HEALTH STATUS ASSESSMENT
Within the last 25 years, the measurement of health-related quality of life (HRQOL), medical- and patient outcomes, and clinical effectiveness has moved beyond its origins in health care research to find wider application across specialties and practice settings. Our society has grown and changed, and with it the need to allocate health service resources rationally across diverse health programs has steadily grown as well, prompting the evolution of new health policy decisions in both the public and private sectors. To assist in formulating those new decisions, caregivers, health care institutions, and even drug and device manufacturers have been called upon to develop and apply measures of the broad effects of illness states on patients’ lives, the effectiveness of intervention options, and their outcomes. Granted, some general instruments have been in use in the United States for 30 years or more, and millions of such questionnaires have been distributed. However, changes in delivery systems, advances in technology, new issues in health care payment systems, and a shift in patient management to include more chronic care and care of the aged have increased the importance of patient participation in assessment of their health care using contemporary health status assessment instruments.
In the past, simple biomedical indicators of change in health status, such as morbidity and avoidable mortality, survival rates, adverse reactions, complications, symptom relief, pain, and physical and biochemical markers of recovery, were the standard metrics of treatment outcome. Although it took an act of Congress and subsequent changes in research funding by the National Institutes of Health (NIH) (Patient Outcomes Research Act of 1989, S. 702), changes in health policy and allocation of health care resources have gradually moved to define health outcomes of intervention in the broadest sense, specifically those focused on the impact of the condition and its treatment on the patient’s emotional and physical functioning and lifestyle. At the center of this trend is the theme that a definition of health can no longer be limited to the absence of disease as measured by the traditional quantifiable criteria, but must also define the impact of disease and disease intervention on overall quality of life (QOL) and satisfaction with care (
1,
2).
THE “QUALITY OF LIFE”
“Quality of life is a vague and ethereal entity, something that many people talk about, but which nobody knows very clearly what to do about” (
3).
A paradox of measuring “quality of life”—a widely-recognized concept in the social and health sciences—is that it has no standard, uniformly accepted definition. While there is general agreement among experts that QOL is subjective and highly individual, its criteria and indicators reflect a similarly wide range of perspectives. These range from the lyrical—“the perceived discrepancy between what is and what might have been”—to the pragmatic—“evaluation of (personal) well-being with physical symptoms, sexual function, work performance, emotional status, etc.” (
4). A multitude of operational definitions exist that reflect philosophical, psychological, sociologic, economic, or biologic perspectives, but the inescapable conclusion is that the perception and achievement of QOL is personal, unique, and dependent on one’s own preferences, circumstances, values, and priorities in life. This has important implications that are explored in a later section.
HRQOL research was first conducted in cancer treatment settings where the balance between quality and duration of life was a pivotal issue in decisions to use new drugs or combinations of drugs of either partial or unknown efficacy but with very serious side effects (
5). Since then, QOL has become an important goal of treatment and marker of success in health interventions of all kinds. By definition, HRQOL is more focused than general QOL and thus
lends itself more readily to criteria for measurement, at least from a functional perspective, related to the ability to perform activities of daily living (ADL) and fulfill role functions adequately. Moreover, sophisticated health technology and treatments have expanded the life span, meaning that more people live—and live longer—with chronic health conditions. Thus, HRQOL includes perceptions of physical, emotional, and social well-being, before, during, and after treatment and the impact of those factors on the ability to lead a satisfying life.
However, it is a double-sided concept, incorporating positive as well as negative aspects of well-being and life, and is multidimensional, encompassing social, psychological, and physical health (
1). Although not routinely explored, a thorough profile of HRQOL would also include some assessment of the patient’s satisfaction with treatment (especially as it relates to the symptoms of greatest personal concern), and prospects for future well-being. In today’s environment of crushing health care costs, issues related to concerns about economic stability might well be included. To be useful, HRQOL must be a personal construct and it must be dynamic for, as health changes for the better or the worse, perspectives on life, roles, relationships, and experiences change. But poor health or the presence of disease is not invariably associated with poor HRQOL; rather, it can be shaped by factors such as coping, expectancy, and adaptation (
6) and studies have shown repeatedly that doctors’ and patients’ assessment of outcome do not always coincide. It is important to understand as well that although health is usually an important component of overall QOL, health status is neither a predictor nor a determinant of QOL: each can vary independently, even paradoxically.
In the most idealistic sense, the goal of medical or surgical treatment is to extend life and to optimize the patient’s health-related quality of that life, however he or she might define it. This is not a new concept. What is relatively new is the interest in formalizing its definition and measurement, and in using it as the unifying theme for assessing the burden of disease, the efficacy or effectiveness of treatment for that disease, and the individual outcome of treatment on the patient.
If HRQOL includes perception of physical, emotional, and social well-being and the impact of those factors on the ability to lead a satisfying life, many of the diseases and disorders treated in otolaryngology—head and neck surgery have an undeniable impact. Who would debate the pleasure of being able to smell a beautiful flower or the bouquet of a fine wine? Allergic or nonallergic rhinosinusitis can deprive patients of this sensation and impose other uncomfortable symptoms such as nasal congestion, fatigue, and pain. Evidence of hearing loss appears on an audiogram but does not reveal how it affects the ability to enjoy music, participate in a conversation in a crowded restaurant, or listen to a child’s whispered secrets. Loss of these elements of QOL can result in depression, anxiety, and withdrawal. The list continues: voice and the ability to speak, to sing, to tell the world who we are; sleep-disordered breathing; the gastroesophageal reflux disorders; the loss of facial symmetry and expression; and the cruel spectrum of head and neck cancers, some of the most visually obvious and functionally disabling cancers of all. The biomedical intervention outcomes for these conditions and others have been documented and reported; a few clinical trials have been completed, enough so that meta-analyses are becoming possible. But if ever there were multitudes of reasons to make measurement of HRQOL and patient-related outcomes a part of the clinical picture, otolaryngology— head and neck surgery has them!
WHY MEASURE HEALTH-RELATED QUALITY OF LIFE AND FUNCTIONAL STATUS?
Given that compassionate patient care of the highest quality is the goal of every health care provider, there is a need to understand the process from the perspective of the patient. While clinical outcomes are certainly important—indeed, would be considered the primary outcome in medical and surgical interventions—they are not always related to how the patient feels about the disease, its impact, the intervention, and
its impact. Those are issues that may influence not only patient satisfaction, but the recovery process itself (
7). The recent social imperative for individuals to take more responsibility for their personal health through prevention, healthy living, and self-education has made awareness of HRQOL a fact of life: most people are not familiar with the traditional biomedical markers of health and disease, but they do consider health an essential part of QOL. And quite simply, patients like to talk about themselves— they
like to be asked that most basic of questions: “How do you feel?” Thus, including both clinical and patient-based outcome allows health care quality to be measured in a meaningful way for both clinicians and patients.
Apart from the humanistic issues related to increasing awareness of the patient’s experience of health, disease, and treatment, there is a growing number of circumstances in which such measurements will play a key role: First, decisions about allocation of resources from third-party payers, managed care, and government agencies are increasingly requiring evidence of the effectiveness of health care interventions in the broadest sense—euphemistically termed “outcomes-adjusted reimbursement.” That is, data will be expected on whether the treatment leads to a life worth living, by providing a more patient-oriented baseline against which the medical effects of the intervention can be evaluated (
1,
8). For example, two or more drugs may have comparable medical effectiveness against the same condition but produce very different HRQOL results; this could prove to be a significant factor for both the patient and the provider in the decision-making process.
Second, the US Food and Drug Administration (FDA) now encourages drug and pharmaceutical manufacturers to develop and use patient-reported outcome measures (PROMs) to support labeling claims, especially those
that are “best known or best evaluated from a patient perspective” as a part of the approval process for new drugs and devices (
9). While evidence of this nature could be an asset to prescribing physicians when counseling patients in the United States, in Europe it is used to determine availability and pricing of medications. It is not unreasonable to anticipate that such a practice will become more widespread here as patient-reported HRQOL data become more influential in health policy and reimbursement decisions.
Another way in which HRQOL data can be used, and which has been somewhat sensationalized, is to assist in determining quality-adjusted life years (QALYs), an approach which has become one of the key considerations in allocation and reimbursement decision processes. A QALY is the arithmetic product of life expectancy and quality of the remaining years. A QALY places a weight on time in different health states: a year of perfect health is worth 1 and a year of less than perfect health is worth less than 1. Death is considered to be equivalent to 0. QALYs can be used to compare the extent of benefits gained in a broader sense from a variety of interventions when compared to their costs, side effects, and survival time (
10). Only a small number of HRQOL measures are used for this application and it remains a subject of considerable controversy when used in the context of governmental control of health care benefit allocation.
Fourth, health technology assessments (HTAs) are used in many European countries to determine the benefits or added value of new technologies for the purpose of reimbursement and pricing decisions and/or the establishment of clinical guidelines. In the United States, HTA is carried out through the Agency for Healthcare Research and Quality. The recent interest in comparative effectiveness research has prompted US expert advisors to call for the HTA process to include not only clinical and biomedical outcomes, but also “… important measures of effectiveness such as patientreported outcomes, including health related quality of life, patient satisfaction, activities of daily living, and work productivity as relevant to the various USA stakeholders” (
11). A large NIH project, the Patient Reported Outcomes Measurement Information Service (PROMIS) offers tools that can be used as primary or secondary endpoints for clinical studies of treatment effectiveness in a wide variety of chronic diseases and in the general population.
Finally, contract research organizations use patientreported HRQOL instruments routinely as a part of their protocols when conducting large-scale clinical trials and field studies for drug and device manufacturers, in preparation for submitting new drug applications to the FDA.
FUNCTIONAL STATUS
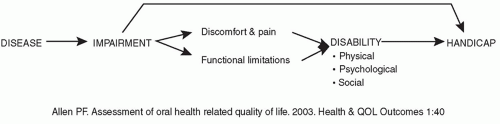
The conceptual framework for measuring multidimensional functional health status is related to the World Health Organization (WHO) classification of impairment, disability, and handicap, and seeks to address the functional and psychosocial outcomes of diseases, disorders, and conditions (
2). Although “impairment,” “disability,” and “handicap” are interrelated concepts, they are not interchangeable. Their relationship is shown in
Figure 49.1. An
impairment is a problem in body structure or function, which can manifest itself as a functional limitation, for example, difficulty in hearing and/or discomfort and pain. Impairment describes the biologic status: loss of a body part, dysfunction secondary to trauma, disease, malformation, etc. If severe enough, the impairment can also—but does not invariably—result in a
handicap, which prevents the individual from carrying out life roles. Functional limitations or discomfort and pain produced by an impairment can lead to a
disability—restriction in performing all or some of the ADL due to physical, psychological, and/or social issues, including such problems as fear, embarrassment, feelings of isolation, or loss of independence. This can be seen with disorders such as Ménière disease and head and neck cancer. While restriction in performing ADL can be measured objectively, the patient’s QOL experience is subjective, affected by how he or she feels about those restrictions. Awareness of how the patient feels about his or her impairment can give useful insights into its life impact, leading to some therapeutic enablement strategies (
12). Disabilities can also lead to handicap (although use of the word “handicap” has fallen into disfavor, imparting some negative psychosocial connotations) (
10).
These broad definitions of biologic states and their associated physical, mental, and social consequences set the stage for the structure common to the tools we use to measure health status, including physical health, mental health, social health, role function, and overall well-being. Most can be characterized in terms of the range and focus of the information they collect. Although there are several
ways in which to group the hundreds of available PROMs and HRQOL measures, for this discussion, we consider the instruments in the broadest sense:
generic, symptom-specific, and
disease-specific. While these three categories of health status and QOL instruments each have their own properties, advantages, and disadvantages (see
Table 49.2), they have an additional important property in common. Because the goal is to learn the impact of disease and interventions on the patient’s HRQOL and functional status, they are designated patient-reported outcome measures (PROMs). As officially defined by the US FDA, a PROM is “… an instrument of any aspect of a patient’s health status that comes directly from the patient (i.e., without interpretation by a physician or anyone else)” (
13). A PRO can describe health status, disability, handicap, a functional outcome, or a factor contributing to QOL in a standardized measure; these data provide a useful supplement to the medical assessment. In a very real sense then, the patient becomes the clinician’s partner in achieving balanced insights into clinical effectiveness.
TYPES OF HEALTH-RELATED QUALITY OF LIFE MEASURES
Generic HRQOL instruments are designed to be applicable across a broad range of diseases, disorders, and conditions. Similarly, they can be used with different groups of patients and to assess the impact of a range of different interventions, making them valuable for comparative effectiveness studies. Both general QOL and HRQOL can be addressed with generic instruments. Some of the most widely used generic scales are the Medical Outcomes Survey SF-36 (MOS SF-36); the Glasgow Benefit Inventory (GBI); and the World Health Organization Quality of Life Assessment Inventory (WHOQOL-BREF). Just as a number of factors contribute to overall QOL, HRQOL instruments are designed to measure those perceptions by separate questions organized into
domains, with the goal of parsing out individual influences on the overall quality score. Over the years, consensus has been established that HRQOL is a multidimensional concept, divided for measurement into three core domains: physical, social, and psychological. It is important, however, to distinguish between
functional scales and HRQOL scales. In the physical domain, perception of normal or disrupted function such as mobility, pain, and sensory function are evaluated. In the social domain, questions are centered on items such as ADL, interactions with family and friends, and responsibilities in and out of the home. The psychological domain would involve emotional and mental functioning, including mood, distress, memory, self-image, special concerns, etc. While these three domains are fundamental, some instruments contain other domains, such as vitality, spirituality, sexuality/intimacy, financial concerns, and role function. Each domain is comprised of a series of questions that are scored separately and/or contribute to an aggregate score. An overall global HRQOL will yield an estimate of the patient’s subjective evaluation of his or her own health; this may be different than simply the sum of the domain scores (
14). A brief list of some of the more commonly used generic HRQOL instruments is presented in
Table 49.1.
Although generic instruments have been in widespread use longer than any other type and thus have very stable psychometric properties and reliability, they often lack the sensitivity to evaluate subtle changes or mild disease, lack relevant subdomains, or fail to demonstrate health impact of certain conditions such as rhinosinusitis or mild to moderate hearing impairment. Disease-specific or possibly symptom-specific instruments might be a better choice to overcome these shortcomings.