Pathology of the Optic Nerve
Diva R. Salomão
Caterina Giannini
Introduction
The optic nerve, second cranial nerve, is approximately 50 mm long. It extends from the optic nerve disc inside the globe to the optic chiasm in the intracranial cavity where it is continuous with the optic tract (Fig. 16.1). The optic nerve is composed primarily of the axons of the ganglion cells in the retina.
Embryology and Development
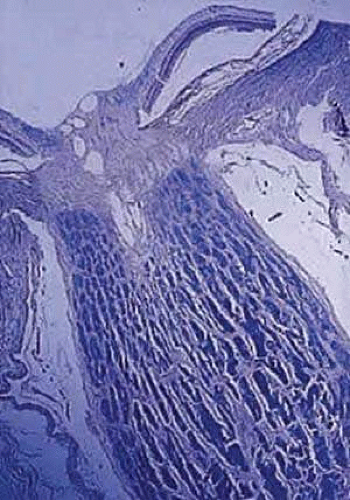
The optic stalk develops from the optic vesicle around the seventh week of gestation. The number of axons rapidly increases during early embryogenesis to reach a peak of about 3 million axons during the second trimester, after which fiber loss occurs until about 1 million fibers remain at birth.1 The diameter of the optic nerve increases throughout embryogenesis—from about 0.15 mm at the ninth week of gestation until full term, when it averages 2.7 mm.2 Myelination by oligodendroglial cells begins near the optic chiasm, at about the 30th week of gestation, and extends in the direction of the lamina cribrosa. Myelination of the optic nerve is complete at or soon after birth (Fig. 16.2).2
Normal Anatomy and Histology
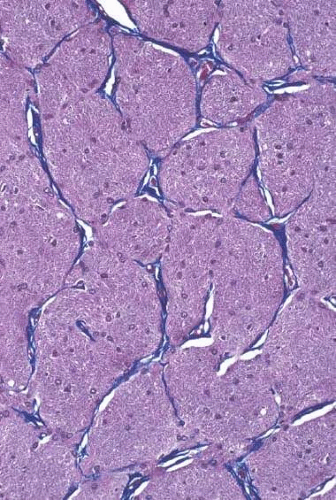
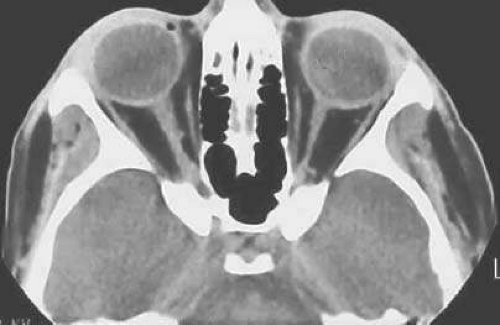
The adult human optic nerve has a cross-sectional area of about 9 mm2 (Fig. 16.3), and it measures about 50 mm in length from the back of the eye to the optic chiasm. The optic nerve is divided into three segments: intraocular, intraorbital, and intracanalicular. The intraocular portion of the nerve, about 0.7 mm in length, has a prelaminar and a laminar portion.
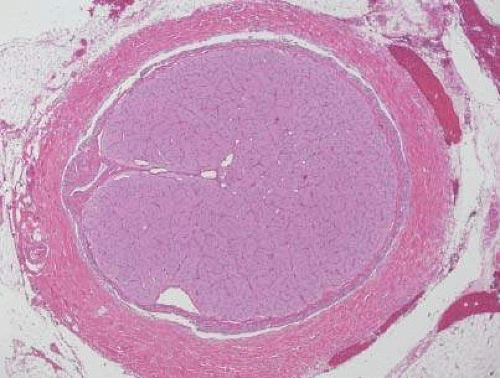 Figure 16.3. Cross-section of normal optic nerve showing thin pial strands dividing nerve into fascicles. Meningeal sheath is closely applied to optic nerve (hematoxylin and eosin staining). |
Axons from the retinal ganglion cells exit the eye through the optic disc, which is 1.5 mm in diameter. The physiologic cup is a small disc-shaped depression the edges of which are parallel to those of the optic disc. It lies slightly temporal to the disc center and is lined by glial tissue (tissue of Kuhnt). The nerve exits the globe slightly nasal to and above the posterior pole. The prelaminar portion of the nerve, anterior to the lamina cribrosa, consists of nonmyelinated nerve fibers and astrocytes. The scleral lamina or lamina cribrosa is composed of astrocytes, collagen, and vessels.
In the adult, the normal optic nerve contains about 1 million nerve fibers.3,4 No difference in nerve-fiber counts was observed between male and female gender, but it seems to exist between right and left eyes. Axon density is higher in the inferotemporal segment of the nerve because a major portion of the papillomacular bundle exits through this quadrant. Axonal diameter varies considerably (from 0.3 to 5 μm) within individual optic nerves and on average is smaller in the temporal and inner regions of the nerve when compared to the nasal and outer areas.5 The optic nerve fibers are arranged in fascicles.6 These fascicle are composed of the nerve fibers in addition to oligodendrocytes, astrocytes, and microglia. The number of fascicles varies only slightly between individuals but changes substantially along the length of the nerve. Immediately behind the globe and in the region of the optic canal, there are about 200 to 300 fascicles; in contrast, in the mid-orbital segment there are fewer than 120 fascicles. This variation maximizes compliance and tensile strength throughout the nerve.
Anteriorly, the axons of the optic nerve traverse the lamina cribrosa, a sieve-like portion of connective tissue in the sclera; where it appears to be some degree of constriction.7 The course of the axons through the lamina cribrosa varies in different areas and is longer and more tortuous peripherally than centrally.8
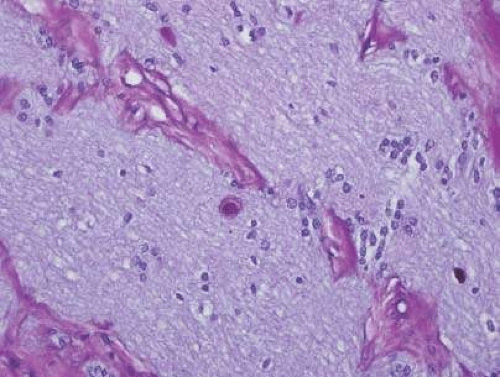
Central vessels penetrate the optic nerve approximately 8 to 15 mm behind the globe, after crossing the meninges nasal to the nerve (Fig. 16.4). The vein follows a course of variable length before entering the nerve. The artery penetrates the nerve at the same level or anterior to the vein. The arrangement of arterial branches within the nerve varies between individuals. The branches are from the ophthalmic, posterior ciliary, central retinal artery (within orbit), or from branches from the circle of Zinn-Haller. The intraocular portion of the nerve receives branches from the intracranial portion of the ophthalmic artery, anterior cerebral, internal carotid, and ophthalmic arteries.9,10
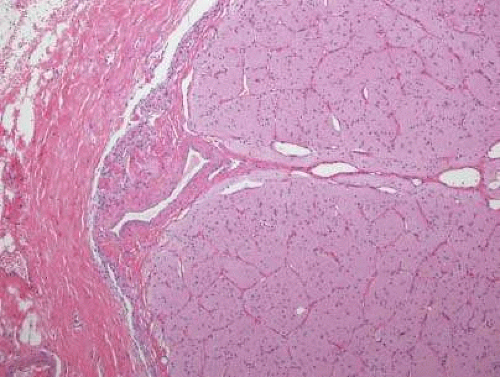 Figure 16.4. High power of normal optic nerve showing vessels entering the nerve (hematoxylin and eosin). |
The optic nerve is invested by a sheath that is continuous posteriorly with the intracranial dura mater and the periorbita in the orbital apex; anteriorly, it fuses with the sclera. The arachnoid is composed of loose connective tissue continuous with that of the cranium. Meningothelial cells (arachnoid cells) may be found in nests, some of which contain calcified bodies (psammoma bodies). The subarachnoid space is continuous with the intracranial subarachnoid space. Investing the nerve closely is the pia, which sends fibrovascular septa into the nerve, dividing it into fascicles (Fig. 16.5). The pia blends with the lamina cribrosa and the connective tissue around the central vessels, so that the central retinal artery and vein have a common connective tissue sheath. The optic nerve sheath, immediately behind the eye, is typically somewhat loose.
Aging Changes
Most studies show that the number of axons within the optic nerve decreases with age at a rate of about 5,000 axons per year, even in the absence of clinical eye disease.4,5,11,12 Whether this is a steady decrease throughout life or starts during adulthood is unclear. Accompanying this loss of axons is an increase in the proportion of the nerve occupied by connective tissue.13 Also noted with advancing age are increased corpora amylacea and lipofuscin deposits within astrocytes14 and an increase in the thickness of the lamina cribrosa. This latter finding might be of relevance in the development of glaucoma.
Corpora amylacea are small (about 10 μm) homogeneous or laminated structures that are frequently found in the optic nerve (Fig. 16.6), retina, brain, and spinal cord. These periodic acid-Schiff (PAS)-positive structures represent filamentous tangles of fibrils associated with microtubules, ultrastructurally.15 Corpora amylacea increase in number with age and decrease in conditions associated with neuronal loss (e.g., glaucoma), which suggests that they represent intraneuronal aging products.13 The immunoreactivity for tau-2 protein, which is present in neurofibrillary tangles and plaques in various neurodegenerative disorders, and also the presence of kynurenine aminotransferases, seen by KAT II immunoreactivity on those, support this idea.16,17
Anomalies and Congenital Malformations
Aplasia of the optic nerve is rare congenital anomaly, characterized by an absence of the optic nerve and disc, retinal ganglion cells, nerve fibers, and retinal blood vessels, which are replaced by fibrous and glial tissue, with capillaries and lobules of adipose tissue.18 It is more often an unilateral anomaly;19 however, bilateral cases have been described.20,21 This anomaly might be associated with other ocular or central nervous system anomalies.22
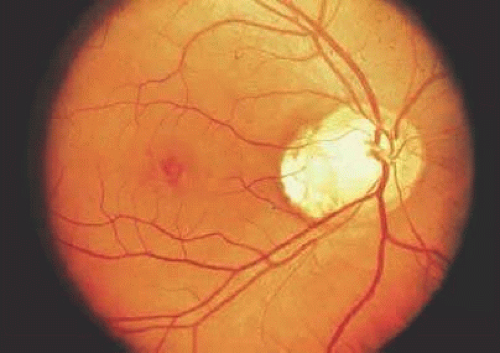
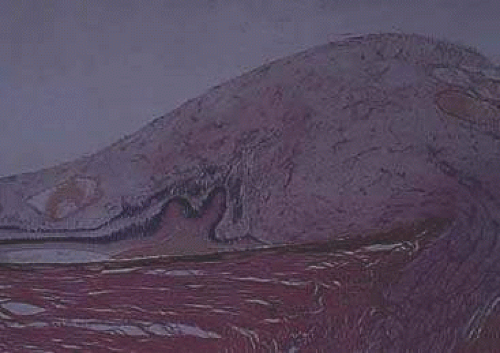
Hypoplasia of the optic nerve is also rare although more common than aplasia (Figs. 16.7 and 16.8). It is increasingly recognized as a significant cause of visual deficit in children. Congenital nerve hypoplasia is most commonly a nonprogressive and nonfamilial condition associated with a reduction in the number of retinal ganglion cells and axons. It may be either unilateral or more commonly bilateral and may involve the entire nerve or be segmental. Although it may be an isolated abnormality, it is frequently associated with other anomalies—both ocular and nonocular.23,24 Additional ocular abnormalities include microphthalmos and congenital nonattachment of the retina. The most common nonocular abnormalities associated with congenital optic nerve hypoplasia are midline central nervous system (CNS) structural defects, including septo-optic dysplasia, corpus callosum, and septum pellucidum abnormalities and ectopia of the posterior pituitary, resulting in diabetes insipidus.25 In optic nerve hypoplasia, the optic disc appears grayish and is about half its normal size. Retinal vessels are present. The optic foramen is often small. Clinically, there may be associated strabismus, nystagmus, and decreased visual acuity. Optic nerve hypoplasia has been related to fetal alcohol syndrome and other maternal substance abuse.26
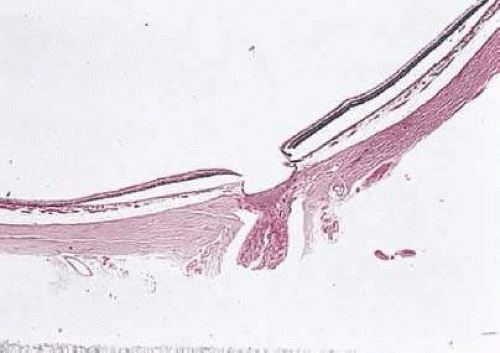 Figure 16.7. Hypoplasia of the optic nerve. Note lack of formation of optic disc tissue (hematoxylin and eosin staining). |
Coloboma results from complete or partial nonclosure of the most anterior portion of the optic stalk. (Fig. 16.9). It may be sporadic or inherited with an autosomal-dominant pattern, an isolated defect or associated with other congenital abnormalities.27 An association with Pax2 gene mutation has been recently described as part of renal-coloboma syndrome.28,29 Colobomas are usually unilateral and vary in appearance from a deep physiologic cup to a large hole with a retrobulbar cyst. Microscopically, a coloboma is characterized by missing portions of the choroidal layer and retinal pigment epithelium with gliosis of the retina in some areas. The wall of the defect may contain adipose tissue and occasionally muscle fibers. The latter may lead to a contractile peripapillary staphyloma. Ocular anomalies, such as microphthalmos with cyst, lenticonus and persistent hyaloid arteries may be associated.30 Macular changes consisting of hemorrhages, cysts, holes, or retinal detachment are present in as many as half of affected patients.31,32
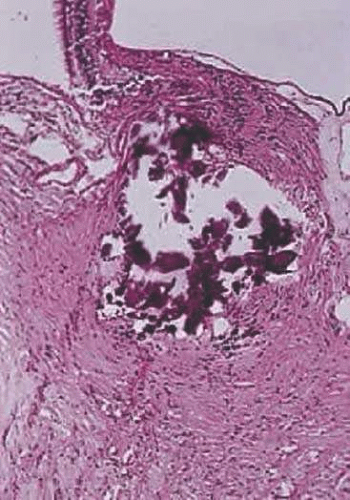
Optic pit, often referred to as an “atypical coloboma” because of its position inferolaterally, represents a small partial coloboma (Fig. 16.10). It consists of a unilateral anomalous development of the optic nerve papilla. It is an outpouching of neuroectoderm surrounded by a connective tissue capsule that passes through a defect in the lamina cribrosa into the subarachnoid space. Occasionally, the affected disc is larger than the contralateral disc.
The myopic disc is oriented obliquely to the horizontal meridian and is raised nasally. Temporally, it appears flattened and has a white scleral crescent. Microscopically, the nasal disc overlaps the neural retina, retinal pigment epithelium (RPE), Bruch membrane, and choroid. Temporally, the RPE and Bruch membrane do not extend to the disc margin.
Meningocele is a saccular dilation of the optic nerve sheath. Optic sheath meningocele is rare, with approximately 30 cases described in the literature.33 The term should be used to describe primary CSF cysts of the optical nerve sheath without apical mass or malformation of the cranial orbital junction. Patients present with headaches, visual complaints, or both.34 Patients with neurofibromatosis type 1 (NF-1) have an increased incidence of meningocele, as isolated finding or in association with optic nerve gliomas.35,36,37
Optic Nerve Head Drusen
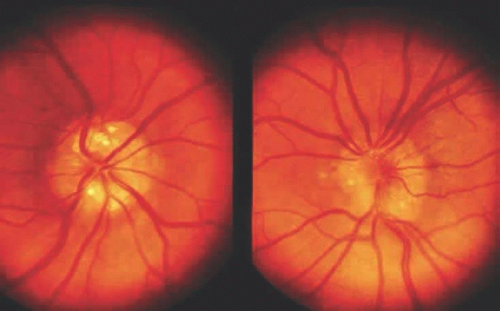
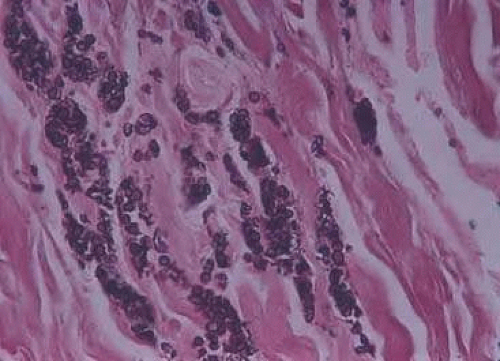
Optic nerve drusen are congenital and developmental anomalies of the optic nerve head commonly seen in clinical practice, often as an incidental finding.38 These have been found in 0.3% of the population with up to 91% presenting bilaterally.39 Drusen are a form of calcific degeneration that may be present pre- or postlamina cribrosa (Fig. 16.11). Those that are anterior to the lamina are usually bilateral and thought to result from mitochondrial calcification within axons, causing disturbance in axoplasmic flow. Drusen may be idiopathic or occasionally inherited in an autosomal-dominant pattern. The presence of drusen may cause enlargement of the papilla and be mistaken for papilledema (Fig. 16.12). The giant drusen of tuberous sclerosis are calcified astrocytic hamartomas (Fig. 16.13). Although benign and usually asymptomatic, an association with hypertension and vascular occlusion has also been noted.40
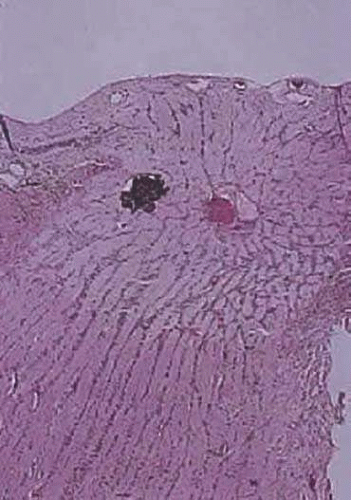 Figure 16.11. Calcified drusen is present in the optic disc (longitudinal section; hematoxylin and eosin staining). |
Optic Neuropathy
Optic neuropathy is a general term applied to optic nerve dysfunction. It may be caused by a variety of conditions such as ischemia (e.g., giant cell arteritis, compression by tumor); metabolic diseases (e.g., diabetes mellitus); drug toxicity (e.g., chloramphenicol); and vitamin deficiencies (e.g., B12 deficiency), among others. The etiology of optic neuropathy is outlined below based in categories according to the main pathological processes (Table 16.1). The histopathological changes observed in some of these diseases are described later in this chapter. Keep in mind that in clinical practice, many of the diseases involved in causing optic neuropathy overlap in several categories.
Table 16.1. Optic Neuropathy | |
|---|---|
|
Inflammation – Optic Neuritis
Inflammation of the optic nerve may occur as a result of infectious and non infectious causes. Optic neuritis produces visual loss resulting of axonal damage secondary to the inflammatory process.
Infectious causes of optic neuritis include bacterial (syphilis and mycobacteria)41,42; viral (cytomegalovirus, herpes zoster)43,44,45; fungal (Cryptococcus, Histoplasma, Aspergillus, zygomycosis)46,47 (Fig. 16.14); and parasitic (toxoplasmosis).48,49 These are rarely a primary process involving the optic nerve, more often these are secondary to infections of adjacent anatomic structures (eye, sinus), or part of a systemic infection, particularly in immunosuppressed and immunocompromised patients (Fig. 16.15). Clinically, there might be unilateral acute blurred vision, with pain or discomfort on movement or palpation of the globe. On examination, visual acuity with a central scotoma is decreased to colored objects but the disc appears normal.
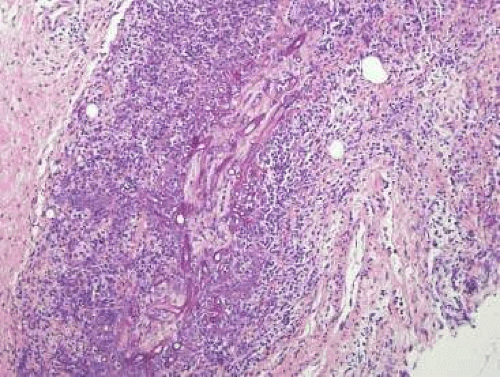 Figure 16.14. Septatae hyphae consistent with mucormyces associated with necrosis (hematoxylin and eosin) |
Noninfectious causes of optic neuritis include sarcoidosis, collagen vascular diseases50 and vasculitis, such as Wegener granulomatosis.51,52 Sarcoidosis affects the optic nerve in approximately 5% of patients with the disease.53 The optic nerve and is the second most frequently involved cranial nerve—the facial nerve being the most common.54,55 Rarely, sarcoidosis may present as tumefactive enlargement of the optic nerve without or prior to manifestation of systemic disease.56,57 Many cases are misdiagnosed initially as meningioma or orbital pseudotumor (Fig. 16.16).54,58 Magnetic resonance imaging reveals diffuse swelling of the nerve and enhancement.59 Histopathology is characterized by noncaseating epithelioid granulomas involving the nerve and the meninges (Fig. 16.17).
