Chapter 40 The clinical evaluation of orbital lesions is treacherous due to the overlap of signs and symptoms of disease caused by orbital inflammatory processes with those of orbital mass lesions. For this reason, the differential diagnosis of orbital inflammation presented in this chapter almost always includes neoplastic and vascular diseases along with infection and systemically associated processes. We presuppose that lesions caused by trauma have been ruled out by the medical history. Attention to several questions during patient evaluation can facilitate the diagnosis and management of inflammatory lesions of the orbit. The onset, duration, localization, and progression of symptoms are of utmost importance. Inflammatory lesions of the orbit are much more common than neoplastic lesions, either benign or malignant. Lesions of chronic duration (months to years) with limited effect on orbital structure and function are more likely to be benign (whether neoplasia or low grade inflammation), as opposed to lesions of short duration (days to weeks), which are most likely to be malignant neoplasia or high grade, acute inflammation.1,2 Hyperacute presentation (hours) suggests either hemorrhage into a preexisting lesion, or fulminant inflammation or infection. Pain is nonspecific and is a frequent complaint in patients with either malignancy or inflammation. However, numbness often indicates neoplastic neural invasion. Working through the differential diagnoses, the history, physical findings, imaging characteristics, results of prudent laboratory investigation, and histopathologic evaluations guides the clinician to the correct diagnosis and treatment for orbital inflammation. The most reasonable format for categorization has been presented separately by Jakobiec and colleagues2 and Rootman et al.1 This chapter utilizes their approach with some modification as is outlined in Table 40–1. Orbital diseases in this category have been lumped in the past into a relatively poorly descriptive grouping labeled “orbital pseudotumor.” More recent schemes suggest dividing diagnostic elements into those with more nonspecific inflammatory processes, or into those with more specific inflammatory patterns, based on a known etiology of the inflammation (infection), or on a specific inflammatory configuration (e.g., granulomatous). In this discussion, those nonspecific idiopathic inflammations of the orbit that primarily affect the anterior orbital structures, the posterior structures, and those with diffuse orbital processes are considered together. Those predominately affecting the extraocular muscles and lacrimal gland are considered subsequently. Since the early 19th century, the term orbital pseudo-tumor has been used to describe a set of orbital lesions composed of benign nonspecific idiopathic inflammatory infiltrates, characterized by accumulation of heterogeneous cellular constituents, and with varying degrees of fibrosis. Several authors have since recommended abandonment of this term in favor of a more practical nomenclature designated as nonspecific idiopathic orbital inflammation (NSIOI).3,4 Several subtypes have been described based on lesion location (anterior orbital inflammation, posterior orbital inflammation, diffuse orbital inflammation, myositis, dacryoadenitis), the degree of inflammation (focal, diffuse), or the amount of fibrosis (sclerosing). The clinical presentation in any of these may be acute (hours to days), subacute (days to weeks), or chronic (weeks to months). This discussion focuses on the more common presentation of multifocal (anterior or posterior orbital) and diffuse orbital inflammation, and not those related to infection or to specific local or systemic processes.
ORBITAL INFLAMMATION AND INFECTION
NONSPECIFIC INFLAMMATIONS OF THE ORBIT
NONSPECIFIC IDIOPATHIC ORBITAL INFLAMMATION
Nonspecific idiopathic orbital inflammation (NSIOI: “orbital pseudotumor”) Anterior Posterior Diffuse Sclerosing Myositis Dacryoadenitis Specific orbital inflammation Inflammation secondary to infection Bacterial Fungal Parasitic Vasculitic Inflammations Giant cell (temporal) arteritis Wegener’s granulomatosis Polyarteritis nodosa Granulomatous inflammations Sarcoidosis Xanthogranulomatous inflammation Erdheim-Chester disease Adult orbital xanthogranuloma Necrobiotic xanthogranuloma Foreign-body granuloma Ruptured dermoid, mucocele Graves’ orbitopathy Sjögren’s syndrome Reactive inflammation Lymphoproliferative (neoplastic) inflammation/infiltration Non-Hodgkin’s lymphoma B cell Burkitt’s T cell Plasma cell Hodgkin’s lymphoma Leukemic Granulocytic sarcoma Histiocytic Langerhans’ cell histiocytosis Malignant histiocytosis |
URGENCY OF EVALUATION
The urgency of evaluation is dependent on the symptom complex. Those patients with severe pain or decreased vision need to be seen promptly. Those with a more indolent course or mild diplopia should be seen as soon as is practicable. If the patient is also diabetic or otherwise immunocompromised, examination should proceed emergently to rule out fungal infection (particularly mucormycosis).
DIAGNOSIS
Demographics
Nonspecific idiopathic orbital inflammation (NSIOI) usually presents between the ages of 30 and 50, but can occur in children and the elderly.1 Although children can present with bilateral disease, adults usually have unilateral involvement. There is no gender or ethnic predilection.
Symptoms
Pain
Pain is a hallmark of NSIOI. In the acute form, the onset is sudden, but depending on the degree of inflammation present, it can also be subacute, with onset over days. Pain is localized to the periorbital region on the side of involvement, and may be exacerbated by ocular movement. If a posterior inflammatory picture is present, pain may be more modest. If associated with scleritis, pain may be referred to the temple.
Lid Swelling and Erythema
Lid involvement varies with degree of inflammation, but is not as dramatic as that seen in orbital cellulitis. Swelling can include the periorbital regions, frequently demarcated by the orbital septum at the orbital rim (Fig. 40–1). Swelling should not be indurated or warm. Lid swelling is less prominent in posterior NSIOI.
Diplopia
Double vision is sometimes described, but is less prominent than that seen with primary inflammation of extraocular muscles (myositis). The diplopia may be caused by muscle restriction, especially in the more fibrotic forms, or due to congestion. Forced ductions and cross-cover analysis sometimes indicates decompensation of a preexisting phoria. Posterior NSIOI usually presents as an orbital apex syndrome often with multiple muscle involvement.
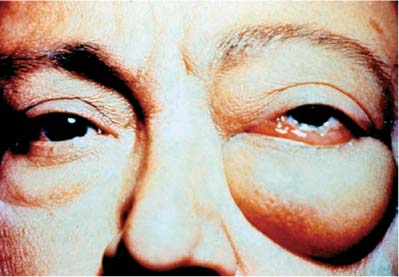
FIGURE 40–1 Anterior nonspecific idiopathic orbital inflammation. Note the proptosis, especially of the left eye. There is also periorbital edema and chemosis.
Vision Loss
Vision loss is uncommon in anterior forms of NSIOI except as secondary to ocular inflammation. In posterior forms, vision decline can occur due to compression or involvement of the optic nerve. Vision loss can also be caused by secondary uveitis or scleritis with retinal involvement. Uveitis is a more common finding in anterior NSIOI.
Fatigue
Patients frequently complain of loss of energy, but do not have systemic signs of infection (afebrile).
Signs
Proptosis
Although proptosis is considered the hallmark of orbital mass effect, the degree of proptosis in NSIOI can be quite variable depending on the degree of inflammation and fibrosis. There is usually resistance to ocular retropulsion, often with pain. Proptosis is less prominent with posteriorly located inflammation (apical).
Conjunctival Injection and Chemosis
This is generalized across the conjunctiva, and the injection is not severe unless significant ocular involvement is present.
Uveitis
With both posterior and anterior forms of NSIOI, some degree of uveal inflammation may be seen. This can include iridocyclitis, pars planitis, posterior uveitis, and posterior scleritis.5 Retinal striae from choroidal involvement or posterior ocular mass effect may be present, and can lead to a hyperopic shift. Secondary exudative retinal detachment has been described.5
Papillitis
Inflammation of the optic nerve head or the surrounding nerve fiber layer has been described uncommonly, both in association with choroiditis, and separately. This can lead to visual acuity and field loss.6
Glaucoma
An increase in intraocular pressure can be seen in anterior NSIOI, with either direct inflammation of trabecular meshwork structures, or secondary to blockage of aqueous outflow passage.7 Cyclitis (inflammation of the ciliary body) without significant cellular release into the aqueous can result in a lowering of intraocular pressure.
Red Flags
The following features should raise the question of alternative diagnoses:
 Bilateral orbital involvement. This is more common in children, but in adults raises the question of thyroid ophthalmopathy, cavernous sinus thrombosis, or systemic vasculitis such as polyarteritis nodosa, Wegener’s granulomatosis, or Erdheim-Chester disease. Lymphoproliferative neoplasia is often bilateral.2
Bilateral orbital involvement. This is more common in children, but in adults raises the question of thyroid ophthalmopathy, cavernous sinus thrombosis, or systemic vasculitis such as polyarteritis nodosa, Wegener’s granulomatosis, or Erdheim-Chester disease. Lymphoproliferative neoplasia is often bilateral.2
 Significant ophthalmoparesis. Suggests alternatives as described previously in this chapter. Aqueous cell and flare and pertinent ocular signs can indicate orbital ischemic syndrome.
Significant ophthalmoparesis. Suggests alternatives as described previously in this chapter. Aqueous cell and flare and pertinent ocular signs can indicate orbital ischemic syndrome.
 Fever, warmth, significant orbital/lid swelling. Orbital cellulitis must be ruled out.
Fever, warmth, significant orbital/lid swelling. Orbital cellulitis must be ruled out.
 Paranasal sinus disease. Although orbital inflammation may follow nonspecifically from a sinus inflammation, sinus and hence orbital infection must be entertained, as well as vasculitic processes such as Wegener’s granulomatosis.8
Paranasal sinus disease. Although orbital inflammation may follow nonspecifically from a sinus inflammation, sinus and hence orbital infection must be entertained, as well as vasculitic processes such as Wegener’s granulomatosis.8
 Presence of diabetes mellitus or immunocompromise. Mucormycosis needs to be ruled out emergently.
Presence of diabetes mellitus or immunocompromise. Mucormycosis needs to be ruled out emergently.
DIFFERENTIAL DIAGNOSIS
Anterior NSIOI
Orbital cellulitis, uveitis without orbital involvement, posterior scleritis without orbital involvement, systemic inflammatory syndromes (collagen vascular disease), Wegener’s granulomatosis, arteriovenous fistula. In children, orbital cellulitis, rhabdomyosarcoma, leukemic infiltrate, metastatic neuroblastoma should be considered.
Posterior NSIOI
Hemorrhage into preexisting orbital mass, Tolosa-Hunt syndrome, lymphoma, fungal infections including mucormycosis, paranasal sinus tumor spread, Wegener’s granulomatosis
ANCILLARY TESTS
Neuroimaging
Because of the possibility of intracranial extension or involvement,9 neuroimaging of both the brain and orbits is absolutely required. This should be performed in both axial and coronal planes (digital reconstruction if not available), and both with and without contrast medium. Computed tomography (CT) is generally ordered initially due to the improved visualization of bone and paranasal sinuses.2 Subsequent magnetic resonance imaging (MRI) is often also ordered for better visualization of the brain, cavernous sinus, and some soft tissue components of the orbit. MRI should also be ordered with additional fat suppression techniques.
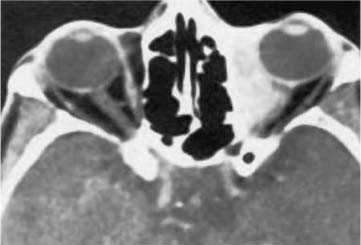
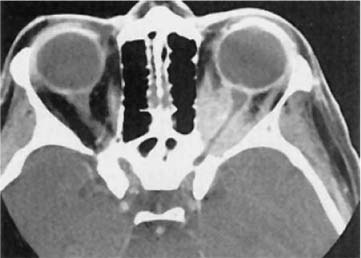
CT with contrast (Fig. 40–2) typically reveals a diffuse orbital infiltrate that, in anterior forms, is ragged on the edges and conforms to the curvature of the globe.10 Scleral involvement appears as thickening and enhancement. Remodeling of bone is not uncommon, sometimes including hyperostosis.11 Bone erosion is more common in neoplasia.2 In posterior NSIOI (Fig. 40–3), CT often depicts enhancement and enlargement of the optic nerve sheath as well as the perineural fat or posterior portions of the extraocular muscles.12
FIGURE 40–2 Computed tomography (CT) of nonspecific idiopathic orbital inflammation affecting the medial aspect of the orbit anteriorly. Posterior involvement is also present. (From Weber AL, Romo LV, Sabates NR. Pseudo-tumor of the orbit. Radiol Clin North Am 1999;37:151–168.)
FIGURE 40–3 CT scan of nonspecific idiopathic orbital inflammation affecting primarily the posterior orbit. Note the ensheathment of the optic nerve and the crowding of the orbital apex. (From Weber AL, Romo LV, Sabates NR. Pseudotumor of the orbit. Radiol Clin North Am 1999;37: 151–168.)
Ultrasonography
Ultrasound in both A and B mode can help in the diagnosis of scleritis and differentiation of orbital mass structures. Posterior scleritis with effusion at Tenon’s capsule can create the “T-sign.”13
Laboratory Studies
Depending on the clinical presentation, especially if atypical, some laboratory tests may be helpful. These include complete blood count (CBC) with differential, treponemal tests [microhemagglutination of Treponema pallidum (MHA-TP)], erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), fasting serum glucose (rule out diabetes and potential mucormycosis), anti-neutrophil cytoplasmic antibodies (ANCA), and angiotensin-converting enzyme (ACE). Based on the initial workup, more directed laboratory studies may be warranted, as might radiographic analysis of other body regions.
Orbital Biopsy
Biopsy, either by direct visualization or by needle, should be considered if the presentation is atypical or if response to treatment is incomplete. Any past history of neoplasia heightens the need for histopathology. If mucormycosis is suspected, biopsy is of paramount importance.
Biopsy results depend on the site selected and the stage of disease. In early disease, edema and a heterogeneous mild inflammatory infiltrate are typically seen, composed of lymphocytes, plasma cells, eosinophils, and sometimes polymorphonuclear cells.2Eosinophils are more prominent in children and can even be elevated in the blood.14 With disease progression, both the inflammatory component and the fibrotic component increase, with cells becoming separated by strands of collagen radiating from the orbital septa and vessels, into the fat. Lymphoid follicles, usually multicentric, are seen in the more chronic forms of NSIOI. Perivascular lymphocytic cuffing is common and does not represent vasculitis. With chronicity, more fibrosis occurs with gradual replacement of orbital structures including fat and muscle.
TREATMENT
Corticosteroids remain the mainstay of acute therapy, and can produce dramatic results in a matter of hours to days. Some authors utilize steroid failure as an indicator for alternative diagnostic workup. A typical regimen includes prednisone 80 to 100 mg by mouth daily for 2 to 3 weeks, with slow taper over the ensuing 3 weeks to prevent recurrence.2 Rootman1advocated taper over 2 to 3 months. Addition of gastric protective agents is important (i.e., ranitidine, 150 mg p.o. b.i.d.; or famotidine, 40 mg p.o qhs; or others) and should be maintained throughout steroid therapy. Steroids are less successful in subacute or chronic cases where fibrosis is already a component.
Radiotherapy (1500 to 2000 cGy) is sometimes administered in refractory cases, with appropriate shielding of the globe.2 If no or poor response to radiation therapy occurs, biopsy or rebiopsy should be considered.
Additional topical steroid medication is sometimes used in fulminant cases of anterior NSIOI with uveitis, depending on severity of the inflammation. Prednisolone acetate 1% applied every 1 to 6 hours with or without a cycloplegic agent may provide local therapy and relief from ciliary spasm. Newer drugs, including topical cyclosporine, may be indicated in chronic cases.
Glaucoma attendant with NSIOI, whether due to ocular inflammation, or ocular compression, should be treated accordingly.
FOLLOW-UP AND REFERRAL
Depending on the reliability of the patient, initial follow-up evaluation should be performed in 3 to 7 days to check for treatment response. Worsening of symptoms after 48 hours of treatment should be reported to the physician, and other causes, especially infectious, entertained. Intraocular pressure monitoring to observe for steroid effects is important.
Recurrence is uncommon, but may indicate need for longer treatment, or alternative antiinflammatory or immunosuppressive drugs. If there is not a dramatic response to initial therapy, referral to an orbital disease specialist can be helpful.
IDIOPATHIC SCLEROSING INFLAMMATIONS OF THE ORBIT
Although some authors have included idiopathic sclerosing inflammation of the orbit (ISIO) as a subset of idiopathic orbital inflammation, such classification is probably inappropriate.2 Rootman et al3 have argued that ISIO represents a unique clinicopathologic entity that is related to other systemic multifocal fibrosclerosing processes such as retroperitoneal fibrosis, mediastinal fibrosis, sclerosing cholangitis, Reidel’s fibrous thyroiditis, and pachymeningitis. ISIO is manifest as a chronic, primarily fibrotic process, with mild inflammation, which is immunologically mediated. It is relatively uncommon, occurring in only 5% of patients with orbital inflammations of nonthyroid designation.1 Most disease begins in the lacrimal region, with 20% beginning in apical orbital locations. Diffuse orbital and myositic subtypes have been encountered. As disease progresses, sclerosis becomes more diffuse, and may spread intracranially, or into the pterygoid fossa.
URGENCY OF EVALUATION
The clinical onset of disease is subacute or chronic. However, because early treatment has been said to decrease further sclerosis, evaluation should proceed in a relatively short time period. If the patient is diabetic or otherwise immunocompromised, evaluation should proceed urgently in order to rule out mucormycosis.
DIAGNOSIS
Demographics
Idiopathic sclerosing inflammation of the orbit occurs in the same population as other idiopathic inflammatory orbital processes; namely, young adults to those of elderly age. There is no predilection for gender or ethnicity.
Symptoms
Pain
Patients may complain of a low-grade, dull pain localized around the orbit or orbits, which fluctuates little over time.
Diplopia
Double vision is more common in later stages of the disease after significant fibrosis has restricted extraocular muscle movements. In some patients, despite severe ocular restriction, no diplopia is noted, possibly due to unilateral or alternating visual suppression.
Vision Loss
Loss of acuity and visual field are not uncommon and suggest compression or stretching of the optic nerve and need for aggressive treatment.
Signs
Proptosis
Proptosis is variable and dependent on the degree of sclerosis. In some patients, the degree of proptosis may be considerable. Associated resistance to ocular retropulsion is present.
Ptosis
Lid droop may be seen with involvement of the superior orbit/levator palpebrae region. Mass effect on the lid can also cause ptosis.
Lid Swelling
When anterior fibrosis adjacent to the lids occurs, lids may swell, but are usually without significant erythema, and there is never induration or signs of orbital cellulitis.
Conjunctival Injection
Episcleral vessels may be injected on the affected side, but chemosis is less prominent than in nonsclerosing inflammatory variants. Significant conjunctival injection suggests possible carotid-cavernous fistulas.
Restriction of Ocular Movement
Depending on the degree and location of involvement, restriction of extraocular muscles occurs. In severe cases this can present as a “frozen globe.”
Red Flags
The following features should raise the question of alternative diagnoses
 Significant lid retraction. This is more common with thyroid eye disease.
Significant lid retraction. This is more common with thyroid eye disease.
 History of previous cancer. Orbital metastasis, including bilaterally, is not uncommon for lung, breast, bowel, prostate, and some other types of cancer.
History of previous cancer. Orbital metastasis, including bilaterally, is not uncommon for lung, breast, bowel, prostate, and some other types of cancer.
 History of diabetes or other form of immunocompromise. Emergent evaluation for possible mucormycosis should ensue.
History of diabetes or other form of immunocompromise. Emergent evaluation for possible mucormycosis should ensue.
 History of previous systemic vascular disease such as Wegener’s granulomatosis.
History of previous systemic vascular disease such as Wegener’s granulomatosis.
 Fever. Suggests possible infection.
Fever. Suggests possible infection.
Differential Diagnosis
The differential diagnosis includes thyroid ophthalmopathy, sarcoidosis, Wegener’s granulomatosis, orbital infections including fungal and mycobacterial, Erdheim-Chester disease (especially if bilateral), meningioma, fulminant sinus disease with spread, sinus neoplasia with spread, lymphoma.
ANCILLARY TESTS
Neuroimaging
Because of the possibility of intracranial extension or meningeal involvement, neuroimaging of both the brain and orbits is absolutely required. This should be performed in both axial and coronal planes (digital reconstruction if not available), and both with and without contrast medium. CT should be ordered initially due to the improved visualization of bone and paranasal sinuses.2 Subsequent MRI is often ordered to better visualize the brain, cavernous sinus, and some soft tissue components of the orbit. MRI should be ordered with additional fat-suppression techniques.
Imaging of the orbit frequently reveals homogeneously enhanced areas of opacification with ragged margins. There may be opacification of extraocular muscles or the lacrimal gland. Posterior infiltration may show stretch of the optic nerve. In severe disease, there may be total wall-to-wall opacification of all orbital structures except the globe.
Laboratory Studies
When the diagnosis is not clear, additional tests might include CBC with differential, ANCA, ANA, thyroid-stimulating hormone (TSH), MHA-TP, serum protein electrophoresis (SPEP), purified protein derivative (PPD) with controls, chest x-ray, ACE, and other directed tests based on results of the workup.
Orbital Biopsy
Because of the extent of the differential diagnosis, and the overlap of clinical and physical characteristics, biopsy of orbital structures is common in this entity. Representative sample should be obtained and a portion of the biopsy sent in saline (i.e., no fixative) for flow cytometric and/or molecular analysis. Pathology usually shows a paucicellular chronic inflammatory infiltrate with significant desmoplastic stroma that infiltrates and obliterates orbital fat and muscles. Inflammatory cells include lymphocytes, plasmacytes, histiocytes, and occasional lymphoid follicles. Eosinophils are not usually seen.
TREATMENT
In the past, as in other NSIOI, sclerosing inflammation has been treated primarily with steroids, with radiation therapy and chemotherapy comprising second-line approaches. However, discouraging results are often obtained, with up to 30% of patients progressing despite treatment of any sort. Because of this, Rootman and colleagues3 have advocated an aggressive approach aimed at early diagnosis and treatment with multiple immunosuppressive agents. Systemic prednisone has been combined with drugs directed against B cells (methotrexate, cyclophosphamide, azathioprine, depending on the age of the patient) and/or against T-cells (cyclosporine). Management with these agents is best accomplished along with a rheumatologist and an oncologist or chemotherapist. Disease arrest without further progression has been reported using this combined approach.1 Resolution of fibrosis already present prior to therapy, however, has not been reported. Occasionally surgical debulking or orbital exenteration has been utilized to stem aggressive disease and intracranial extension.4
FOLLOW-UP AND REFERRAL
Because of the aggressive and relentless nature of idiopathic sclerosing inflammation of the orbit, and because of the need for multidisciplinary management of treatment paradigms, most patients should be referred to an orbital specialist who will coordinate the approach.
NONSPECIFIC ORBITAL INFLAMMATORY MYOSITIS
Orbital myositis is one of the most common NSIOIs, and involves idiopathic inflammation primarily of the extraocular muscles. It is differentiated from the more anterior, posterior, or diffuse subsets of NSIOI discussed previously. Fifty percent of patients with orbital myositis possess underlying immunologic abnormalities.1 Other systemic associations include prior upper respiratory tract infections,15 antecedent flu-like illness, sinusitis, allergic rhinitis, asthma,16 and influenza vaccination.17
URGENCY OF EVALUATION
Urgency of evaluation is linked to the severity of the presenting symptoms and their developmental course. If pain or diplopia is extreme, a more rapid evaluation is necessary. If symptom presentation is subacute, the patient should be seen soon, but not necessarily urgently. If there is any associated visual acuity or field loss (indicating additional anterior, posterior, or diffuse orbital involvement), the patient should be seen urgently. The patient with diabetes or who is otherwise immunocompromised should be seen emergently to rule out mucormycosis.
DIAGNOSIS
Demographics
Myositis can occur anytime from adolescence to elderly age, with no gender or ethnic predilection.
Symptoms
Diplopia
Fifty percent of patients present with double vision of acute or subacute onset. There are cases with chronic development of symptoms (weeks to months).
Pain
Most patients present with periorbital and retrobulbar pain of sudden onset (over hours to days) that is worsened by extraocular movement. Subacute and chronic forms have also been reported.
Vision Loss
Visual changes are a rare, but concerning manifestation of myositis and indicate possible posterior orbital muscle inflammation or posterior apex involvement. Patients with vision loss should be evaluated and treated quickly.
Signs
Extraocular Muscle Restriction
The movement of the affected extraocular muscle is usually reduced in the appropriate field of action, and forced ductions are often positive. Muscle restriction may result in retraction of the globe on attempted gaze opposite the side of the involved muscle.18 Any extraocular muscle, vertical or horizontal, can be affected in equal proportion.1 Recurrent disease is more likely to involve multiple muscles and is often bilateral.
Proptosis
Proptosis is frequently present, but often not pronounced unless multiple muscles are involved. There is usually resistance to retropulsion of the globe, sometimes with associated pain.
Conjunctival Chemosis and Injection
Injection of episcleral and conjunctival vessels with associated chemosis may be seen overlying the insertion of the involved muscle or muscles. Significant chemosis or injection, however, should be a red flag for possible infection.
Red Flags
The following features should raise the question of alternative diagnoses:
 Significant lid retraction. This is more likely a feature of thyroid ophthalmopathy.
Significant lid retraction. This is more likely a feature of thyroid ophthalmopathy.
 Significant ophthalmoparesis. This may be related to orbital ischemic syndrome, carotid cavernous fistula, or fulminant infection.
Significant ophthalmoparesis. This may be related to orbital ischemic syndrome, carotid cavernous fistula, or fulminant infection.
 Significant chemosis or conjunctival injection. As in number 2.
Significant chemosis or conjunctival injection. As in number 2.
 Significant ptosis without lid swelling. This may relate to myasthenia gravis or more widespread anterior NSIOI.
Significant ptosis without lid swelling. This may relate to myasthenia gravis or more widespread anterior NSIOI.
 History of previous cancer. Metastatic disease to extraocular muscles has been reported.
History of previous cancer. Metastatic disease to extraocular muscles has been reported.
 Visual loss. Although myositis with involvement of the posterior portion of the muscles can cause optic nerve compression and vision loss, multiple other causes may be involved including posterior NSIOI, infection, or neoplasia. Occasionally lymphoproliferative processes can mimic posterior myositis.
Visual loss. Although myositis with involvement of the posterior portion of the muscles can cause optic nerve compression and vision loss, multiple other causes may be involved including posterior NSIOI, infection, or neoplasia. Occasionally lymphoproliferative processes can mimic posterior myositis.
 Young age at onset. Consideration for acute presentation of rhabdomyosarcoma is of paramount importance.
Young age at onset. Consideration for acute presentation of rhabdomyosarcoma is of paramount importance.
 Fever. Suggests infectious causes.
Fever. Suggests infectious causes.
Differential Diagnosis
Differential diagnosis includes thyroid ophthalmopathy; carotid-cavernous fistula; myasthenia gravis; Tolosa-Hunt syndrome; lymphoma; orbital metastasis; primary extraocular muscle tumors; amyloidosis; sarcoidosis; systemic lupus erythematosus; Wegener’s granulomatosis; orbital infection including fungus, spirochetes, and parasites; and other nonspecific orbital inflammation syndromes.
ANCILLARY TESTS
Neuroimaging
To rule out other orbital processes, some of which may involve other central nervous system (CNS) regions, neuroimaging of both the brain and orbits is absolutely required. This should be performed in both axial and coronal planes (coronal digital reconstruction if not available directly), and both with and without contrast medium. CT is generally ordered initially due to the additional visualization of bone and somewhat improved images of the paranasal sinuses.2 Subsequent MRI is often also ordered for better visualization of the brain, cavernous sinus, and some soft tissue components of the orbit. MRI should be ordered with additional fat-suppression techniques.
CT images usually demonstrate the characteristic diffuse enlargement of the entirety of the affected extraocular muscle, including the tendon (as opposed to thyroid ophthalmopathy, which typically spares the tendon). Orbital tissue adjacent to the muscle may be locally infiltrated showing a ragged enhancement on contrast CT adjacent to the muscle substance.
Laboratory Studies
In atypical cases, or where the diagnosis is in doubt, ancillary lab tests might include CBC with differentiation, ESR, CRP, TSH, ANA, ANCA, ACE, acetylcholine receptor antibodies (binding, blocking, and modulating forms), SPEP, MHA-TP, and Lyme Western blot.19
Orbital Biopsy
If there is no response to treatment or if the diagnosis is in doubt, a biopsy of extraocular muscle and associated orbital tissue should be considered. Needle biopsy is not usually adequate in this circumstance. Attempts should be made to obtain an affected portion of muscle. The pathologic characteristics of myositis are similar to those of other forms of nonspecific idiopathic orbital inflammation, namely, a polymorphous infiltrate of mixed lymphocytic, plasmocytic, eosinophilic, and less commonly polymorphonuclear cells. Histopathologic differentiation from thyroid ophthalmopathy can be difficult, but eosinophilia is not usually present in the latter.2 With chronicity, variable amounts of fibrosis can be present with replacement of muscle fibers.
TREATMENT
As in other forms of NSIOI, corticosteroids remain primary in treatment of myositis. Single muscle involvement may require less hefty doses of steroids (prednisone, 40–60 mg p.o. per day tapered on a symptomatic basis). Alternatively, treatment with nonsteroidal antiinflammatory drugs (NSAIDs) has been advocated, or even observation if disease is on the wane.1 Disease in such patients usually improves rapidly with treatment, and is monophasic. If disease is bilateral or if multiple muscles are involved, higher doses of prednisone are indicated (prednisone 80–100 mg p.o. per day for 2 to 3 weeks). Recurrence and refractivity are more common in subacute and chronic presentations, requiring longer treatment periods with slower taper protocols over 4 to 6 weeks.1 Pulse intravenous steroids have been advocated in extreme cases including apical orbital disease with visual loss (IV methylprednisolone 250–1000 mg over 1 to 2 hours every month or every 2 months, changing to orals with taper based on symptomatic response). Appropriate gastric protection should be provided to patients on steroids via an H-2 blocker (ranitidine, cimetidine) or proton pump inhibitor (omeprazole, rabeprazole, lansoprazole).
Radiotherapy has been advocated by some in recalcitrant cases. Considerable rates of recurrence have occurred in these patients, however,20 prompting suggestions that higher radiation dosage might be required.21
FOLLOW-UP AND REFERRAL
Patients should be seen in follow-up within 3 to 7 days after onset of therapy, to evaluate response. Any worsening of symptoms during this period should be reported to the physician immediately, and infectious causes should be evaluated, including mucormycosis in diabetic or immunocompromised patients. Intraocular pressure should be monitored, as some patients develop steroid-induced glaucoma. Subsequent follow-up should be based on response to therapy. If there is no response to therapy, referral to an orbital specialist should be considered.
NONSPECIFIC INFLAMMATORY DACRYOADENITIS
In dacryoadenitis, the primary location of nonspecific idiopathic orbital inflammation is in the lacrimal gland. Because there is normally a small resident population of lymphocytes found at lacrimal ductules, rapid development of lacrimal inflammation in part arising from this locus is not surprising. Dacryoadenitis is the second most common presentation of NSIOI, following only myositis in frequency.1 Evolution of symptoms can be acute, subacute, or chronic. Fifty percent of patients presenting with an enlarged lacrimal gland do not have true nonspecific inflammatory disease but are likely to have an associated systemic disorder, especially if the course is chronic. Close attention to historical, physical, radiologic, and pathologic characteristics helps discern the true nature of the lesion.
URGENCY OF EVALUATION
Urgency of evaluation is linked to the severity of the presenting symptoms. If lacrimal gland swelling is particularly painful, or if there is induration or fever, more rapid evaluation should follow to rule out infection. Although chronic presentation is not urgent, evaluation and workup should proceed directly in order to rule out tumor.
DIAGNOSIS
Demographics
Dacryoadenitis can occur at any age from young adulthood to an elderly age. There is no predilection for disease by gender or race.
Symptoms
Pain
In acute dacryoadenitis, there is sudden onset of pain and tenderness over the superotemporal region of the orbit overlying the lacrimal gland. If inflammation has additionally involved the lateral rectus, there may be pain on extraocular movement.
Diplopia
Diplopia is not usually a presenting symptom unless concomitant involvement of the lateral rectus is present.
Signs
Lid Swelling and Erythema
The temporal portion of the upper lid on the affected side is frequently swollen and red. The region can sometimes feel warm, but is without induration or other signs of orbital cellulitis. A typical S-shape deformity of the lid is produced, sometimes resulting in ptosis.
Enlarged Lacrimal Gland
The palpebral lobe of the lacrimal gland is frequently enlarged enough to be easily seen in the temporal conjunctival fornix, and may be tender to palpation.
Conjunctival Injection and Chemosis
Lateral regions of the bulbar conjunctiva adjacent to the lacrimal gland may be injected and chemotic. This will be more prominent if the lateral rectus is also involved. Chemosis and injection of the conjunctiva distant from the temporal region suggests more widespread involvement, an infection, or arteriovenous fistulae.
Proptosis
Proptosis is usually minimal. There may be slight downward and inward displacement of the globe.
Extraocular Muscle Restriction
The lateral rectus is not usually restricted, but may show slight decrease in range of function if additionally involved. Significant muscle restriction indicates a myositic component.
Red Flags
The following features should raise the question of alternative diagnoses:
 Bilateral presentation. This may suggest a lymphoproliferative process or associated systemic disease such as Sjögren’s syndrome, Crohn’s disease, or sarcoid.
Bilateral presentation. This may suggest a lymphoproliferative process or associated systemic disease such as Sjögren’s syndrome, Crohn’s disease, or sarcoid.
 Significant conjunctival chemosis and injection. Attention should be directed to possible orbital cellulitis or carotid-cavernous fistulas.
Significant conjunctival chemosis and injection. Attention should be directed to possible orbital cellulitis or carotid-cavernous fistulas.
 Significant proptosis. Lacrimal gland inflammation usually molds to the globe in the orbit without much mass effect. Prominent proptosis or displacement of the globe suggests neoplasia, mucocele, or other noninflammatory processes.
Significant proptosis. Lacrimal gland inflammation usually molds to the globe in the orbit without much mass effect. Prominent proptosis or displacement of the globe suggests neoplasia, mucocele, or other noninflammatory processes.
 History of previous cancer. Metastatic disease to the lacrimal gland is possible.
History of previous cancer. Metastatic disease to the lacrimal gland is possible.
 Bone erosion on CT. This is more indicative of neoplasia.
Bone erosion on CT. This is more indicative of neoplasia.
 Fever. This suggests orbital cellulitis.
Fever. This suggests orbital cellulitis.
 Visual loss. This suggests more fulminant inflammation, or addition ocular or posterior orbit processes.
Visual loss. This suggests more fulminant inflammation, or addition ocular or posterior orbit processes.
 Severe pain and/or numbness. This raises the question of perineural invasion by a malignant process.
Severe pain and/or numbness. This raises the question of perineural invasion by a malignant process.
Differential Diagnosis
Differential diagnosis includes specific inflammatory processes such as sarcoidosis; infections including bacterial, mycobacterial, spirochetal, or viral infection; lymphoma; Sjögren’s syndrome; Wegener’s granulomatosis; ruptured dermoid cyst; or lacrimal gland involvement in systemic autoimmune disease such as Crohn’s disease.22 Historical, physical, and radiographic, and biopsy findings can usually help differentiate these diseases from typical inflammatory dacryoadenitis.
ANCILLARY TESTS
Neuroimaging
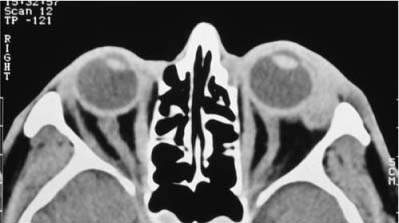
Radiologic evaluation of the orbit is absolutely necessary in the evaluation of lacrimal gland lesions. CT scan with and without contrast should be ordered in axial and coronal planes, with thin cuts. MRI may add information, especially in atypical cases. CT characteristics of nonspecific inflammatory dacryoadenitis include an oblong enlargement of the lacrimal gland, which molds to the curvature of the globe as it extends posteriorly (Fig. 40–4). Primary epithelial tumors of the lacrimal gland will have a more rounded appearance. The gland may appear in a V shape if the orbital lobe and palpebral lobe are involved.2 The bony orbit is normal, and if erosion is seen, then a neoplasm is likely. Occasionally infiltration of the lateral rectus is seen.
Ultrasound
Both A- and B-mode ultrasound investigation can be useful in differentiating lacrimal gland processes.1The lacrimal gland shows enlargement with internal reflectivity. Echolucency is apparent anteriorly near the sclera if the lateral rectus is also involved.
Laboratory Studies
In typical cases, laboratory tests are not usually helpful. In atypical cases, consideration should be given to CBC with differential, ANA, ANCA, ACE, chest x-ray, MHA-TP, SPEP, ESR, CRP, mumps immunoglobulin M (IgM) titer, and other tests as suggested by the workup.
FIGURE 40–4 CT scan of nonspecific idiopathic inflammation of the lacrimal gland on the left. Note the mild proptosis and thickening of anterior orbital structures. (From Weber AL, Romo LV, Sabates NR. Pseudotumor of the orbit. Radiol Clin North Am 1999;37:151–168.)
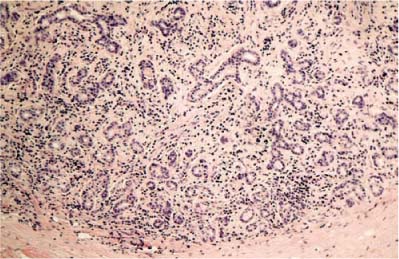
FIGURE 40–5 Photomicrograph of nonspecific idiopathic inflammation of the lacrimal gland. Note the destruction of acini. There is a chronic inflammatory infiltrate present composed of lymphocytes, plasma cells, and eosinophils (H&E original magnification 40X).
Lacrimal Gland Biopsy
Lacrimal gland biopsy is necessary because of the overlap in symptoms caused by lacrimal gland inflammation, lacrimal gland neoplasia, and gland enlargement secondary to systemic processes. Care should be taken to obtain a representative sample, which may require percutaneous anterolateral orbitotomy. This approach has the added benefit of avoiding the proximal lacrimal ducts. A sample large enough to allow for flow cytometric or molecular analysis of the inflammatory cells will help diagnose lymphoma. In this case, the pathologist should be consulted prior to biopsy and a portion submitted in saline without delay. Needle biopsy is usually not sufficient for diagnosis. Biopsy results typically show the same inflammatory infiltrate as seen in other NSIOI (see previous), with associated fibrosis and loss of lacrimal acini, depending on chronicity (Fig. 40–5).
TREATMENT
As with other NSIOIs, corticosteroids are the primary treatment for inflammatory dacryoadenitis. Moderate dosage of prednisone, 40 to 60 mg by mouth every morning should be instituted initially.1 To prevent relapse, taper should take place over 1 to 3 months depending on symptom expression, decreasing by 5 to 10 mg per week. As always, gastric protection with H-2 blockers or proton pump inhibitors is helpful in reducing ulceration (see previous). Although NSAIDs have been advocated by some, the recurrence rate in NSAID treated patients is high compared with steroids.23 In refractory patients, low-dose radiation and surgical debulking have been performed.24,25 These measures are seldom necessary, as response to corticosteroids is typically rapid.
FOLLOW-UP AND REFERRAL
Initial follow-up should occur within 3 to 7 days after treatment initiation, with strict directions to the patient to contact the physician immediately should the symptoms worsen (an indication of possible infection). Intraocular pressure should be checked while the patient is on steroids. Patients who do not respond to treatment may benefit from referral to an orbital specialist.
SPECIFIC INFLAMMATIONS OF THE ORBIT
The specific inflammations of the orbit can be best understood if divided into three categories: (1) inflammations associated with known and identifiable pathogens (infection); (2) inflammations with a specific constellation of findings (either locally or systemically); and (3) inflammations with specific histopathologic patterns.1
ORBITAL INFLAMMATION CAUSED BY INFECTION
Recognition of orbital infection is critical, as some infections can cause rapid and devastating disease with considerable morbidity and mortality. For the immunocompromised host, orbital infections are particularly hazardous. The proximity of the orbit to the brain and the contiguity of orbital structures with intracranial structures provide a readily accessible scaffold for intracranial extension. Evaluation of possible orbital infection, therefore, is urgent.
Infections of the orbit run the gamut of the microbe world including bacterial, viral, fungal, and parasitic. Interestingly, true viral infections of orbital tissues, even in immunocompromised hosts, have not been described outside of viral-induced dacryoadenitis following mumps or upper respiratory infections.2Hence viral infections are not discussed here.
ORBITAL CELLULITIS
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree