Chapter 27 Pediatric Thyroid Cancer
Introduction
Much like many childhood diseases, there are major differences in the treatment of pediatric patients with thyroid cancers when compared to their adult counterparts. Although there is still much to be learned, we do know that a child with pediatric thyroid cancer cannot be treated “just like an adult.” This chapter highlights some of those differences while discussing modern treatment of pediatric thyroid cancer. Thyroid cancer accounts for approximately 1% to 3% of all childhood malignancies and 7% of all pediatric head and neck tumors.1,2 Childhood thyroid cancer is a rare disease; it is important to recognize that even the largest series includes only a few hundred cases that took more than three decades to accumulate.3 Over ninety percent of childhood thyroid carcinomas are papillary thyroid cancer (PTC) and its follicular variant (FVPTC), and therefore much of our discussion in this chapter will focus on childhood PTC.4 The male/female ratio of thyroid carcinoma varies with age: 6:1 for ages 5 to 9 years; 1:1 for ages 10 to 14 years; and 2:5 for ages 15 to 19 years; the peak incidence is between the ages of 15 and 19.1,5
Etiology
The association between thyroid cancer and prior radiation exposure has been well documented for decades. Historical data illustrated an epidemiologic causal relationship between radiation exposure and the development of PTC (see Chapter 28, Chernobyl and Radiation-Induced Thyroid Cancer).6 Radiation was previously given as treatment for benign diseases such as enlarged tonsils or thymic glands and acne, and up until the 1970s, most children with thyroid malignancy had a prior history of radiation exposure.7,8 A number of studies on residents of the Marshall Islands, Hiroshima, and Nagasaki and the long-term outcome study by the Michael Reese Hospital (Chicago) have looked specifically at the increased susceptibility to thyroid cancer from external irradiation in the pediatric population.9,10 These children often presented with locally advanced disease with a high rate of metastases. Since the early 1970s when radiation use as a mode of therapy decreased, so did the incidence of childhood thyroid cancer. However, the 1986 Chernobyl nuclear power plant disaster was a powerful reminder of how devastating to children radiation exposure can be in regard to childhood thyroid malignancy. The average dose in children around the power plant was 2.1 to 4.7 Gy.11 After this nuclear fallout, the incidence of thyroid cancer increased by a factor of more than 60 in local countries, such as Belarus. Furthermore, these tumors unfortunately behaved aggressively with more advanced disease. Regional and distant metastases were present in 57% and 15%, respectively.11 Other thyroid conditions including nodular goiter, hyperplasia, and chronic thyroiditis were also overrepresented. Another important observation in children has been the latency period between radiation exposure and the onset of malignancy. Although latency periods of 8 to 11 years have been traditional time lines in children, periods as brief as 3 years have been documented in the Chernobyl studies and the Michael Reese Hospital study.12
Other known risk factors for thyroid cancer include Hodgkin’s lymphoma, Graves’ disease, Hashimoto’s thyroiditis, autonomous nodules, endemic goiter, and therapeutic doses of 131I.13,14 Increasing data have linked treatment of other childhood malignancies with chemotherapeutics, such as alkylating agents, as a risk factor for developing thyroid cancer decades later.15 We stress this point, as we anticipate an increasing number of secondary malignancies including thyroid cancer with the excellent modern outcomes seen in children with leukemias and lymphomas.16 With advances in pediatric oncology, more children today than ever before are receiving aggressive, but appropriate, chemotherapeutic agents and are subsequently living well into adulthood; attention should be given to this risk factor by clinicians as more data on this subject accumulate.
Pathology
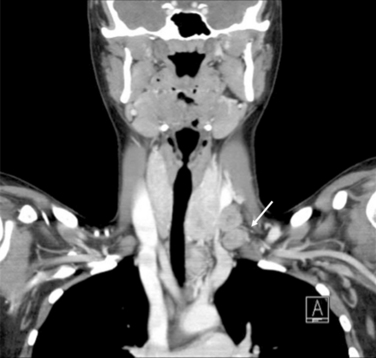
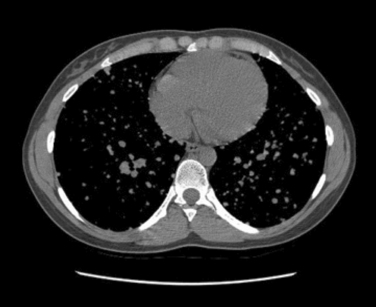
The most common histologic subtype of childhood thyroid cancer is papillary thyroid cancer and its follicular variant (> 90%) (see Chapter 44, Surgical Pathology of the Thyroid Gland). Other childhood thyroid cancers are rare and include pure follicular thyroid cancer (3% to 5%), Hurthle cell tumors (< 1%), medullary thyroid carcinoma (5%), anaplastic carcinoma (< 1%), thyroid sarcomas (< 1%), and lymphoma (< 1%). PTC usually presents as a firm, partially encapsulated or nonencapsulated mass. Its histologic features are not significantly different than adult PTC. However, PTC in childhood often presents with more extracapsular thyroid invasion than in adults. PTC can directly invade local structures such as the esophagus, trachea, recurrent laryngeal nerve, and blood vessels, creating significant local morbidity (Figure 27-1). Childhood PTC often invades lymphatics, giving rise to multifocal PTC through intraglandular spread about 30% to 80% of the time.1 There is also a high incidence of regional nodal metastases, up to 70% to 90% (Figure 27-2). Centers with experience in treating childhood PTC have learned to meticulously look for lymph node metastasis with the aid of preoperative ultrasound and computed tomography (CT).17 PTC in children more often displays distant spread (lungs); however, importantly, it causes fewer deaths from PTC than comparably staged adult patients. In fact, the Mayo Clinic group and others have demonstrated that children with PTC and pulmonary metastasis have a much lower cause-specific mortality than adults over the age of 45 (Figure 27-3).18
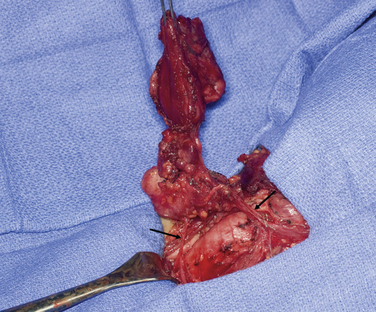
Figure 27-1 Left thyroid lobe with metastatic nodes encasing recurrent laryngeal nerve (RLN). Arrows point to RLN.
FVPTC represents a significant portion of PTC seen in children (20% to 25%).19 These tumors are more likely to appear encapsulated, and some authorities have questioned whether older studies diagnosed these lesions as follicular carcinoma.1,20 Therapy of FVPTC is similar to that for PTC. Another important subtype of PTC is the diffuse sclerosing variant of PTC that causes goitrous enlargement with replacement of both lobes by hard, calcified tumor. It tends to be a more aggressive form of the disease affecting children, teenagers, and younger adults. In one series, there was a 100% incidence of nodal metastases at diagnosis with a 25% incidence of subsequent pulmonary involvement.21 Other less common but aggressive subtypes that occur in children are tall cell, columnar cell, and solid (poorly differentiated) variants, all of which have a worse prognosis (see Chapter 44, Surgical Pathology of the Thyroid Gland).
Pure follicular carcinoma and its Hurthle cell variant, like PTC, are both well-differentiated cancers thought to be increased by radiation exposure.22 They are rare in children, and features such as microscopic capsular or vascular invasion determine the minimally invasive subtype. The widely invasive subtype is easily diagnosed on gross inspection. Although metastasis to lymph nodes is rare, metastasis to lungs and bone via a hematological route are more commonly seen in the widely invasive subtype. Childhood follicular tumors are usually of the minimally invasive type, are smaller than 4 cm, and are generally associated with extremely low recurrence and mortality rates.
Medullary thyroid carcinoma (MTC), unlike thyroid cancers of follicular cell origin, originates from the parafollicular cells that secrete calcitonin (see Chapters 23, Sporadic Medullary Thyroid Carcinoma, 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, and 25, Sporadic Medullary Thyroid Microcarcinoma). These tumors are derived from neural crest cells, and genetic mutations have strong implications not only for childhood MTC treatment but for other disorders of neural crest origin with genetic mutations.
Genetics
Quantitative and perhaps qualitative differences in genetic mutations and growth factor expression patterns exist in childhood thyroid carcinomas compared with those of adults (see Chapter 17, Molecular Pathogenesis of Thyroid Neoplasia).23 MTC is an inherited autosomal dominant disease and has become the most commonly recognized hereditary thyroid cancer. Although genetic aspects of MTC are covered in detail in other chapters in this book, features related to pediatric MTC are discussed here (see Chapters 23, Sporadic Medullary Thyroid Carcinoma, 24, Syndromic Medullary Thyroid Carcinoma: MEN 2A and MEN 2B, and 25, Sporadic Medullary Thyroid Microcarcinoma). MTC can be associated with familial MTC and multiple endocrine neoplasia (MEN) types 2A and 2B. Two additional variants have been identified, which include Hirschsprung’s disease and cutaneous lichen amyloidosis.24 Overall, MTC has a hereditary or associated familial syndrome in up to one third of cases. The gene that is associated with MEN 2 is the rearranged (during transfection) (RET) proto-oncogene on chromosome 10. It is a tyrosine kinase receptor protein and is involved in the control, survival, proliferation, differentiation, and migration of the enteric nervous system progenitor cells. RET is now known to be the common etiologic factor linking Hirschsprung’s disease and MEN 2 syndromes.25 Mutations in the RET proto-oncogene may cause either a loss of function causing Hirschsprung’s disease or a gain of function causing MEN 2. Although oncogenic RET mutations may vary between different population groups, RET analysis in MEN has revolutionized the management of children with MEN 2. The ability to accurately predict the risk of MTC by genetic RET proto-oncogene analysis now exists. Codon-risk management protocols have been used to guide therapy; specifically, the C634 and M918T mutations (associated with MEN 2A and MEN 2B, respectively) are particularly associated with early aggressive disease and distant metastasis requiring prophylactic thyroidectomy as early as one year of age.
It has become well documented that genetic factors play an important role in the development of thyroid malignancy. Familial nonmedullary thyroid cancer (FNMTC), which includes cases with two or more first-degree relatives diagnosed with nonmedullary thyroid cancer, occurs in approximately 5% of all thyroid cancer cases (see Chapter 29, Familial Nonmedullary Thyroid Cancer).25 FNMTC may also occur as a component of familial cancer syndromes that are of particular interest for clinicians caring for pediatric patients. Gardner’s syndrome (APC), Cowden’s disease (PTEN), Carney complex type 1 (PRKAR1a), Werner syndrome (WRN), and McCune-Albright syndrome (GNAS1) have been associated with FNMTC.26 FNMTC is transmitted in an autosomal dominant pattern of inheritance with incomplete penetrance. Approximately 90% of FNMTC cases are PTC. Today, the specific gene or genes have not yet been identified for nonsyndromic FNMTC; however, linkage analysis studies have identified chromosomal loci that are associated with the condition including chromosomal loci 1q21, 2q21, and 19p13.2. Multiple investigators have excluded RET, PTEN, MET, APC, and MNG1 as candidate genes for FNMTC. Many studies suggest that FNMTC is more aggressive than sporadic differentiated thyroid cancer and the outcome of the nonindex cases of FNMTC appear to be better.27 Therefore, total or near-total thyroidectomy with a prophylactic central neck lymph node dissection appears to be appropriate surgical treatment for patients diagnosed with FNMTC.26 This would include younger patients who have positive findings either on screening or genetic testing. Uchino et al. screened patients as young as 3 years old.28 As we learn more about the genetics of FNMTC, pediatric endocrine surgeons should always elicit a detailed family history and document this recently described entity (see Chapter 29, Familial Nonmedullary Thyroid Cancer).
FNMTC is not to be confused with sporadic nonmedullary thyroid carcinomas whose genetic factors are also being elucidated. At least 12 different types of RET rearrangements have been identified in sporadic and radiation-induced PTCs.24 These are somatic mutations that have led to the activation of RET in the follicular cells of the thyroid, which may cause well-differentiated malignancies.
Presentation
Evaluation
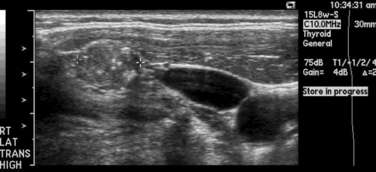
Although early studies have reported a high prevalence of malignant disease among thyroid nodules in children (30% to 50%), more recent studies demonstrate a smaller risk of malignancy in solitary thyroid nodules in children approximating 20% to 30%.29,30 Thyroid nodules, in any child, irrespective of nodule size, growth, or number present, should be considered malignant until proved otherwise. It should also be noted that management of cystic lesions in children is controversial, as cysts may harbor occult PTC. Although diagnosis and workup of nodules is beyond the scope of this chapter, we will discuss certain aspects unique to children, in particular the use of fine-needle aspiration (FNA) (see Chapters 11, The Evaluation and Management of Thyroid Nodules, 12, Fine-Needle Aspiration of the Thyroid Gland, and 13, Ultrasound of the Thyroid and Parathyroid Glands).30,31 It should be stated that the use of ultrasound (US) in experienced hands is very useful in identifying nonpalpable nodes in the lateral neck compartments (Figure 27-4). It is so useful in planning the extent of the lymph node dissection; we recommend preoperative US of the contralateral lobe and bilateral cervical nodes for all patients undergoing thyroidectomy for cancer. Other modalities, such as preoperative nuclear scintigraphy, have been mostly abandoned unless evaluating the patient with nodular disease in the setting of a suppressed serum thyroid-stimulating hormone (TSH) level. Also, a chest x-ray should be routinely obtained. Noncontrast CT can demonstrate micronodular and interstitial patterns of pulmonary metastatic spread. If there is concern by palpation or US for invasion of the upper aerodigestive tract or major vascular structures, magnetic resonance imaging (MRI) or CT is also useful for evaluating the neck and mediastinum. Positron emission tomography (PET) as a diagnostic tool in thyroid disease has not been adequately studied in children.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree