subsided in about 2 weeks.
More Than 5 Weeks After PCA Occlusion
Of the 13 eyes followed for more than 5 weeks, 11 showed optic atrophy of variable extent. In four eyes with optic atrophy, there were enlarged collateral vessels on the disc, connecting the retinal vessels on the disc surface with the peripapillary choroid. These were on the temporal side in three (Fig. 18.30) and on the nasal side in one. No such vessels were seen before occluding the PCAs in these eyes. Their exact significance is not known, because they were not seen in other eyes with or without optic atrophy.
These findings represent anterior ischemic optic neuropathy. Fluorescein fundus angiography immediately after occlusion of the PCAs mostly revealed, during the early part of the transit of the dye, reduced fluorescence of the part of the disc supplied by the occluded PCA, e.g., usually the temporal part in lateral PCA occlusion and the nasal part in medial PCA occlusion. This study not only confirmed the role of the ciliary circulation in the blood supply of the optic nerve head (see Chap. 5) but also helped by giving more information about the origins of fluorescence of the optic disc during the different phases of the retinal transit of the dye in fluorescein fundus angiography. During the retinal arterial phase, before the retinal capillaries fill, the disc fluorescence represents the deep ciliary supply to the optic disc, while in the arteriovenous phase when the retinal capillaries are completely filled, the fluorescence of the disc is mainly due to the retinal capillary fluorescence and very little to the deep ciliary fluorescence. The one eye in which the cilioretinal artery did not fill on occlusion of the lateral PCA demonstrated this very clearly, because the area where the retinal capillaries were missing was much less fluorescent than the area where they were full (Fig. 18.31).
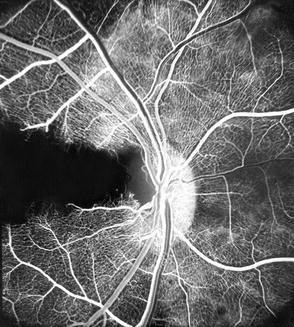

Fig. 18.31
Fluorescein fundus angiogram of the right eye of a rhesus monkey eye with a cilioretinal artery and occlusion of lateral PCA. It shows filling defect in the distribution of the occluded PCA as well as filling defect in retinal vascular bed in the distribution of the cilioretinal artery (a branch of lateral PCA) (Reproduced from Hayreh and Baines [13])
Histopathological Studies
In the optic discs showing ophthalmoscopic evidence of atrophy, histological examination revealed atrophic changes in the disc and the retrolaminar part supplied by the PCAs (Figs. 18.32 and 18.33). In some eyes, the atrophic area had a well-marked border with the normal optic nerve (Fig. 18.32). This well-marked infarction of the optic nerve has also been reported in patients with arteritic ischemic optic neuropathy [36–39] (Figs. 18.34, 18.35, 18.36, 18.37, and 18.38). In other monkey eyes, the junction between the atrophic and normal regions was not always so well defined, although the distribution of atrophic changes was similar. In some of these, the atrophic changes were more marked in the peripheral part of the optic nerve. There was a localized patch of optic atrophy in the peripheral part of the retrolaminar optic nerve on the occluded side in a significant number of these nerves.


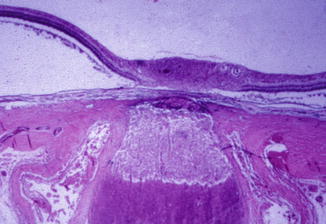
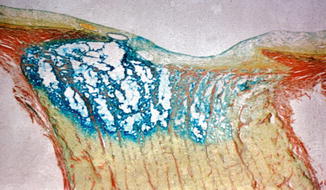
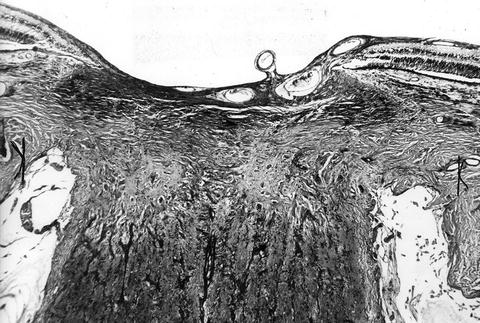
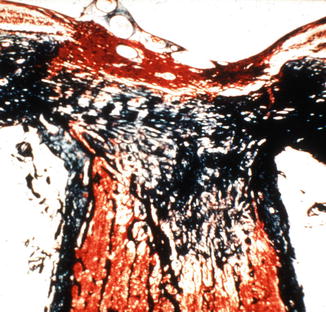
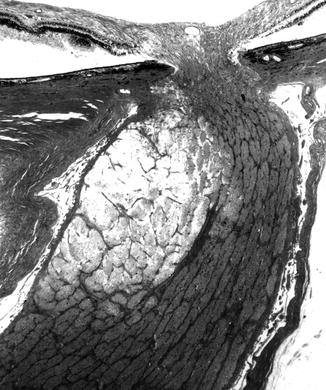

Fig. 18.32
Photomicrograph of optic disc and optic nerve 36 days after occlusion of all PCAs in rhesus monkey (Masson’s trichrome stain). ×38

Fig. 18.33
Photomicrograph of optic disc and optic nerve 42 days after occlusion of lateral PCA in rhesus monkey (Masson’s trichrome stain). ×38

Fig. 18.34
Photomicrographs of the optic nerve head and retrolaminar optic nerve of the right eye of a patient with 4-week-old arteritic anterior ischemic optic neuropathy show a well-defined area of infarction (Verhoeff’s modified elastic stain) (Reproduced from MacMichael and Cullen [38])

Fig. 18.35
Photomicrographs of the optic nerve head and retrolaminar optic nerve of a left eye with 18-day-old arteritic anterior ischemic optic neuropathy show a sharply defined area of necrosis in the anterior part of the optic nerve and a partially disintegrated lamina cribrosa. There is presence of hyaluronidase-sensitive material (blue) similar to that found in cavernous degeneration of the optic nerve (Alcian blue stain) (Reproduced from Hinzpeter and Naumann [39])

Fig. 18.36
Photomicrographs of the optic nerve head and retrolaminar optic nerve of the left eye with 4-month-old arteritic anterior ischemic optic neuropathy and no light perception vision. It shows atrophy of prelaminar tissue and fibrosis and gliosis of laminar and retrolaminar tissue (Verhoeff elastic stain) (Reproduced from Henkind et al. [37])

Fig. 18.37
Photomicrographs of the optic nerve head and retrolaminar optic nerve of an eye with arteritic anterior ischemic optic neuropathy show atrophy of prelaminar tissue and fibrosis of the laminar and retrolaminar tissue (Reproduced by courtesy of late Dr. Paul Henkind)

Fig. 18.38
Photomicrograph of the optic nerve head and retrolaminar optic nerve of an eye with non-arteritic anterior ischemic optic neuropathy showing infarction of temporal half of the optic nerve head and retrolaminar optic nerve (Reproduced from Cogan [36])
Conclusion
Experimental occlusion of the various PCAs produced optic disc edema during the initial phases. Optic atrophy developed after 5–6 weeks in 16, 60, and 85 % of the eyes after occlusion of the medial, lateral, and all PCAs, respectively. These changes in the optic disc are typical of ischemic optic neuropathy. Histopathological studies revealed involvement of the optic nerve head and retrolaminar part of the optic nerve – the parts of the optic nerve supplied by the posterior ciliary circulation, as has been shown in arteritic anterior ischemic optic neuropathy eyes of patients with giant cell arteritis [36–39].
Simultaneous Occlusion of PCAs and Vortex Veins
My studies [14, 15] discussed above, dealing with experimental occlusion of various PCAs in monkeys, showed patchy infarction of the retinal pigment epithelium and outer parts of the retina and development of anterior ischemic optic neuropathy. The lesions were located in the distribution of the occluded PCAs. One consistent, intriguing finding was the great variation in the extent of the ischemic lesions produced by identical experimental PCA occlusions, not only among different studies [14, 15, 40–42] but also within the same study. For example, my studies [14] showed a notable variation in the extent of the lesions after occlusion of the lateral, medial, and all PCAs – not only between the three groups but, much more importantly, between eyes in the same group. Other investigators [40, 41] also reported similar notable variation in the incidence and extent of the retinal lesions after PCA occlusion. The same holds true of the incidence and severity of anterior ischemic optic neuropathy seen in different studies after identical PCA occlusion [15, 40–42]; this led some authors [40, 43] to question the validity of my theory that anterior ischemic optic neuropathy is caused by a disturbance in the PCA circulation [15, 44]. My study [14] suggested that the extent of the retinal infarcts after occlusion of the PCA depends largely on the extent of choroidal filling from other sources, because I found that the lesions usually appeared in areas devoid of choroidal filling on fluorescein fundus angiography immediately after the occlusion. One important source of choroidal filling was considered to be the vortex vein system [13]; as discussed above, the territory of the uveal tract drained by one vortex vein is usually supplied not only by the medial and lateral PCAs but also by a large number of the anterior ciliary arteries [13, 45]. Thus, occlusion of the PCAs cuts down the blood flow in the posterior tributaries of the vortex vein draining the nonperfused part of the choroid; however, the anterior tributaries of the vortex vein are still receiving blood from the uvea in front of the equator (mainly supplied by the anterior ciliary arteries). I [13, 45] postulated that in these eyes the blood would reflow from the main stem of the vortex vein into its empty posterior tributaries, causing retrograde filling of the choroid in the region of the occluded artery (Fig. 18.11). This was suggested by the fact that, in the occluded PCA territory, isolated areas of the choroid started to fill with fluorescein on angiography during the late venous phase (Figs. 18.12 and 18.13) [13]. The normal ocular pulsation, by acting as a pumping mechanism, would help in the filling and emptying of the veins in the occluded territory. The high concentration of oxygen in the blood of the vortex veins should help substantially in preventing ischemic damage [13, 45]. Occluding the vortex vein in such eyes should force much more blood into the occluded choroid than is seen normally and should protect the choroid and the tissues in the region of the occluded artery from ischemic damage.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree