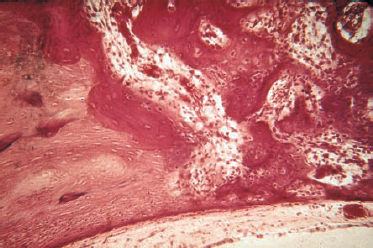
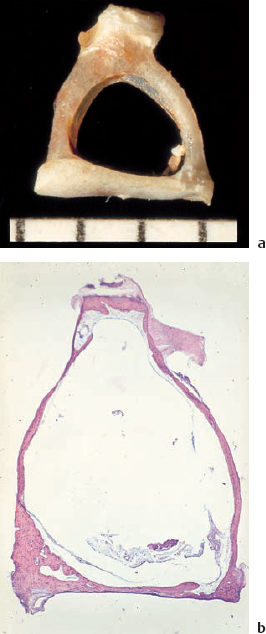
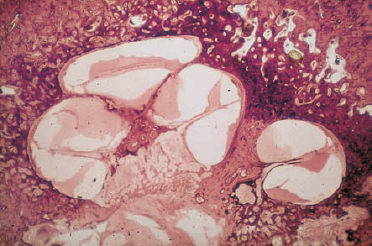
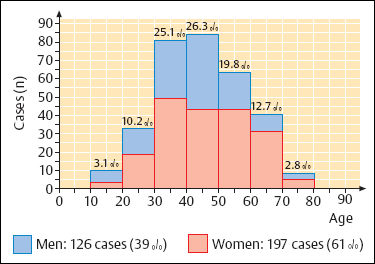
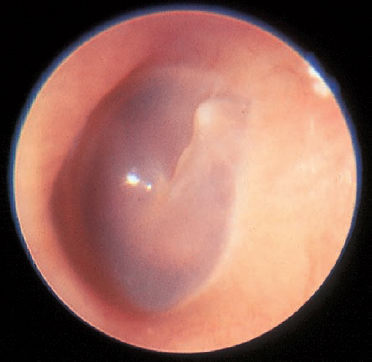
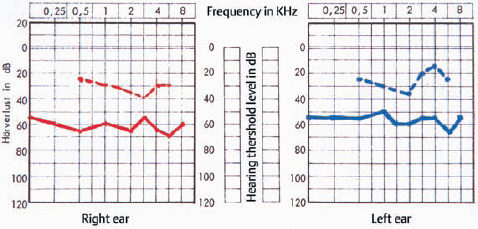
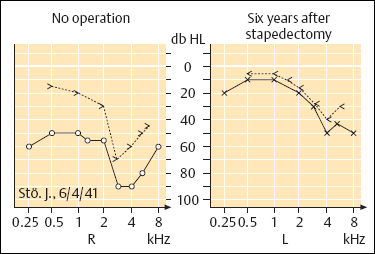
7 Otosclerosis There is no doubt that surgery for otosclerotic deafness was the pioneering development for all forms of functional microsurgery of the ear. It sent the exciting message round the world that hearing could be restored to the deaf by means of minor operations with reproducible results, and it prompted many otologists to learn how to carry out surgery for otosclerosis. Although Friedrich Kessel in Jena (Germany) conducted stapes surgery as early as the 1870s, with unsatisfactory results (Kessel 1876, 1878), the credit for developing this field belongs to those who pioneered fenestration of the lateral semicircular canal—such as Sourdille (1937), Holmgren (1937), Lempert (1938), Shambaugh (1949, 1959), and others who established a variety of successful operations to treat otosclerosis and improve disturbed hearing. Moritz (1950), Wullstein (1952, 1968), and Zöllner (1952), who opened up the field of functional surgery for chronic otitis media, were basically influenced by the techniques used and problems encountered in fenestration operations. Their various backgrounds in otosclerotic surgery can be traced to the development of tympanoplasty. In contrast to the widely varying opinions and assumptions that are still under debate in relation to the various forms of chronic otitis media, there has been little further debate on the best ways of treating otosclerotic deafness since the reintroduction of stapedectomy by Shea (1958), and discussions have been confined to minor technical details, analysis of failures, and epidemiological matters. The following description of the surgical techniques involved can therefore be kept very short. Otosclerosis is a specific bone disease, marked by local, focal and noninflammatory bone destruction and disorganized re-formation (Fig. 7.1), limited to the otic capsule. While the normal cochlear and labyrinthine bone has a lamellar structure, the growth pattern of an otosclerotic focus is spongy. In this respect, the popular term “otospongiosis” is a more accurate one. After Toynbee (1861), Politzer (1894) was the first to give a detailed description of the histopathological features of the disease and to distinguish between otosclerosis and the group of inflammatory hearing disorders. There is a consensus that fresh processes are well vascularized, like bone marrow, and that they contain many osteoclasts and osteoblasts. This zone is deeply stained by hematoxylin, while older foci develop a denser bone structure with few signs of osteoclastic activity and appear more “sclerotic.” Fig. 7.1 Otosclerotic bone in the cochlear wall. The otosclerotic, spongy area, characterized by a disorderly network, with wide vascular channels and fresh, deeply red-stained bone, has a sharp border against the healthy laminar bone (left), with no signs of inflammation (hematoxylin-eosin stain, original magnification 200 ×). Although the typical form of bone necrosis can occur in any compartment of the otic capsule, even in the semicircular canals, most foci are situated in the cochlear shell, with the anterior margin of the oval window (fossula ante fenestram) as a site of predilection. Here, the annular ligament and stapes footplate are most often involved, resulting in ankylosis (Fig. 7.2). During tympanoscopy, typical pale and thickened areas can also be recognized on the promontory side of the oval window or on the facial canal with the operating microscope. In advanced stages, the footplate can become completely thickened, and the focus may extend into the stapes crura. Similarly, massive bone production can fill out the oval niche like an atretic plate, and in such cases the term “obliteration” is used. However, the outer bony walls of the cochlea, and less frequently the semicircular canals, can also be included in a massive process of osteospongiotic bone destruction and reorganization (Fig. 7.3), which then protrudes into the spiral ligament and even into the three spiral ducts or the limbus. Otosclerotic occlusion of the inner ear has been reported. The diagnosis of cochlear otosclerosis in vivo can be made using high-resolution computed tomography (CT), which demonstrates radiolucent areas in the labyrinthine capsule. The bone density loss can be measured densitometrically. Using this technique, De Groot et al. (1987) were able to demonstrate some impressive examples of active spongiotic foci, although they were unable to distinguish between hypermineralized foci and normal capsular bone. The importance of densitometry was emphasized by the fact that in their study, 37% of the affected ears without visible foci on the CT had a moderately decreased capsular density. Youssef et al. (1998) have pointed out that there is no scientific evidence to link the radiological findings with histological substrates of capsular otosclerosis. Magnetic resonance imaging may delineate large, immature foci. In patients with progressive bilateral sensorineural hearing loss, the presence of cochlear otosclerosis should therefore be considered despite the limited sensitivity of current diagnostic techniques, in view of the therapeutic options now available—such as sodium fluoride treatment (Shambaugh 1969, Glass and Philipp 1988). Final conclusions on questions of pathogenesis and histogenesis have not yet been found. Autosomal dominant heredity of the disease was postulated at an early stage (Albrecht 1922), and this has been repeatedly confirmed (Nylen and Nylen 1952, Larsson 1960, Causse and Causse 1984). Most authors favor the idea that otosclerosis has autosomal dominance, but that it is virtually recessive, as only a minority of those affected present symptoms. Guild (1930) calculated that this ratio was about 10%. Larsson (1960) concluded that the clinical entity has a monohybrid, autosomal-dominant heredity, with a penetrance of the pathological gene of 25–40%. This would imply that 19.6% of the children of one otosclerotic father or mother would become deaf. However, the female-male sex distribution of the disease is about 1.6 :1.0 (Soifer et al. 1970); other authors have reported a ratio of 2.0 : 1.0, based on surgical statistics. Molecular-genetic investigations have located a gene for otosclerosis at chromosome 15q25-q26 (Tomek et al. 1998). It is unclear why those of Caucasian ethnicity are more likely to develop otosclerosis, while the disease is comparatively rare (1 : 7) in African-Americans and almost un-known among Africans and American Indians. It is also infrequent in Japan and China (Altmann et al. 1967). The relationships between the disease and other bone diseases, such as Paget’s disease or monostotic fibrous dysplasia (localized osteitis fibrosa), have not been fully elucidated. Simultaneous occurrence of otosclerosis and abnormal bone fragility and blue sclera is seen—van der Hoeve’s syndrome (van der Hoeve and de Kleijn 1917). Many otologists have observed that pregnancy and delivery can have a triggering function for the start of clinical symptoms. The common observation that there is an increased incidence during the third to the fifth decades of life (Fig. 7.4) is in accordance with the assumption that hormonal co-factors are involved (this is why information leaflets on the side effects of contraceptives usually mention otosclerosis). However, the ways in which hormones interact with bone metabolism have not yet been fully explained; and various attempts to attribute local bone destruction to occasional mechanical overload in deformities of the skull base have also not yet fully elucidated the processes involved. Fig. 7.2 Otosclerotic alteration of a stapes footplate. a There is marked thickening of the footplate with whitish new bone in the anterior margin here. b Histological section of another stapes, with otosclerotic accumulation at the anterior crus and overgrowth of the annular ligament. Fig. 7.3 Capsular otosclerosis. This histological section of a cochlea (from the collection of the University of Würzburg, Germany), shows severe deformation of the outer cochlear walls, with wide-meshed otospongiosis and small spicules below the annular ligament, as well as invasion of the osseous limbus (hematoxylin-eosin stain, original magnification 100 ×). Fig. 7.4 Sex and age distribution in our own series of 323 patients with otosclerosis. The female : male ratio was 1.6 : 1. Recent information (Arnold and Friedmann 1988, Arnold et al. 1996) describing pathological data from otosclerotic bone has suggested that viral infection (measles) may have a causative role, but this requires further confirmation. Even athorough genetic study of 151 otosclerotic families came to the conclusion that a substantial proportion of the cases might arise from non-genetic causes (Sabitha et al. 1997). The cardinal symptom of otosclerosis is a sudden or slow onset of unilateral deafness. The second ear may remain unaffected, but involvement of it more often follows after an interval of two or three years, with the same pattern of hearing loss gradually developing. Most patients complain of concomitant whispering or rushing noises in the ear, which may be temporarily interrupted by high-tone tinnitus. Unsystematic vertigo or short-term giddiness can be an initial symptom, but this is more often ignored, as is a soft intra-aural sensation of pressure. Many patients do not visit a doctor until they become unable to understand a phone call with the second ear. The otoscopic examination shows a normal, well-angled and reflecting drum membrane. Rarely, the drumhead can have a reddish translucence in the direction of the promontory (Fig. 7.5). Known as Schwartze’s sign, this is said to suggest an active phase in an otosclerotic focus at the oval niche (Schwartze 1885). I have not found that the sign is reliable, except in cases of familial juvenile otosclerosis with a fulminant spontaneous course. Impedance measurements show undisturbed ventilation of the tympanic cavity, but the stapedial reflex cannot be recorded on the affected side, due to the ankylosis. The audiogram reveals an air-bone gap of 10–20 dB in the early phase, but it is not uncommon to find a conduction loss of 30–40 dB even at the first consultation (Fig. 7.6). The bone conduction threshold may remain stable for several years, but more often shows a typical virtual depression in the middle frequencies, with a maximum of 15–20 dB at 2 kHz (the Carhart notch). This limited deterioration in bone conduction is attributed to a blockade of the hydrodynamic perilymphatic vibrations within the cochlea by the fixation of the stapes footplate. It is eliminated after a successful intervention. Sensorineural hearing loss with tinnitus but without any conductive impairment can be regarded as symptomatic of purely capsular otosclerosis. Our own observations of patients who developed sound conduction deficits after several years, and then had otosclerotic foci verified histologically after removal of the stapes, suggest the existence of capsular otosclerosis (Fig. 7.3). Presurgical imaging diagnosis of otospongiotic bone has not become routine. High-resolution CT may be able to visualize broad foci as zones of decreased density surrounding the cochlea. However, small erosions and the typical focus at the fossula ante fenestram are usually not evident. A radiogram using Schüller’s projection will normally show well-developed pneumatization with no specific signs of inflammation. There are, of course, patients with a history of chronic otitis during childhood who develop a sclerotic mastoid, with later suspicion of otosclerosis. Fig. 7.5 The left tympanic membrane in a case of acute hearing deterioration due to otosclerosis. There is a normal appearance, but a reddish shadow over the promontory has become translucent (Schwartze’s sign). Fig. 7.6 A typical audiogram in bilateral otosclerosis. Both ears present with marked combined hearing losses of 20–35 dB air conduction factors and 20–40 dB bone conduction factors. The sensorineural notches at 2–3 kHz are known as Carhart notches, and are caused by an intracochlear increase in impedance due to ankylosis in one window. A reduction in, or even disappearance of, these notches can be expected after stapedectomy. The diagnosis of otosclerosis is usually reached by excluding other similar syndromes. There are only a few conditions that imitate the triad of conductive hearing loss, normal appearance of the drum membrane, and low-frequency tinnitus: The prognosis for improved hearing after stapes surgery for otosclerotic deafness is excellent, and the procedure is associated with only a minimal risk of injury to the inner ear or facial paralysis. Another strong argument in favor of surgical treatment is Wullstein’s experience that cochlear function, expressed in terms of bone conduction thresholds, remains more stable in operated ears than in un-operated ones (Fig. 7.7). Against this background, it can be explained to the patient that there is an indication for stapedotomy or stapedectomy, as the therapy of choice. When progressive deafness has been verified by a series of audiograms, any delay in carrying out surgery would be counterproductive—the spontaneous course would reduce the chances of success. It should also be emphasized that surgery is always more difficult in the advanced stages of ankylosis and will involve a higher complication rate, with obliteration of the oval niche. Patients who refuse surgery and prefer a hearing aid should be aware of this. Fig. 7.7 Protection of inner ear function by stapedectomy treatment for otosclerosis. The bone conduction level in the operated left ear was unchanged from the preoperative level six years after stapedectomy. The right ear—initially with better hearing—in which surgery was refused, developed severe sensorineural hearing loss (near deafness), over the same period. The majority of our patients underwent surgery between the ages of 30 and 60. Interventions for juvenile otosclerosis were indicated after rapid deterioration of the air conduction and bone conduction thresholds, with early total deafness being expected. Operations in patients over the age of 70 were exceptional, but were equally successful. Additional arguments in favor of intervention are that surgery can be carried out using local anesthesia, and that there is a high probability of at least partial relief of the tinnitus. There are no real contraindications, with the exception of intervention in the only hearing ear, and concomitant chronic otitis media with infection. A bacteria-free middle ear is mandatory for stapes surgery.
 Clinical Entity
Clinical Entity
 Symptoms and Signs
Symptoms and Signs
 Differential Diagnosis
Differential Diagnosis
 Indications for Surgery
Indications for Surgery
 Surgical Techniques
Surgical Techniques
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