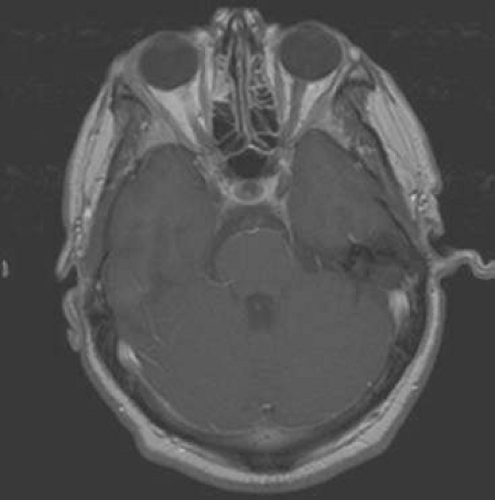 Figure 16.16. Sarcoidosis of the right optic nerve. Abnormal signal with enhancement surrounds the orbital intraorbital portion of the nerve (MRI). |
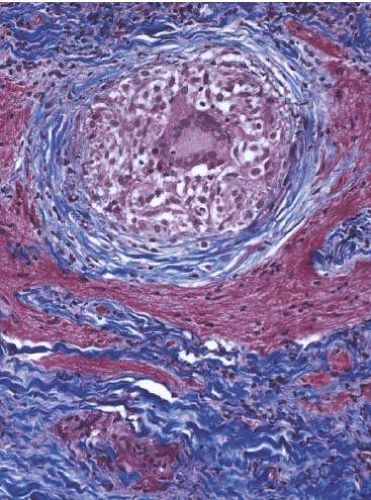 Figure 16.17. Sarcoidosis of the optic nerve. A well-formed granuloma with large central multinucleated giant cells (Masson’s trichrome). |
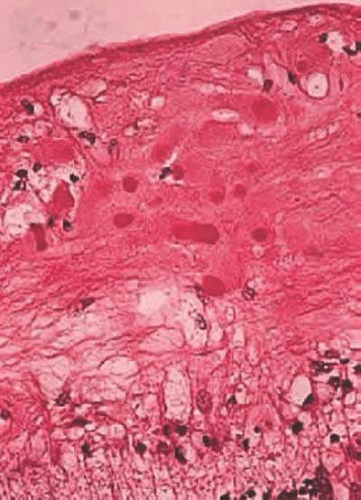
Papillitis refers to an inflammation of the disc resulting in swollen optic nerve head. It is usually accompanied by a vitreous haze due to the presence of vitreous inflammatory cells. Commonly, it is seen with suppurative endophthalmitis and panophthalmitis. The disc shows edema, perivenous cuffing by chronic inflammatory cells, and astrocytic proliferation. Thin bands of connective tissue may spread from the disc surface into the vitreous and cover the adjacent retina. Regardless of the cause, the histopathologic alterations are similar. The earliest change is nerve-fiber swelling in the region of the intermediary tissue of Kuhnt, the outermost fibers around the disc margins (Fig. 16.18). Bulging fibers initially displace the retina laterally, and the outer layers eventually buckle. The glial lamina is displaced anteriorly, and the vessels appear congested. Bundles of nerve fibers appear pale staining because of accumulation of watery interstitial fluid. The swollen nerve head protrudes forward against the vitreous and laterally displaces the central elements of the peripapillary retina from the disc margins. Proteinaceous material often accumulates between the sensory retina and the pigment epithelium around the disc margins, resulting in an enlarged blind spot.
Vascular Optic Neuropathy
Ischemic optic neuropathy represents an acute ischemic disorder of the optic nerve, manifesting clinically by sudden, unilateral, painless visual loss in most patients. This condition is divided in two types: anterior ischemic optic neuropathy, which involves the optic nerve head, and the less frequent, posterior ischemic optic neuropathy, which involves the remaining retrolaminar segment of the nerve. The anterior ischemic optic neuropathy is further classified in arteritic, which is secondary to giant cell arteritis, and nonarteritic, a result of idiopathic vascular insufficiency. The latter has been associated with a number of risk factors, such as hypertension, diabetes mellitus, medication, and hypotension, among others.60,61
In arteritic anterior ischemic optic neuropathy, various histological changes, including infarction in the optic nerve head, inflammation, thrombosis, and occlusion within the posterior ciliary arteries have been reported.62 The clinical diagnosis of giant cell (temporal) arteritis is usually confirmed by temporal artery biopsy.63 Because of the impending risk of permanent visual loss, with involvement also of the fellow eye, treatment with steroids is usually implemented before confirming the diagnosis with a biopsy. However, the biopsy should be performed as soon as possible since the inflammation might disappear after 2 weeks of treatment. Histologically, the inflammation is segmental, with areas of uninvolved arterial wall.64,65,66 It is characterized by an inflammatory infiltrate with lymphocytes, epithelioid histiocytes and multinucleated giant cells, along the internal elastic layer and the media of the temporal artery (Fig. 16.19), which extends to involve the optic nerve vessels and posterior ciliary vessels causing narrowing of the lumen. The inflammation disrupts and destroys the elastic layer.67 With treatment and scarring, the disruption of the elastic layer might be the only sign of a healed arteritis. This finding is highlighted with an elastic staining.
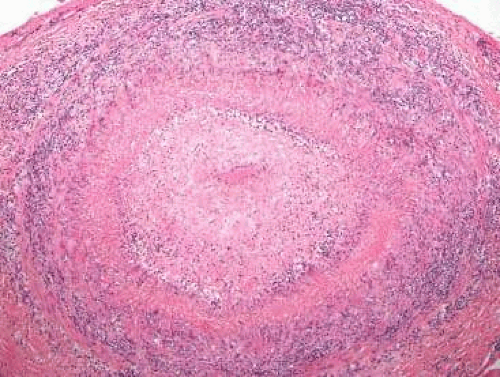 Figure 16.19. Giant cell arteritis. The arterial wall is markedly thickened with extensive chronic inflammation and scattered giant cells. The tumor is nearly occluded (hematoxylin and eosin). |
Posterior ischemic optic neuropathy is thought to result of infarction of the intraorbital optic nerve through unknown mechanisms. This condition has been reported in perioperative setting following a variety of surgical procedures, related to hypotension, anemia, excessive blood loss, among other causes hypoxia of the optic nerve and optic tract during surgical time.68,69 It has also been described in association with giant cell arteritis and a nonarteritic systemic vascular disease.70
Blockage of axoplasmic flow at the lamina cribrosa with accumulation of mitochondria and other organelles within the axon results in prelamina axonal swelling. Because the condensed axoplasm appears similar to a nucleus within a cell, the individual component of the cotton-wool spot is known as a cytoid body (Figs. 16.20 and 16.21).
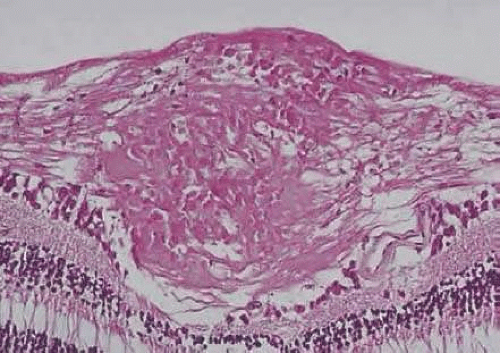 Figure 16.20. Cotton-wool spot within nerve-fiber layer of the retina (hematoxylin and eosin staining). |
Optic Neuropathies with Axonal Degeneration
Traumatic injuries to the optic nerve often result in optic nerve degeneration. The injury might be direct to the nerve, due to craniofacial trauma, including penetrating wounds, orbital and skull fractures, and rarely sinonasal surgeries; or indirect injuries which include contusions due to blunt trauma,71,72 “shaken baby syndrome,”73,74 and radiation-induced optic neuropathy.75,76 Histologically, the affected optic nerve might show a variety of changes including transsection, hemorrhage, edema, secondary demyelination, reactive gliosis, and eventually atrophy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree