Pediatrics
8.1 LEUKOCORIA
Definition
A white pupillary reflex (see Figure 8.1.1).
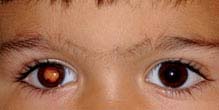
FIGURE 8.1.1. Leukocoria.
Etiology
 Retinoblastoma: A malignant tumor of the retina that appears as a white, nodular mass that breaks through the internal limiting membrane into the vitreous (endophytic), as a yellowish subretinal mass lesion often underlying a serous retinal detachment (exophytic), or as a diffusely spreading lesion simulating uveitis (diffuse infiltrating). Iris neovascularization is common. Pseudohypopyon and vitreous seeding may occur. Cataract is uncommon, and the eye is normal in size. May be bilateral, unilateral, or multifocal. Diagnosis is usually made between 12 and 24 months of age. A family history may be elicited in about 10%.
Retinoblastoma: A malignant tumor of the retina that appears as a white, nodular mass that breaks through the internal limiting membrane into the vitreous (endophytic), as a yellowish subretinal mass lesion often underlying a serous retinal detachment (exophytic), or as a diffusely spreading lesion simulating uveitis (diffuse infiltrating). Iris neovascularization is common. Pseudohypopyon and vitreous seeding may occur. Cataract is uncommon, and the eye is normal in size. May be bilateral, unilateral, or multifocal. Diagnosis is usually made between 12 and 24 months of age. A family history may be elicited in about 10%.
 Toxocariasis: A nematode infection that may appear as a localized, white, elevated granuloma in the retina or as a diffuse endophthalmitis. Associated with localized inflammation of ocular structures, vitreous traction bands and related macular dragging, traction retinal detachment, and cataract. It is rarely bilateral and is usually diagnosed between 6 months and 10 years of age. Paracentesis of the anterior chamber may reveal eosinophils; serum enzyme-linked immunosorbent assay (ELISA) test for Toxocara organisms is positive. The patient may have a history of contact with puppies or eating dirt. Toxocariasis may also be acquired prenatally and present as a congenital infection.
Toxocariasis: A nematode infection that may appear as a localized, white, elevated granuloma in the retina or as a diffuse endophthalmitis. Associated with localized inflammation of ocular structures, vitreous traction bands and related macular dragging, traction retinal detachment, and cataract. It is rarely bilateral and is usually diagnosed between 6 months and 10 years of age. Paracentesis of the anterior chamber may reveal eosinophils; serum enzyme-linked immunosorbent assay (ELISA) test for Toxocara organisms is positive. The patient may have a history of contact with puppies or eating dirt. Toxocariasis may also be acquired prenatally and present as a congenital infection.
 Coats disease (see Figure 8.1.2): A retinal vascular abnormality resulting in small multifocal outpouchings of the retinal vessels. Leukocoria may develop secondary to an exudative retinal detachment or to extensive yellow intraretinal and subretinal exudate. Usually develops in boys during the first decade of life; more severe cases occur in early childhood. Coats disease is rarely bilateral. No family history.
Coats disease (see Figure 8.1.2): A retinal vascular abnormality resulting in small multifocal outpouchings of the retinal vessels. Leukocoria may develop secondary to an exudative retinal detachment or to extensive yellow intraretinal and subretinal exudate. Usually develops in boys during the first decade of life; more severe cases occur in early childhood. Coats disease is rarely bilateral. No family history.
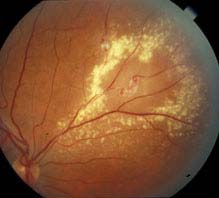
FIGURE 8.1.2. Coats disease.
 Persistent fetal vasculature (PFV)/persistent hyperplastic primary vitreous (PHPV): A developmental ocular abnormality consisting of a varied degree of glial and vascular proliferation in the vitreous cavity. It is usually associated with a slightly small eye. Typically there is a membrane behind the lens that may place traction on the ciliary processes. This is a progressive condition with a cataract present at birth or early in life. The membrane and lens may rotate anteriorly, shallowing the anterior chamber and resulting in secondary glaucoma. Retinal detachments may be seen. Rarely bilateral. No family history.
Persistent fetal vasculature (PFV)/persistent hyperplastic primary vitreous (PHPV): A developmental ocular abnormality consisting of a varied degree of glial and vascular proliferation in the vitreous cavity. It is usually associated with a slightly small eye. Typically there is a membrane behind the lens that may place traction on the ciliary processes. This is a progressive condition with a cataract present at birth or early in life. The membrane and lens may rotate anteriorly, shallowing the anterior chamber and resulting in secondary glaucoma. Retinal detachments may be seen. Rarely bilateral. No family history.
 Congenital cataract: Opacity of the lens present at birth; may be unilateral or bilateral. There may be a family history or an associated systemic disorder. See 8.8, Congenital Cataract.
Congenital cataract: Opacity of the lens present at birth; may be unilateral or bilateral. There may be a family history or an associated systemic disorder. See 8.8, Congenital Cataract.
 Retinal astrocytoma: A sessile to slightly elevated, yellow-white retinal mass that may be calcified and is often associated with tuberous sclerosis and rarely neurofibromatosis. May occur on the optic nerve head (giant drusen) in patients with tuberous sclerosis.
Retinal astrocytoma: A sessile to slightly elevated, yellow-white retinal mass that may be calcified and is often associated with tuberous sclerosis and rarely neurofibromatosis. May occur on the optic nerve head (giant drusen) in patients with tuberous sclerosis.
 Retinopathy of prematurity (ROP): Predominantly occurs in premature children. Leukocoria is usually the result of a retinal detachment. See 8.2, Retinopathy of Prematurity.
Retinopathy of prematurity (ROP): Predominantly occurs in premature children. Leukocoria is usually the result of a retinal detachment. See 8.2, Retinopathy of Prematurity.
 Others: Retinochoroidal coloboma, retinal detachment, familial exudative vitreoretinopathy (FEVR), myelinated nerve fibers, uveitis, incontinentia pigmenti, toxoplasmosis.
Others: Retinochoroidal coloboma, retinal detachment, familial exudative vitreoretinopathy (FEVR), myelinated nerve fibers, uveitis, incontinentia pigmenti, toxoplasmosis.
Work-Up
1. History: Age at onset? Family history of one of the conditions mentioned? Prematurity? Contact with puppies or habit of eating dirt?
2. Complete ocular examination, including a measurement of corneal diameters (look for a small eye), an examination of the iris (look for neovascularization), and an inspection of the lens (look for a cataract). A dilated fundus examination and anterior vitreous examination are essential.
3. Any or all of the following may be helpful in diagnosis and planning treatment:
—B-scan ultrasonography (US), especially if there is no view of the fundus. This can be used to look for calcification within a suspected lesion.
—Intravenous fluorescein angiogram (Coats disease, ROP, retinoblastoma).
—Computed tomographic (CT) scan or magnetic resonance imaging (MRI) of the orbit and brain (retinoblastoma), particularly for bilateral cases or those with a family history, as well as for cases of advanced Coats disease.
—Serum ELISA test for Toxocara (positive at 1:8 in the vast majority of infected patients).
—Systemic examination (retinal astrocytoma, retinoblastoma) by pediatrician.
—Anterior chamber paracentesis (toxocariasis). See Appendix 13, Anterior Chamber Paracentesis.
NOTE: Anterior chamber paracentesis in a patient with a retinoblastoma can possibly lead to tumor cell dissemination.
4. Consider examination under anesthesia (EUA) in young or uncooperative children, particularly when retinoblastoma, toxocariasis, Coats disease, or ROP is being considered as a diagnosis. See 8.8, Congenital Cataract, for a more specific cataract work-up.
Treatment
1. Retinoblastoma: Chemoreduction, intra-arterial chemotherapy, cryotherapy, thermotherapy, laser photocoagulation, or plaque radiotherapy. These treatment modalities are typically used in combination. Enucleation is now reserved for cases not amenable to the above treatment options. Systemic chemotherapy is used in metastatic disease. Irradiation is no longer used as it is associated with a high incidence of secondary tumors later in life.
2. Toxocariasis:
—Steroids (topical, periocular, or systemic routes may be used, depending on the severity of the inflammation).
—Consider a surgical vitrectomy when vitreoretinal traction bands form or when the condition does not improve or worsens with medical therapy.
—Consider laser photocoagulation of the nematode if it is visible.
3. Coats disease: Laser photocoagulation or cryotherapy to leaking vessels. Though associated with a poor outcome, surgery may be required for a retinal detachment.
4. PFV/PHPV:
—Cataract and retrolental glial membrane extraction.
—Treat any amblyopia, though visual outcome is often poor secondary to foveal hypoplasia associated with PFV.
5. Congenital cataract: See 8.8, Congenital Cataract.
6. Retinal astrocytoma: Observation.
7. ROP: See 8.2, Retinopathy of Prematurity.
Follow-Up
Variable, depending on the diagnosis.
8.2 RETINOPATHY OF PREMATURITY
Risk Factors
 Prematurity, especially <32 weeks of gestation.
Prematurity, especially <32 weeks of gestation.
 Birthweight <1,500 g (3 lb, 5 oz), especially <1,250 g (2 lb, 12 oz).
Birthweight <1,500 g (3 lb, 5 oz), especially <1,250 g (2 lb, 12 oz).
 Supplemental oxygen therapy, neonatal sepsis, hypoxemia, hypercarbia, and concurrent illness.
Supplemental oxygen therapy, neonatal sepsis, hypoxemia, hypercarbia, and concurrent illness.
 Risk factors mentioned above, when present concurrently, have an additive effect on the risk for development of ROP.
Risk factors mentioned above, when present concurrently, have an additive effect on the risk for development of ROP.
Signs
Critical. Avascular peripheral retina. Demarcation line between vascular and avascular retina.
Other. Extraretinal fibrovascular proliferation, vitreous hemorrhage, retinal detachment, or leukocoria, sometimes bilateral. Engorgement and tortuosity of the vessels in the posterior pole and/or iris in plus disease. Poor pupillary dilation despite mydriatic drops. In older children and adults, decreased visual acuity, amblyopia, myopia, strabismus, macular dragging, lattice-like vitreoretinal degeneration, or retinal detachment may occur.
Differential Diagnosis
 FEVR: Appears similar to ROP, except FEVR is most commonly autosomal dominant (though family members may be asymptomatic); asymptomatic family members often show peripheral retinal vascular abnormalities. There usually is no history of prematurity or oxygen therapy. See 8.3, Familial Exudative Vitreoretinopathy.
FEVR: Appears similar to ROP, except FEVR is most commonly autosomal dominant (though family members may be asymptomatic); asymptomatic family members often show peripheral retinal vascular abnormalities. There usually is no history of prematurity or oxygen therapy. See 8.3, Familial Exudative Vitreoretinopathy.
 Incontinentia pigmenti: X-linked dominant condition that only occurs in girls. Lethal in males. Characterized by skin changes including erythematous macular papular lesions, vesicles, hypopigmented patches, and alopecia. Central nervous system and dental abnormalities may be seen.
Incontinentia pigmenti: X-linked dominant condition that only occurs in girls. Lethal in males. Characterized by skin changes including erythematous macular papular lesions, vesicles, hypopigmented patches, and alopecia. Central nervous system and dental abnormalities may be seen.
 See 8.1, Leukocoria, for additional differential diagnoses.
See 8.1, Leukocoria, for additional differential diagnoses.
Classification
Location
 Zone I: Posterior pole: Twice the disc–fovea distance, centered around the disc (poorest prognosis).
Zone I: Posterior pole: Twice the disc–fovea distance, centered around the disc (poorest prognosis).
NOTE: With the nasal edge of the optic disc at one edge of the field of view with a 28D lens, the limit of Zone I is at the temporal field of view.
 Zone II: From zone I to the nasal ora serrata; temporally equidistant from the disc.
Zone II: From zone I to the nasal ora serrata; temporally equidistant from the disc.
NOTE: ROP should not be considered zone III until one is sure the nasal side is vascularized to the ora serrata.
 Zone III: The remaining temporal periphery.
Zone III: The remaining temporal periphery.
Extent
 Number of clock hours (30-degree sectors) involved.
Number of clock hours (30-degree sectors) involved.
Severity
 Stage 1: Flat demarcation line separating the vascular posterior retina from the avascular peripheral retina (see Figure 8.2.1).
Stage 1: Flat demarcation line separating the vascular posterior retina from the avascular peripheral retina (see Figure 8.2.1).
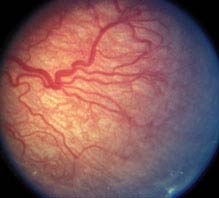
FIGURE 8.2.1. Retinopathy of prematurity: Stage 1.
 Stage 2: Ridged demarcation line.
Stage 2: Ridged demarcation line.
 Stage 3: Ridged demarcation line with fibrovascular proliferation or neovascularization extending from the ridge into the vitreous (see Figure 8.2.2).
Stage 3: Ridged demarcation line with fibrovascular proliferation or neovascularization extending from the ridge into the vitreous (see Figure 8.2.2).
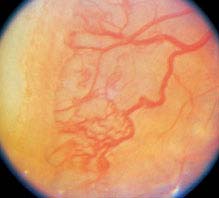
FIGURE 8.2.2. Retinopathy of prematurity: Stage 3.
 Stage 4A: Extrafoveal partial retinal detachment.
Stage 4A: Extrafoveal partial retinal detachment.
 Stage 4B: Foveal partial retinal detachment.
Stage 4B: Foveal partial retinal detachment.
 Stage 5: Total retinal detachment.
Stage 5: Total retinal detachment.
NOTE: The overall stage is determined by the most severe manifestation; however, it is recommended to define each stage and record its extent.
“Plus” Disease
(See Figure 8.2.3.)
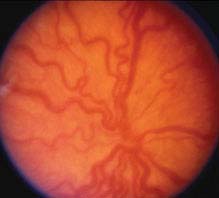
FIGURE 8.2.3. Retinopathy of prematurity: Plus disease.
At least two quadrants of engorged veins and tortuous arteries in the posterior pole; iris vascular engorgement, poor pupil dilatation, and vitreous haze with more advanced plus disease. If plus disease is present, a “+” is placed after the stage (e.g., stage 3+). If vascular dilatation and tortuosity are present but inadequate to diagnose plus disease, it is called “pre-plus” disease and noted after the stage (e.g., stage 3 with pre-plus disease). Posterior ROP (usually Zone I) with plus disease out of proportion to the peripheral retinopathy, or so-called “rush” disease (also known as aggressive posterior disease), may progress rapidly to stage 5 ROP without passing through the other stages. This aggressive ROP may also show hemorrhages at the junction between vascular and avascular retina.
Threshold Disease
Defines high-risk eyes that meet the criteria for treatment:
—Zone I, any stage with plus disease.
—Zone I, stage 3, no plus disease.
—Zone II, stage 2 or 3 with plus disease.
Prethreshold Disease
Defines less severely advanced eyes that should be monitored closely for progression to threshold disease:
—Zone I, stage 1 and 2 without plus disease.
—Zone II, stage 3 without plus disease.
Screening Recommendations
 Birth weight <1,500 g.
Birth weight <1,500 g.
 Gestational age ≤32 weeks.
Gestational age ≤32 weeks.
 Selected infants with birth weight of 1,500 to 2,000 g or gestational age ≥32 weeks with unstable clinical course thought to be at high risk.
Selected infants with birth weight of 1,500 to 2,000 g or gestational age ≥32 weeks with unstable clinical course thought to be at high risk.
 Timing of first eye examination is based on gestational age at birth.
Timing of first eye examination is based on gestational age at birth.
Work-Up
1. Dilated retinal examination with scleral depression at 31 to 32 weeks after date of mother’s last menstrual period, or 4 weeks after birth, whichever is later.
2. Can dilate with combination phenylephrine 1.0% and cyclopentolate 0.2%.
Treatment
Based on severity. The presence of plus disease is typically an indication for early treatment.
1. Prethreshold disease: Close observation.
2. Threshold disease: Ablative treatment of avascular zone of retina. Laser photocoagulation is preferred over cryotherapy. Treatment should be instituted within 72 hours (see Figure 8.2.4).
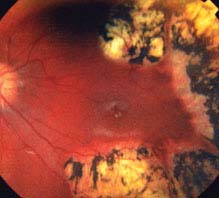
FIGURE 8.2.4. Retinopathy of prematurity after laser treatment.
3. Stages 4 and 5: Surgical repair of retinal detachment by scleral buckling, vitrectomy surgery, or both.
Follow-Up
 One week or less: Zone I, stage 1 or 2; or zone II, stage 3.
One week or less: Zone I, stage 1 or 2; or zone II, stage 3.
 One to 2 weeks: Immature vascularization, zone I; or zone II, stage 2; or zone I, regressing ROP.
One to 2 weeks: Immature vascularization, zone I; or zone II, stage 2; or zone I, regressing ROP.
 Two weeks: Zone II, stage 1; or zone II, regressing ROP.
Two weeks: Zone II, stage 1; or zone II, regressing ROP.
 Two to 3 weeks: Immature vascularization, zone II, no ROP; or zone III, stage 1 or 2; or zone III, regressing ROP.
Two to 3 weeks: Immature vascularization, zone II, no ROP; or zone III, stage 1 or 2; or zone III, regressing ROP.
1. Children who have had ROP have a higher incidence of myopia, strabismus, amblyopia, macular dragging, cataracts, glaucoma, and retinal detachment. An untreated fully vascularized fundus needs examination at age 6 months to rule out these complications.
NOTE: Because of the possibility of late retinal detachments and other ocular complications, ROP patients should be followed at yearly intervals for life.
2. Acute-phase ROP screening can be discontinued when any of the following signs is present, indicating that the risk of visual loss from ROP is minimal or passed:
—Zone III retinal vascularization attained without previous zone I or II ROP. If there is doubt about the zone or if the postmenstrual age is unexpectedly young, confirmatory examinations may be warranted.
—Full retinal vascularization.
—Postmenstrual age of 45 weeks and no prethreshold or worse ROP.
8.3 FAMILIAL EXUDATIVE VITREORETINOPATHY
Symptoms
Majority are asymptomatic, but patients may report decreased vision.
Signs
(See Figure 8.3.1.)

FIGURE 8.3.1. FEVR with a falciform fold.
Critical. Peripheral retinal capillary nonperfusion, most prominently temporally, and may be present for 360 degrees. Bilateral but may be asymmetric. Peripheral retinal vessels have a fimbriated border. Present at birth.
Other. Peripheral neovascularization and/or fibrovascular proliferation at the border of vascular and avascular retina; temporal dragging of macula through contraction of fibrovascular tissue; vitreous hemorrhage; tractional, exudative, or rhegmatogenous retinal detachment; peripheral intraretinal and subretinal lipid exudation. May present with strabismus or leukocoria in childhood. Cataract, band keratopathy, neovascular glaucoma, or phthisis possible.
Differential Diagnosis
 Retinopathy of prematurity: Appears similar to FEVR, but there should be no family history, and there should be a history of prematurity. See 8.2, Retinopathy of Prematurity.
Retinopathy of prematurity: Appears similar to FEVR, but there should be no family history, and there should be a history of prematurity. See 8.2, Retinopathy of Prematurity.
 See 8.1, Leukocoria, for additional differential diagnosis, e.g., retinoblastoma, Coats, PFV/PHPV, incontinentia pigmenti, Norrie disease, X-linked retinoschisis, and peripheral retinal nonperfusion. Positive family history and bilaterality can help distinguish from others.
See 8.1, Leukocoria, for additional differential diagnosis, e.g., retinoblastoma, Coats, PFV/PHPV, incontinentia pigmenti, Norrie disease, X-linked retinoschisis, and peripheral retinal nonperfusion. Positive family history and bilaterality can help distinguish from others.
Etiology
Due to defects in the Wnt signaling pathway. Usually autosomal dominant, though autosomal recessive, and X-linked cases have been reported. No history of prematurity or oxygen therapy. FEVR is similar to ROP, histologically.
Work-Up
1. History: Positive family history? No history of prematurity or oxygen therapy?
2. Complete ocular examination, including dilated retinal examination with a slit lamp and a 60- or 90-diopter lens looking for temporal dragging of the macula. Indirect ophthalmoscopy looking for peripheral nonperfusion, fibrovascular membranes, tractional retinal detachment. All family members should also have dilated retinal examinations.
Treatment
Laser of peripheral nonperfused retina is sometimes considered if there is documented progression. Small, stable tufts of neovascularization can be observed. Scleral buckling and vitrectomy can be considered for retinal detachments. Often complicated by proliferative vitreoretinopathy. Treat amblyopia as needed.
Follow-Up
Asymptomatic patients should be followed every 6 to 12 months throughout life to monitor for progression.
8.4 ESODEVIATIONS IN CHILDREN
Signs
(See Figure 8.4.1.)
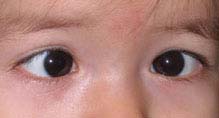
FIGURE 8.4.1. Esotropia.
Critical. Either eye is turned inward. The nonfixating eye turns outward to fixate straight ahead when the previously fixating eye is covered during the cover–uncover test. See Appendix 3, Cover/Uncover and Alternate Cover Tests.
Other. Amblyopia, overaction of the inferior oblique muscles.
Differential Diagnosis
 Pseudoesotropia: The eyes appear esotropic; however, there is no ocular misalignment detected during cover–uncover testing. Usually, the child has a wide nasal bridge, prominent epicanthal folds, or a small interpupillary distance (see Figure 8.4.2).
Pseudoesotropia: The eyes appear esotropic; however, there is no ocular misalignment detected during cover–uncover testing. Usually, the child has a wide nasal bridge, prominent epicanthal folds, or a small interpupillary distance (see Figure 8.4.2).
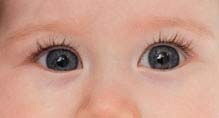
FIGURE 8.4.2. Pseudoesotropia.
 See 8.6, Strabismus Syndromes.
See 8.6, Strabismus Syndromes.
Types
Comitant or Concomitant Esotropic Deviations
A manifest convergent misalignment of the eyes in which the measured angle of esodeviation is nearly constant in all fields of gaze at distance fixation.
1. Congenital (infantile) esotropia: Manifests by age 6 months, the angle of esodeviation is usually large (>40 to 50 prism diopters) and mostly equal at distance and near fixation. Refractive error is usually normal for age (slightly hyperopic). Amblyopia is uncommon but may be present in those who do not cross-fixate. Family history may be present but is not mandatory. Latent nystagmus, inferior oblique overaction, and dissociated vertical deviation may develop as late findings. Congenital esotropia can occur in up to 30% of children with neurological and developmental disorders (e.g., cerebral palsy, hydrocephalus).
2. Accommodative esotropia: Convergent misalignment of the eyes associated with activation of the accommodative reflex. Average age of onset is 2.5 years.
Subtypes of accommodative esotropia:
—Refractive accommodative esotropia: These children are hyperopic in the range of +3.00 to +10.00 diopters (average, +4.75). The measured angle of esodeviation is usually moderate (20- to 30-prism diopters) and is relatively equal at distance and near fixation. Full hyperopic correction eliminates the esodeviation. The accommodative convergence–accommodation angle ratio (AC/A) is normal. Amblyopia is common at presentation.
—Nonrefractive accommodative esotropia (high AC/A ratio): The measured angle of esodeviation is greater at near fixation than at distance fixation. The refractive error may range from normal for age (slight hyperopia) to high hyperopia (may be seen in conjunction with refractive-type accommodative esotropia) or even myopia. Amblyopia is common.
—Partial or decompensated accommodative esotropia: Refractive and nonrefractive accommodative esotropias that have a reduction in the esodeviation when given full hyperopic correction, but still have a residual esodeviation. The residual esodeviation is the nonaccommodative component.
3. Sensory-deprivation esotropia: An esodeviation that occurs in a patient with a monocular or binocular condition that prevents good vision.
4. Divergence insufficiency: A convergent ocular misalignment that is greater at distance fixation than at near fixation. This is a diagnosis of exclusion and must be differentiated from divergence paralysis, which, when sudden in onset, can be associated with pontine tumors, neurologic trauma, and elevated intracranial pressure. See 10.8, Isolated Sixth Nerve Palsy.
Incomitant Esodeviations
The measured angle of esodeviation increases in lateral gaze at distance fixation.
1. Central nervous system pathology causing increased intracranial pressure: Acute and new onset of diplopia secondary to an acquired sixth nerve palsy, which may be accompanied by nystagmus, headache, or other focal neurologic deficits depending on etiology.
2. Medial rectus restriction (e.g., thyroid disease, medial orbital wall fracture with entrapment).
3. Lateral rectus weakness (e.g., isolated sixth cranial nerve palsy, slipped or detached lateral rectus from trauma or previous surgery).
4. See 8.6, Strabismus Syndromes, and 10.8, Isolated Sixth Nerve Palsy, for additional etiologies.
Other
1. Esophoria: Latent esodeviation controlled by fusion. Eyes are aligned under binocular conditions.
2. Intermittent esotropia: Esodeviation that is intermittently controlled by fusion. Becomes manifest spontaneously, especially with fatigue or illness.
Work-Up
1. History: Age of onset, frequency of crossing, prior therapy (e.g., glasses, patching).
2. Visual acuity of each eye, with best correction and pinhole. Color vision and stereopsis.
3. Ocular motility examination; observe for restricted movements or oblique overactions.
4. Measure the distance deviation in all fields of gaze and the near deviation in the primary position (straight ahead) using prisms (see Appendix 3, Cover/Uncover and Alternate Cover Tests). Look specifically for an esotropia increasing in either side gaze.
5. Manifest and cycloplegic refractions especially if <7 years of age.
6. Complete eye examination. Look for any cranial nerve abnormalities and causes of sensory deprivation.
7. If acute-onset non-accommodative esotropia, divergence insufficiency or paralysis, paralysis, or incomitant esotropia is present, a head CT scan (axial and coronal views) and/or an MRI is necessary to rule out an intracranial or orbital process, extraocular muscle pathology, or bony lesion.
8. With incomitant esodeviation greater in side gaze, determine whether the lateral rectus function is deficient or the medial rectus is restricted. Forced-duction testing may be necessary for that distinction (see Appendix 6, Forced-Duction Test and Active Force Generation Test). This may need to be done under anesthesia for children. Consider thyroid function tests or a work-up for myasthenia gravis, or look for characteristics of strabismus syndromes (see 8.6, Strabismus Syndromes).
Treatment
In all cases, correct refractive errors of +2.00 diopters or more, and in children treat any amblyopia (see 8.7, Amblyopia).
1. Congenital esotropia: When equal vision is obtained in the two eyes, strabismus surgery is indicated.
2. Accommodative esotropia: Glasses must be worn full time.
a. If the patient is <6 years, correct the hyperopia with the full cycloplegic refraction.
b. If the patient is >6 years, attempts should be made to give as close to the full-plus refraction as possible, knowing that some may not tolerate the full prescription. Attempts to push plus lenses during the manifest (noncycloplegic) refraction until distance vision blurs may be tried to give the most plus lenses without blurring distance vision. The goal of refractive correction should be straight alignment without sacrificing visual acuity.
c. If the patient’s eyes are straight at distance with full correction, but still esotropic at near fixation (high AC/A ratio), treatment options include the following:
 Bifocals (executive type) +2.50 or +3.00 diopter add, with top of the bifocal at the lower pupillary border.
Bifocals (executive type) +2.50 or +3.00 diopter add, with top of the bifocal at the lower pupillary border.
 Extraocular muscle surgery targeting the near deviation only may be indicated. This typically requires posterior fixation sutures to the muscle to modify the surgical effect for near only.
Extraocular muscle surgery targeting the near deviation only may be indicated. This typically requires posterior fixation sutures to the muscle to modify the surgical effect for near only.
 Wearing full-plus distance glasses only.
Wearing full-plus distance glasses only.
NOTE: There is no universal agreement on the treatment of patients with excess crossing at near only.
3. Non-accommodative, partially accommodative, or decompensated accommodative esotropia: Muscle surgery is usually performed to correct only the significant residual esotropia that remains when glasses are worn.
4. Sensory-deprivation esotropia:
 Attempt to identify and correct the cause of poor vision.
Attempt to identify and correct the cause of poor vision.
 Amblyopia treatment.
Amblyopia treatment.
 Give the full cycloplegic correction (in fixing eye) if the patient is <6 years of age, otherwise give the full manifest refraction.
Give the full cycloplegic correction (in fixing eye) if the patient is <6 years of age, otherwise give the full manifest refraction.
 Muscle surgery to correct the manifest esotropia.
Muscle surgery to correct the manifest esotropia.
 All patients with very poor vision in one eye need to wear protective polycarbonate lens glasses at all times.
All patients with very poor vision in one eye need to wear protective polycarbonate lens glasses at all times.
Follow-Up
At each visit, evaluate for amblyopia and measure the degree of deviation with prisms (with glasses worn).
1. If amblyopia is present, see 8.7, Amblyopia, for management.
2. In the absence of amblyopia, the child is reevaluated in 3 to 6 weeks after a new prescription is given. If no changes are made and the eyes are straight, the patient should be followed several times a year when young, decreasing to annually when stable.
3. When a residual esotropia is present while the patient wears glasses, an attempt is made to add more plus power to the current prescription. Children <6 years should receive a new cycloplegic refraction; plus lenses are pushed without cycloplegia in older children. The maximal additional plus lens that does not blur distance vision is prescribed. If the eyes cannot be straightened with more plus power, then a decompensated accommodative esotropia has developed (see preceding).
4. Hyperopia often decreases slowly after age 5 to 7 years, and the strength of the glasses may need to be reduced so as not to blur distance vision. If the strength of the glasses must be reduced to improve visual acuity and the esotropia returns, then this is a decompensated accommodative esotropia (see preceding).
8.5 EXODEVIATIONS IN CHILDREN
Signs
(See Figure 8.5.1.)
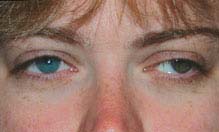
FIGURE 8.5.1. Exotropia.
Critical. Either eye is constantly or intermittently turned outward. On the cover–uncover test, the uncovered eye moves from the out-turned position to the midline to fixate when the previously fixating eye is covered (see Appendix 3, Cover/Uncover and Alternate Cover Tests).
Other. Amblyopia, “A” pattern deviation (superior oblique overaction producing an increased deviation in downgaze compared to upgaze), “V” pattern deviation (inferior oblique overaction producing an increased deviation in upgaze compared to downgaze), vertical deviation.
Differential Diagnosis
Pseudoexotropia: The patient appears to have an exodeviation, but no movement is noted on cover–uncover testing despite good vision in each eye. A wide interpupillary distance, a naturally large angle k (angle between the pupil and the visual axis), or temporal dragging of the macula (e.g., from ROP, FEVR, toxocariasis, or other retinal disorders) may be responsible.
Types
1. Exophoria: Exodeviation controlled by fusion under conditions of normal binocular vision. Usually asymptomatic, but prolonged strenuous visual activity may cause asthenopia.
2. Intermittent exotropia: The most common type of exodeviation in children. Onset is usually before age 5. Frequency often increases over time. Amblyopia is rare.
A. There are three phases:
—Phase 1: One eye turns out at distance fixation, spontaneously or when it is covered. Usually occurs when the patient is fatigued, sick, or not attentive. The eyes become straight within one to two blinks or when the cover is removed. The eyes are straight at near fixation. Patient often closes one eye or squints in bright sunlight. This is likely due to dissociation and breakdown of their binocular alignment.
—Phase 2: Increasing frequency of exotropia at distance fixation. Exophoria begins to occur at near fixation.
—Phase 3: There is a constant exotropia at distance and near fixations.
B. These phases can be seen in all four types of intermittent exotropia:
—Basic: Exodeviation is approximately the same with distance and near fixation.
—True divergence excess: Exodeviation that remains greater at distance than near after a period of monocular occlusion.
—Simulated divergence excess: Exodeviation that is initially greater with distance fixation than near that becomes approximately the same after an interval of monocular occlusion.
—Convergence insufficiency: Exodeviation is greater at near than distance. Distinct from isolated convergence insufficiency. See 13.4, Convergence Insufficiency.
3. Constant exotropia: Encountered more often in older children. There are three types:
—Congenital exotropia: presents before age 6 months with a large angle deviation. Uncommon in otherwise healthy infants and may be associated with a central nervous system or craniofacial disorder.
—Sensory-deprivation exotropia: An eye that does not see well for any reason may turn outward.
—Consecutive exotropia: Follows previous surgery for esotropia.
4. Duane syndrome, type 2: Limitation of adduction of one eye, with globe retraction and narrowing of the palpebral fissure on attempted adduction. Rarely bilateral. See 8.6, Strabismus Syndromes.
5. Neuromuscular abnormalities:
—Third nerve palsy: See 10.5, Isolated Third Nerve Palsy.
—Myasthenia gravis: See 10.11, Myasthenia Gravis.
—Internuclear ophthalmoplegia: See 10.13, Internuclear Ophthalmoplegia.
6. Dissociated Horizontal Deviation: A change in horizontal ocular alignment caused by a change in the balance of visual input from the two eyes. Not related to accommodation. Seen clinically as a spontaneous unilateral exodeviation or an exodeviation of greater magnitude in one eye during prism and alternate cover testing.
7. Orbital disease (e.g., tumor, idiopathic orbital inflammatory syndrome): Proptosis and restriction of ocular motility are usually evident. See 7.1, Orbital Disease.
8. Isolated convergence insufficiency: Usually occurs in patients >10 years. Blurred near vision, asthenopia, or diplopia when reading. An exophoria at near fixation, but straight or small exophoria at distance fixation. Must be differentiated from convergence paralysis. See 13.4, Convergence Insufficiency.
9. Convergence paralysis: Similar to convergence insufficiency, but with a relatively acute onset, an exotropia at near, and an inability to overcome base out prism. Often secondary to an intracranial lesion.
Work-Up
1. Evaluate visual acuity of each eye, with correction and pinhole, to evaluate for amblyopia. Color vision and stereopsis.
2. Perform motility examination; observing for restricted eye movements or signs of Duane syndrome.
3. Measure the exodeviation in all cardinal fields of gaze at distance and in primary position (straight ahead) at near, using prisms. See Appendix 3, Cover/Uncover and Alternate Cover Tests.
4. Perform pupillary, slit-lamp, and fundus examinations; check for causes of sensory deprivation (if poor vision).
5. Refraction (cycloplegic or manifest depending on age of the patient).
6. Consider work-up for myasthenia gravis when suspected by evidence of fatigability. See 10.11, Myasthenia Gravis.
7. Consider a CT scan (axial and coronal views) or an MRI of the orbit and brain, when orbital or neurologic disease is suspected.
Treatment
In all cases, correct significant refractive errors and treat amblyopia. See 8.7, Amblyopia.
1. Exophoria:
—No treatment necessary unless it progresses to intermittent exotropia.
2. Intermittent exotropia:
—Phase 1: Follow patient closely.
—Phase 2: Muscle surgery may be considered to maintain normal binocular vision.
—Phase 3: Muscle surgery is often indicated at this point. Bifixation or peripheral fusion can occasionally be attained.
3. Sensory-deprivation exotropia:
—Correct the underlying cause, if possible.
—Treat any amblyopia.
—Muscle surgery may be performed for manifest exotropia.
—When one eye has very poor vision, protective glasses (polycarbonate lens glasses) should be worn at all times to protect the good eye.
4. Congenital exotropia:
—Muscle surgery early in life, as in patients with congenital esotropia.
5. Consecutive exotropia:
—Additional muscle surgery may be considered.
—Prism correction in glasses can be used.
—Over-minus or under-plus correction can stimulate accommodative convergence.
6. Dissociated horizontal deviation:
—Muscle surgery may be considered.
7. Duane syndrome: See 8.6, Strabismus Syndromes.
8. Third nerve palsy: See 10.5, Isolated Third Nerve Palsy.
9. Convergence insufficiency: See 13.4, Convergence Insufficiency.
10. Convergence paralysis:
—Base-in prisms at near to alleviate diplopia.
—Plus lenses if accommodation is also weakened.
Follow-Up
1. If amblyopia is present, see 8.7, Amblyopia.
2. If no amblyopia is present, then reexamine every 4 to 6 months. The parents and patient are asked to return sooner if the deviation increases, becomes more frequent, stays out longer, or patient begins to close one eye.
8.6 STRABISMUS SYNDROMES
Motility disorders that demonstrate typical features of a particular syndrome.
Syndromes
 Duane syndrome: A congenital motility disorder, usually unilateral (85%), characterized by limited abduction, limited adduction, or both. The globe retracts and the eyelid fissure narrows on adduction. In unilateral cases, the strabismus will be incomitant and the patient will often adopt a face-turn to allow them to use both eyes together. Classified into three types:
Duane syndrome: A congenital motility disorder, usually unilateral (85%), characterized by limited abduction, limited adduction, or both. The globe retracts and the eyelid fissure narrows on adduction. In unilateral cases, the strabismus will be incomitant and the patient will often adopt a face-turn to allow them to use both eyes together. Classified into three types:
—Type 1 (most common): Limited abduction. Primary position frequently esotropia. In unilateral cases, nearly always with face turn toward affected side.
—Type 2: Limited adduction. Primary position usually exotropia. In unilateral cases, often with face turn away from affected side.
—Type 3: Limited abduction and adduction. Esotropia, exotropia, or no primary position deviation. Significant globe retraction.
 Brown syndrome: A motility disorder characterized by limitation of elevation in adduction. Elevation in abduction is normal. Typically, eyes are aligned in primary gaze. Usually congenital, but may be acquired secondary to trauma, surgery, or inflammation in the area of the trochlea, or idiopathic. Bilateral in 10% of patients.
Brown syndrome: A motility disorder characterized by limitation of elevation in adduction. Elevation in abduction is normal. Typically, eyes are aligned in primary gaze. Usually congenital, but may be acquired secondary to trauma, surgery, or inflammation in the area of the trochlea, or idiopathic. Bilateral in 10% of patients.
 Monocular elevation deficiency (double-elevator palsy): Congenital. Unilateral limitation of elevation in all fields of gaze secondary to restriction of the inferior rectus or paresis of the inferior oblique and/or superior rectus. There may be hypotropia of the involved eye that increases in upgaze. Ptosis or pseudoptosis may be present in primary gaze. The patient may assume a chin-up position to maintain fusion if a hypotropia in primary gaze is present.
Monocular elevation deficiency (double-elevator palsy): Congenital. Unilateral limitation of elevation in all fields of gaze secondary to restriction of the inferior rectus or paresis of the inferior oblique and/or superior rectus. There may be hypotropia of the involved eye that increases in upgaze. Ptosis or pseudoptosis may be present in primary gaze. The patient may assume a chin-up position to maintain fusion if a hypotropia in primary gaze is present.
 Möbius syndrome: Rare congenital condition associated with both sixth and seventh nerve palsies. Esotropia is usually present. Limitation of abduction and/or adduction, with a unilateral or bilateral, partial or complete facial nerve palsy. Other cranial nerve palsies as well as deformities of the limbs, chest, and tongue may occur.
Möbius syndrome: Rare congenital condition associated with both sixth and seventh nerve palsies. Esotropia is usually present. Limitation of abduction and/or adduction, with a unilateral or bilateral, partial or complete facial nerve palsy. Other cranial nerve palsies as well as deformities of the limbs, chest, and tongue may occur.
 Congenital fibrosis syndrome: Congenital group of disorders with restriction and fibrous replacement of the extraocular muscles. Usually involves all of the extraocular muscles with total external ophthalmoplegia and ptosis. Usually both eyes are directed downward, so the patient assumes a chin-up position to see. Often autosomal dominant.
Congenital fibrosis syndrome: Congenital group of disorders with restriction and fibrous replacement of the extraocular muscles. Usually involves all of the extraocular muscles with total external ophthalmoplegia and ptosis. Usually both eyes are directed downward, so the patient assumes a chin-up position to see. Often autosomal dominant.
Work-Up
1. History: Age of onset? History of trauma? Family history? History of other ocular or systemic diseases?
2. Complete ophthalmic examination, including alignment in all fields of gaze. Note head position. Look for retraction of globe and narrowing of interpalpebral fissure in adduction (common in Duane syndrome).
3. Pertinent physical examination, including cranial nerve evaluation.
4. Radiologic studies (e.g., MRI or CT scan) may be indicated for acquired, atypical, or progressive motility disturbances.
5. Forced-duction testing is used to differentiate the two etiologies of monocular elevation deficiency (test will be positive with inferior rectus fibrosis and negative with superior rectus and inferior oblique paresis). Forced ductions can also confirm the diagnosis of Brown syndrome.
Treatment
1. Treatment is usually indicated for a cosmetically significant abnormal head position, or if a significant horizontal or vertical deviation exists in primary gaze.
2. Surgery, when indicated, depends on the particular motility disorder, extraocular muscle function, and the degree of abnormal head position.
Follow-Up
Follow-up depends on the condition or conditions being treated.
8.7 AMBLYOPIA
Symptoms
Usually none. Usually found when decreased vision is detected by vision testing in each eye. A history of patching, strabismus, or muscle surgery as a child may be elicited.
NOTE: Amblyopia occasionally occurs bilaterally as a result of bilateral visual deprivation (e.g., congenital cataracts not treated within the first months of life).
Signs
Critical. Poorer vision in one eye that is not improved with refraction and not entirely explained by an organic lesion. The involved eye nearly always has a higher refractive error. The decrease in vision develops during the first several years of life. Central vision is primarily affected, while the peripheral visual field usually remains normal.
Other. Individual letters are more easily read than a full line (crowding phenomenon). In reduced illumination, the visual acuity of an amblyopic eye is reduced much less than an organically diseased eye (neutral-density filter effect).
NOTE: Amblyopia, when severe, may cause a trace relative afferent pupillary defect. Care must be taken to be sure that the light is directed along the same axis in each eye, particularly in patients with strabismus. Directing the light off-axis may result in a false-positive result.
Etiology
 Strabismus: Most common form (along with anisometropia). The eyes are misaligned. Vision is worse in the consistently deviating, nonfixating eye. Strabismus can lead to or be the result of amblyopia.
Strabismus: Most common form (along with anisometropia). The eyes are misaligned. Vision is worse in the consistently deviating, nonfixating eye. Strabismus can lead to or be the result of amblyopia.
 Anisometropia: Most common form (along with strabismus). A large difference in refractive error (usually ≥1.50 diopters) between the two eyes. Can be seen in cases of eyelid hemangioma or congenital ptosis inducing astigmatism.
Anisometropia: Most common form (along with strabismus). A large difference in refractive error (usually ≥1.50 diopters) between the two eyes. Can be seen in cases of eyelid hemangioma or congenital ptosis inducing astigmatism.
 Media opacity: A unilateral cataract, corneal scar, or PFV/PHPV may cause a preference for the other eye and thereby cause amblyopia.
Media opacity: A unilateral cataract, corneal scar, or PFV/PHPV may cause a preference for the other eye and thereby cause amblyopia.
 Occlusion: Amblyopia that occurs in the fellow eye as a result of too much patching or excessive use of atropine. Prevented by examining at appropriate intervals (1 week per year of age), patching part-time, or using the full cycloplegic refraction when using atropine.
Occlusion: Amblyopia that occurs in the fellow eye as a result of too much patching or excessive use of atropine. Prevented by examining at appropriate intervals (1 week per year of age), patching part-time, or using the full cycloplegic refraction when using atropine.
Work-Up
1. History: Eye problem in childhood, such as misaligned eyes, patching, or muscle surgery?
2. Ocular examination to rule out an organic cause for the reduced vision.
3. Cover–uncover test to evaluate eye alignment. See Appendix 3, Cover/Uncover and Alternate Cover Tests.
4. Refraction; cycloplegic in children too young to cooperate.
Treatment
1. Patients younger than 12 years:
—Appropriate spectacle correction (full cycloplegic refraction or reduce the hyperopia in both eyes symmetrically ≥1.50 diopters). If vision remains reduced after period of refractive adaptation (6 to 12 weeks), begin patching or penalization of fellow eye.
—Patching: Patch the eye with better corrected vision 2 to 6 hours/day for 1 week per year of age (e.g., 3 weeks for a 3-year-old), with at least 1 hour of near activity. Adhesive patches placed directly over the eye are most effective. A patch can be worn over the glasses as long as the child does not peek around the patch. If a patch causes local irritation, use tincture of benzoin on the skin before applying the patch and use warm water compresses on the patch before removal.
—Penalization with atropine: Atropine 1% once daily (used with glasses) has been shown to be equally effective as patching in mild-to-moderate amblyopia (20/100 or better). If vision does not improve, the effect of the atropine can be increased by removing the hyperopic lens from the glasses of the nonamblyopic eye. If the child is experiencing difficulty with school work with the use of atropine, he/she can wear full hyperopic correction with a +2.50 bifocal during school.
—Optical degradation: Use a high plus lens (e.g., +9.00 diopters or an aphakic contact lens) to blur the image. If the child is highly myopic, may remove the minus lens from the preferred eye.
 Continue patching until the vision is equalized or shows no improvement after three compliant cycles of patching. If a recurrence of amblyopia is likely, use part-time patching to maintain improved vision.
Continue patching until the vision is equalized or shows no improvement after three compliant cycles of patching. If a recurrence of amblyopia is likely, use part-time patching to maintain improved vision.
 If occlusion amblyopia (a decrease in vision in the patched eye) develops, patch the opposite eye for a short period (e.g., 1 day per year of age), and repeat the examination.
If occlusion amblyopia (a decrease in vision in the patched eye) develops, patch the opposite eye for a short period (e.g., 1 day per year of age), and repeat the examination.
—In strabismic amblyopia, delay strabismus surgery until the vision in the two eyes is equal, or maximal vision has been obtained in the amblyopic eye.
2. If treatment of amblyopia fails or patient presents outside of treatment age range, protective glasses should be worn to prevent accidental injury to the nonamblyopic eye. Any child who does not have vision improved to at least 20/40 needs to wear eye protection during sports (one-eyed athlete rule).
3. Treatment of media opacity: Remove the media opacity and begin patching the nonamblyopic eye.
4. Treatment of anisometropic amblyopia: Give the appropriate spectacle correction at the youngest age possible. If vision remains reduced after period of refractive adaptation (6 to 12 weeks), begin patching or penalization of fellow eye.
8.8 CONGENITAL CATARACT
Signs
(See Figure 8.8.1.)
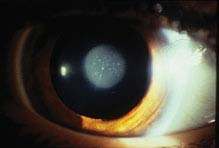
FIGURE 8.8.1. Congenital nuclear cataract.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


