Pathogenesis of Pseudomonas aeruginosa Keratitis
Linda D. Hazlett
Grant Support: NIH grants R01EY02986, R01EY16058, and P30EY04068
Microbial keratitis often is associated with complications caused by extended-wear contact lens usage. It has an incidence of 25,000 to 30,0000 cases annually. Treatment cost is approximately $15 to $30 million, which poses a considerable economic and medical impact.1
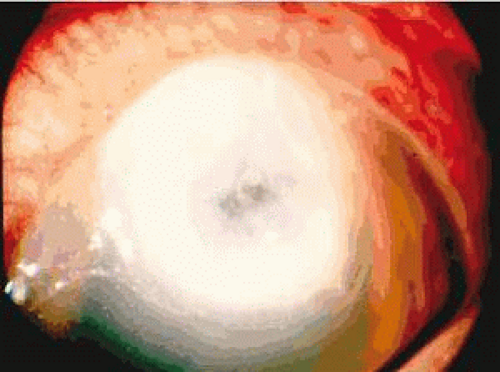
Pseudomonas aeruginosa is a gram-negative pathogen that induces keratitis and typically requires corneal injury for invasion.2 Because the bacterium has few nutritional requirements, it can adapt to a variety of ecologic conditions and niches, such as preserved ophthalmic solutions and the hospital environment. Pseudomonal and other gram-negative bacterial infections usually present as a rapidly progressing, suppurative stromal infiltrate with a marked mucopurulent exudate (Fig. 42.1). Yellowish coagulative necrosis surrounded by inflammatory epithelial edema is distinctive, and stromal ulceration can lead to significant stromal tissue destruction and loss. A ring infiltrate may appear in the surrounding paracentral cornea, and hypopyon (a dense inflammatory coagulum) usually is present. In addition, descemetocele formation or corneal perforation is not uncommon.3
Animal models of bacterial keratitis continue to be of value in the study of this disease and are produced by topical application of bacteria after abrading the epithelium, by intrastromal inoculation, or by placement of a contaminated suture or contact lens on the cornea.4,5,6,7 These approaches and models have led to an increased understanding of the mechanisms of corneal inflammation during bacterial keratitis. The newly developed extended-wear contact lens model in the rat also should prove particularly valuable, because no wounding is required to establish bacterial infection, unlike in other rodent models of disease (Fig. 42.2).6,7
Cytokines, Chemokines, Proteases, and Cell Infiltrate
Macrophage Inflammatory Protein-2, Interleukin-1, and Polymorphonuclear Cell Counts
In a bacterial infection, the innate immune response often includes local polymorphonuclear (PMN) cell recruitment (Fig. 42.3), which is essential to control bacterial replication and host survival. PMN cell persistence, however, also is associated with pathology and, in the cornea, this includes both stromal scarring and perforation.8,9,10 PMN cell infiltration into inflamed tissue is controlled largely by the local production of inflammatory mediators. In the mouse, two members of the C-X-C family of chemokines, macrophage inflammatory protein-2 (MIP-2; a functional homologue of human IL-8) and KC are potent chemoattractants and activators of PMN. In corneal infections, MIP-2 is the major chemokine that attracts PMN cells into the P. aeruginosa-infected cornea, and the persistence of PMN cells in the susceptible (cornea perforates) C57BL/6 (B6) mouse cornea correlates with higher MIP-2 chemokine levels (both mRNA and protein).11
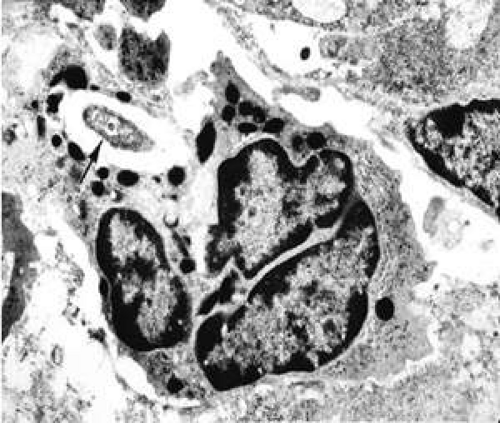 Figure 42.3. PMN cells in corneal stroma of infected mouse eye. Note the phagocytosed bacterium (arrow) in the cell cytosol surrounded by granules (12,000×). |
Interleukin-1 (IL-1), produced by macrophages, monocytes, and resident corneal cells12,13 also influences PMN cell influx into tissues.14,15 When tested, IL-1α and β (mRNA and protein) were elevated in B6 (susceptible) and BALB/c (resistant) mice, and levels peaked at 1 day postinfection (p.i.). Significantly greater amounts of IL-1 protein were detected in B6 versus BALB/c mice at 1 and 3 days p.i. and, at 5 days p.i., IL-1α and β (mRNA and protein) levels remained elevated in B6, but began to decline in BALB/c mice. Neutralization of IL-1β in infected B6 mice16 reduced disease severity, as evidenced by a reduction of PMN cells in cornea (myeloperoxidase [MPO] assay), decreased bacterial load, and decreased MIP-2 mRNA and protein in the cornea. The data confirmed IL-1 as an important contributor to P. aeruginosa-induced keratitis.16
Caspase-1 Inhibition
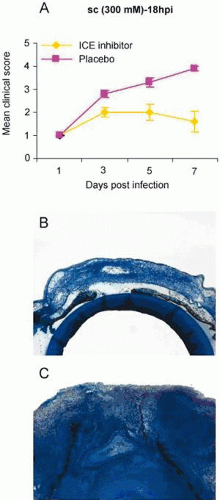
Studies using an inhibitor of the enzyme caspase-1, which converts the pro- to the active form of IL-1β, tested whether administering an inhibitor of IL-1β processing after the onset of infection would reduce experimental keratitis in B6 mice. Treatment with the inhibitor at 18 hours after infection reduced disease (Fig. 42.4), decreased PMN cell number, and reduced IL-β and MIP-2 protein levels in the cornea for two strains of P. aeruginosa, one of which was a clinical isolate.17 A combination of the inhibitor with the antibiotic ciprofloxacin also proved more efficacious for both bacterial strains than did treatment using the antibiotic alone. Last, treatment with the inhibitor provided better outcome than treatment with ciprofloxacin when a ciprofloxacin-resistant strain of the bacteria was used for infection.11 The use of caspase-1-deficient mice confirmed these results,18 but clinical testing of these provocative animal data awaits trial (18).
Matrix Metalloproteinases
Matrix metalloproteinases (MMPs) are a family of protein-cleaving enzymes that degrade extracellular matrix and basement membrane components.19 The most widely studied members of the MMP family in eye tissues are the gelatinases, MMP-2 and MMP-9, because they preferentially degrade basement membrane components such as type IV collagen.16 Recently, MMP-9 also has been found to process cytokines and chemokines, and thus has been suggested to be a prototypic example of the regulation of immune functions through proteolysis.20
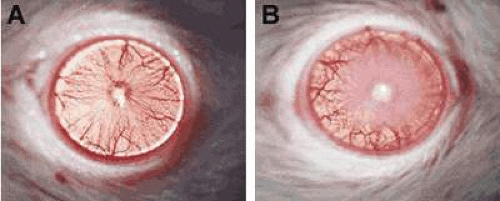
In P. aeruginosa corneal infection, Kernacki et al. showed that, when compared with susceptible (cornea perforates) nonimmunized mice, resistant immunized animals displayed lower levels of both the active and latent forms of MMP-9.21 In the genetically susceptible B6 versus. resistant BALB/c mouse models of P. aeruginosa (Fig. 42.5), greater fold increases in MMP-9 over MMP-2 were detected by gene array (Table 42.1) in the infected cornea of B6 (susceptible) over BALB/c (resistant) mice. The data were confirmed by reverse transcriptase-polymerase chain reaction (RT-PCR).22 Furthermore, latent and active forms of MMP-9 only were detected in the infected cornea using zymography. This showed that, in the infected cornea, B6 mice appeared to have increased levels of both forms of the enzyme when compared with BALB/c mice. The use of MMP-9 Ab neutralization and the injection of rMMP-9 protein, as well as MMP-9-/- mice, explored further the contribution of MMP-9 to corneal immunopathology. Using a clinical scoring system, rMMP-9-injected resistant BALB/c mice exhibited worsened disease, whereas Ab-neutralized B6 mice and MMP-9-/- mice had less disease than did control groups. Various other studies have indicated that MMP-9 (or gelatinase B) is a key player in the mechanisms underlying immunopathology. Active forms of gelatinases 2 and 9 were detected in endogenous corneal ulcer cases in tear fluid, and it was concluded that the active form of gelatinase expression may be related to the severity of ulceration.23
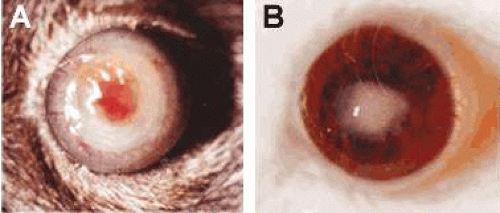 Figure 42.5. A perforated cornea in a B6-susceptible mouse at 7 days after infection is shown in A, while a resistance response in a BALB/c mouse (B) is shown for comparison (10×). |
Whether MMP-9 was crucial for Langerhans cells (LC) migration also was tested. LC are dendritic cells that capture foreign antigens and migrate with them to regional lymph nodes, where they are presented to naïve T cells. These cells have been shown to be critical in the innate immune response to P. aeruginosa, 24,25 and increased numbers of cells in the cornea have been associated with corneal perforation. When using the same paradigm of testing as described above, MMP-9 Ab-neutralized and knockout mice had fewer LC in the infected cornea, and BALB/c mice injected with rMMP-9 had more, thus suggesting that MMP-9 may be involved in LC transmigration into the cornea. When tested, however, soluble MMP-9 was not chemotactic.22 In this regard, others have reported that membrane-associated MMP-2 and -9 rather than soluble MMPs appear involved in cell migration.26 Membrane type MMPs (MT1-6) have been detected in the cornea of mice infected with P. aeruginosa, and peak expression of all three showed a good correlation with inflammation and stromal destruction.27,28
In inflammation, leukocytosis and blood-cell homeostasis are crucial in host defense. Gelatinase B assists in the migration of leukocytes in response to chemokines into sites of inflammation.29 The use of MMP-9 neutralization and MMP-9-/- animals showed that PMN cells are decreased in these two groups versus control mouse groups. Consistent with these data, PMN cells were increased in the cornea of rMMP-9-injected mice, suggesting that MMP-9 contributes to the regulation of PMN cell transmigration into the cornea. Immunostaining for collagen IV confirmed an interrupted basal lamina in the WT versus MMP-9-/- mouse cornea, possibly contributing to PMN cell transmigration into the cornea.22
Gelatinase B was first identified in PMN cells30 and later in monocytes and macrophages.31 In general, these types of cells secrete gelatinase A constitutively, but the expression of gelatinase B requires induction by adequate triggering.32 IL-1, lectins, double-stranded RNA, and endotoxin (LPS) are soluble inducers of MMP-9.32,33 Immunostaining showed that PMN cells in the cornea (and anterior chamber) were a source of MMP-9 (gelatinase B) in the P. aeruginosa-infected cornea.22 PMN-cell gelatinase B has been shown to be released in the extracellular medium as a latent proform, then activated mainly by PMN-cell elastase in vitro.34
Gelatinase B functions as a regulator and effector in leukocyte biology, because it is able to activate pro-IL-1β into the active form of the cytokine, both important in the susceptibility response of B6 mice.16,35 rMMP-9-treated mice had enhanced levels for IL-1β and MIP-2, whereas knockout or MMP-9-neutralized mice had decreased levels of both, suggesting that MMP-9 proteolysis modulates cytokine expression in the cornea. Because gelatinase B and cytokines both function in the extracellular milieu and might be secreted together at particular sites within tissues, it is not unexpected that they would interact. In fact, others have shown that the treatment of mice with an anti-IL-1β antibody before P. aeruginosa challenge resulted in a significant reduction of MMP-9 (36). Together with data from the current study, evidence suggests that, in the P. aeruginosa infected cornea, a reciprocal interaction occurs between the two.
CD4+ T Cells and Genetic Susceptibility to P. aeruginosa
The role of T cells in P. aeruginosa corneal infection was first examined in inbred B6 wild-type and β2 microglobulin knockout mice on a B6 genetic background.37 The corneas of both groups of mice perforated by 7 days p.i. and the histopathology was similar, with infiltration of PMN cells seen within 24 hours p.i. After infection, CD4+ and CD8+ (latter absent in the knockout mouse) T cells were present in the cornea by 3 days; by 5 days, activated (IL-2R, CD25+) cells were detected by positive immunostaining. The corneas of wild-type mice depleted of CD4+ T cells and infected with P. aeruginosa did not perforate at 7 days p.i., versus mice depleted of CD8+ T cells or treated with an irrelevant antibody. Antibody neutralization of interferon-γ (IFN-γ before infecting B6 mice also prevented perforation and was associated with a lower delayed-type hypersensitivity (DTH) response over B6 mice similarly treated with an irrelevant antibody. These data support the conclusion that a CD4+ T cell (T helper 1, Th1)-dominant response following P. aeruginosa infection is associated with genetic susceptibility and corneal perforation in B6 mice and provided initial evidence that CD4+ T cells are important in the development of keratitis.38).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree